Abstract
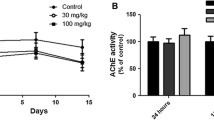
In order to better understand the effects of repeated low-dose exposure to organophosphorus (OPs) on physiological and behavioural functions, we analysed the levels of endogenous monoamines (serotonin and dopamine) in different brain areas after repeated exposure of mice to sublethal dose of soman. Animals were injected once a day for 3 days with 0.12 LD50 of soman (47 μg/kg, i.p.). They did not show either severe signs of cholinergic toxicity or pathological changes in brain tissue. 24 h after the last injection of soman, inhibition of cholinesterase was similar in plasma and brain (32% and 37% of inhibition respectively). Afterwards, recovery of cholinesterase activity was faster in the plasma than in the brain. Dopamine levels were not significantly modified. On the other hand, we observed a significant modification of the serotoninergic system. An increase of the 5-HIAA/5-HT ratio was maintained for 2 and 4 weeks after exposure in the hippocampus and the striatum respectively. This study provides the first evidence of a modification of the 5-HT turnover in the hippocampus and the striatum after repeated low-dose intoxication with a nerve agent. Further experiments are necessary to evaluate the relationship between these modifications and the unexpected neuropsychological disorders usually reported after chronic exposure of organophosphorus.

Similar content being viewed by others
References
Taylor P (2001) Anticholinesterase agents. In: The pharmacological basis of therapeutics, 10th edn. McGraw-Hill, New-York, pp 175–191
McDonough JH, Shih TM (1997) Neuropharmacological mechanisms of nerve agent induced seizure and neuropathology. Neurosci Biobehav Rev 21:559–579
Shih TM, Duniho SM, McDonough JH (2003) Control of nerve agents-induced seizures is critical for neuroprotection and survival. Toxico Appl Pharmacol 188:69–90
Pope C, Karanth S, Liu J (2005) Pharmacology and toxicology of cholinesterase inhibitors: uses and misuses of a common mechanism of action. Environ Toxicol Pharmacol 19:433–446
Rosenstock L, Keifer M, Daniell WE et al (1991) Chronic central nervous systems effects of acute organophosphate pesticide intoxication. Lancet 338:223–227
Stephens R, Spurgeon A, Calvert IA et al (1995) Neuropsychological effects of long-term exposure to organophosphates in sheep dip. Lancet 345:1135–1139
Russel RW, Booth RA, Lauretz SD et al (1986) Behavioral, neurochemical and physiological effects of repeated exposure to subsymptomatic levels of the anticholinesterase, soman. Neurobehav Toxicol Teratol 8:675–685
Raslear TG, Kaufman LW (1983) Diisopropyl phosphorofluoridate (DFP) disrupts circadian activity patterns. Neurobehav Toxicol Teratol 5:407–411
Kant GJ, Kenian CC, Meyenhoff BL (1984) Effects of diisopropylfluorophosphate (DFP) and other cholinergic agents on release of endogenous dopamine from rat brain striatum in vitro. Biochem Pharmacol 33:1823–1825
Ellman G, Courney KD, Andres V et al (1961) A new and rapid colorimetric determination of acetylcholinesterase activities. Biochem Pharmacol 7:88–95
Bielavska M, Kassa J (2000) Simultaneous determination of dopamine, serotonin, and their metabolites in the rat brain by HPLC method with colourometric detection. Collect Czech Chem Commun 65:1677–1682
Perrier NA, Salani M, Falasca C et al (2005) The readthrough variant of acetylcholinesterase remains very minor after heat shock, organophosphate inhibition and stress, in cell culture and in vivo. J Neurochem 94:629–638
Wolthuis O, Benshop HP, Berends F (1981) Persistence of the anti-cholinesterase soman in rats, antagonism with a non-toxic simulator of this OP. Eur J Pharmacol 69:379–383
Greenfield SA (1984) Acetylcholinesterase may have novel functions in the brain. Trends Neurosci 7:364–368
Raush JL, Sanowsky DS, Rich SC et al (1985) Physostigmine effects on serotonine uptake in human blood. Eur J Pharmacol 109:91–96
Blier P, de Montigny C (1994) Current advances and trends in the treatment of depression. TIPS 15:220–225
Murphy DL, Wichems C, Li Q et al (1999) Molecular manipulations as tools for enhancing our understanding of 5-HT1A neurotransmission. TIPS 20:246–251
Mayoshi M, Shirai Y, Bandoh T et al (2006) Alteration of 5-HIAA levels in frontal cortex and dorsal raphe nucleus in rats treated with combined administration of tryptophan and ethanol. J Toxicol Sci 31:235–246
Aldridge JE, Meyer A, Seidler FJ et al (2005) Alterations in central nervous system serotoninergic and dopaminergic synaptic activity in adulthood after prenatal or neonatal chlorpyrifos exposure. Environ Health Perspec 113:1027–1031
Soblosky JS, Thurmond JB (1986) Biochemical and behavioural correlates of chronic stress: effects of tricyclic antidepressants. Pharmacol Biochem Behav 24:1361–1368
Adell A, Trulas R, Gelpi E (1988) Time course in serotonin and noradrenaline in rat brain after predictable or unpredictable shock. Brain Res 459:54–59
Miura H, Naoi M, Nakamura D et al (1993) Changes in monoamine levels in mouse brain elicited by forced swimming stress and the protective effect of a new monoamine oxidase inhibitor. J Neural Trans 94:175–189
Taysse L, Christin D, Delamanche S et al (2005) Peripheral ChE inhibition modulates brain monoamine levels and c-fos oncogene in mice subjected to a stress situation. Neurochem Res 30:391–402
Sunanda BS, Shankaranarayana R, Raju TR (2000) Restraint stress-induced alterations in the levels of biogenic amines, amino acids and AChE activity in the hippocampus. Neurochem Res 25:1547–1552
Malyszko J, Urano T, Takada Y, Takada A (1994) Serotoninergic systems in brain and blood under stress and tranycypromine treatment in rats. Brain Res Bull 35:9–13
Nishi M, Azmitia EC (1996) 5-HT1A receptor expression is modulated by corticosteroid receptor agonist in primary rat hippocampal culture. Brain Res 722:190–194
Sevaljevic L, Marinkovic S, Bogojevic D et al (1989) Soman intoxication-induced changes in serum acute phase protein levels, corticosterone concentration and immunosuppressive potency of the serum. Arch Toxicol 63:406–411
Acknowledgements
This work was supported by grants from the Délégation Générale pour l’Armement. We wish to thank G. Guillot, P. Desforges, S. Morio and H. Cocher for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Christin, D., Daulon, S., Delamanche, S. et al. Effects of Repeated Low-dose Soman Exposure on Monoamine Levels in Different Brain Structures in Mice. Neurochem Res 33, 919–926 (2008). https://doi.org/10.1007/s11064-007-9535-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-007-9535-2