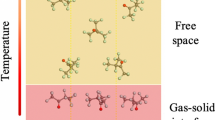
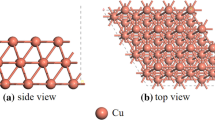
Abstract
In gas-insulated equipment, the decomposition of SF6 is closely related to the residual traces of O2 and H2O in the equipment. However, the decomposition mechanism has not yet been clarified. In this paper, the adsorption and defluorination of SF6 on the preadsorbed Cu(111) surface were calculated based on density flooding theory and transition state theory. Besides, the influence mechanism of O atoms and H2O molecules on the defluorination process is analyzed by comparing the energy barrier, reaction heat, and density of states. The results show that pre-adsorption of O atoms changes the adsorption sites of SFx, but has no significant effect on the adsorption energy. In addition, the O atom has a certain inhibitory effect on the decomposition process, and SF → S + F is the key to determining the reaction rate. In contrast, H2O will not only promote the adsorption of SFx to the surface but also reduce the total reaction heat of the decomposition reaction by 135.37 kcal·mol−1, driving the decomposition process of SF6. In this paper, stable co-adsorption configurations of low-fluorosulfide and co-adsorption groups were determined, and the effects of O and H2O preadsorption on SF6/Cu gas–solid interactions were initially revealed.









Similar content being viewed by others
Data Availability
The data that support the findings of this study are available upon reasonable request from the authors.
References
Chu FY (1986) SF6 decomposition in gas-insulated equipment. IEEE Trans Electr Insul EI-21:693–725
Ma J, Zhang Q, You H, Wu Z, Wen T, Guo C, Wang G, Gao C (2017) Study on insulation characteristics of GIS under combined voltage of DC and lightning impulse. IEEE Trans Dielectr Electr Insul 24:893–900
Zargar ZH, Akram KJ, Biswal GR, Islam T (2021) A linear polar sensor for ppm hydro measurement in SF6 gas-insulated switchgear. IEEE Trans Instrum Meas 70:1–8
Zeng FP, Li HT, Cheng HT, Liu YL (2021) SF6 decomposition and insulation condition monitoring of GIE: a review. High Volt 6(6):955–966
Zeng FP, Zhu KX, Feng XX, Li HT, Tang J, Zhang SL (2022) Adsorption characteristics of SF6 and its main over-thermal decomposition components on Ag (111) surface. IEEE Trans Dielectr Electr Insul 29(2):551–558
Tang J, Rao XJ, Zeng FP, Cai W, Chen L, Zhang CH (2017) Influence mechanisms of trace H2O on the generating process of SF6 spark discharge decomposition components. Plasma Chem Plasma Process 37:325–340
Zhong LP, Liu K, Ji SC, Wang F, Sun QQ, Chen S, Liu J (2020) Effects of voltage forms, pressure, and adsorbent on the SF6 decomposition characteristics under corona discharge. IET Sci Meas Technol 14(4):486–493
Cui ZL, Zhang XX, Yuan T, Pan XY, Luo Y, Tang J (2020) Plasma-assisted abatement of SF6 in a dielectric barrier discharge reactor: investigation of the effect of packing materials. J Phys D Appl Phys 53(2):025205
Yashdeep Y, Biswal GR, Choudhury T, Islam T, Mukhopadhyay SC, Vashisht V (2017) Design and modeling of MEMS-based trace-level moisture measurement system for GIS applications in smart grid environment. IEEE Sens J 17:7758–7766
Liu H, Wang J, Wang JR, Hu Q, Chang YN, Li QM (2020) Study on pyrolysis characteristics of SF6 in a trace-oxygen (O2) environment: ReaxFFSFO force field optimization and reactive molecular dynamics simulation. ACS Omega 5(41):26518–26526
Zeng FP, Li HT, Zhang MX, Li C, Yao Q, Tang J (2021) Establishment of a Reax force field to study SF6 gas over-thermal decomposition. J Phys D 54(11):115501
Zeng FP, Li HT, Zhang MX, Li C, Zhu KX, Tang J (2021) Kinetic analysis of the effect of O2 on SF6 over-thermal decomposition. J Phys D 54(49):495502
Li HT, Zeng FP, Zhang MX, Zhu KX, Yao Q, Wei G, Ma GM, Tang J (2022) Isotope tracing experiment on the mechanism of O2 on the over-thermal decomposition of SF6. Plasma Chem Plasma Process 42:505–518
Fu YW, Rong MZ, Yang K, Yang AJ, Wang XH, Gao QQ, Liu DX, Murphy AB (2016) Calculated rate constants of the chemical reactions involving the main byproducts SO2F, SOF2, SO2F2 of SF6 decomposition in power equipment. J Phys D Appl Phys 49(15):155502
Fu YW, Wang XH, Wang XX, Yang AJ, Rong MZ (2020) The decomposition pathways of SF6 in the presence of organic insulator vapors. Plasma Chem Plasma Process 40:449–467
Zhong LP, Ji SC, Wang F, Sun QQ, Chen S, Liu J, Hai B, Tang L (2019) Theoretical study of the chemical decomposition mechanism and model of sulfur hexafluorid (SF6) under corona discharge. J Fluor Chem 220:61–68
Ge JY, Dai JQ, Zhang JZH (1999) Dissociative adsorption of O2 on Cu(110) and Cu(100): three-dimensional quantum dynamics studies. J Phys Chem 100:11432–11437
Zhao J, Qin P, Fang T (2018) Mechanism of ammonia decomposition on clean and oxygen-covered Cu(111) surface: a DFT study. Phys Chem 8:055215
Jiang Z, Fang T (2016) Insight into the promotion effect of pre-covered X (C, N and O) atoms on the dissociation of water on Cu(111) surface: a DFT study. Appl Surf Sci 376:219–226
Bai MB, Chen J, Feng ZT (2022) Density functional theory study on the decomposition mechanism of HFC-32 on a Cu(111) surface: the impact of H2O and O2. J Mol Liq 345(15):118027
Zeng FP, Tang J, Fan QT, Pan JY, Zhang XX, Yao Q, He JJ (2014) Decomposition characteristics of SF6 under thermal fault for temperature below 400°C. IEEE Trans Dielectr Electr Insul 21(3):995–1004
Monterroso R, Fan MH, Argyle M (2011) Chapter 9—mercury removal. In: David AB, Brian FT, Fan MH (eds) Coal gasification and its applications. William Andrew Publishing, pp 247–292
Pisarev AA (2012) 1-Hydrogen adsorption on the surface of metals. In: Richard PG, Brian PS (eds) In Woodhead publishing series in metals and surface engineering, gaseous hydrogen embrittlement of materials in energy technologies, vol 1. Woodhead Publishing, pp 3–26
Wan YC, Xu JC, Lv RT (2019) Heterogeneous electrocatalysts design for nitrogen reduction reaction under ambient conditions. Mater Today 27:69–90
Delley B (2000) From molecules to solids with the DMol3 approach. J Chem Phys 113(18):7756–7764
Delley B (1990) An all-electron numerical method for solving the local density functional for polyatomic molecules. J Chem Phys 92(1):508–517
Hammer B, Hansen LB, Nørskov JK (1999) Improved adsorption energetics within density-functional theory using revised Perdew–Burke–Ernzerhof functionals. Phys Rev B 59:7413
Zhang Y, Yang W (1998) Comment on ‘‘Generalized gradient approximation made simple”. Phys Rev Lett 80:890
Perdew J, Burke K, Ernzerhof M (1996) Generalized gradient approximation made simple. Phys Rev Lett 77:3865
Delley B (2002) Hardness conserving semilocal pseudopotentials. Phys Rev B 66(15):155125
Monkhorst HJ, Pack JD (1976) Special points for Brillouin-zone integrations. Phys Rev B 13:5188
Huo E, Liu C, Xu X, Li Q, Dang C (2018) Dissociation mechanisms of HFO-1336mzz (Z) on Cu(111), Cu(1 1 0) and Cu(1 0 0) surfaces: a density functional theory study. Appl Surf Sci 443:389–400
Zhao J, Qin P, Fang T (2017) Decomposition mechanism of formic acid on Cu(111) surface: a theoretical study. Appl Surf Sci 396:857–864
Halgren TA, Lipscomb WN (1977) Chem Phys Lett 49:225
Zhao J, Fang T (2016) Dissociation mechanism of H2O on clean and oxygen-covered Cu(111) surfaces: a theoretical study. Vacuum 128:252–258
Houska J, Kozak T (2017) Relationships between the distribution of O atoms on partially oxidized metal (Al, Ag, Cu, Ti, Zr, Hf) surfaces and the adsorption energy: a density-functional theory study. J Appl Phys 121:225303
Zhang R, Duan T, Ling L, Wang B (2015) CH4 dehydrogenation on Cu(111), Cu@Cu (111), Rh@Cu(111) and RhCu(111) surfaces: a comparison studies of catalytic activity. Appl Surf Sci 341:100–108
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant No. 51877157) and the Hubei Province Science Fund for Distinguished Young Scholars (Grant No. 2020CFA097).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zeng, F., Kexin, Z., Su, D. et al. Influence Mechanism of O2/H2O Adsorption on Cu(111) Surface on SF6 Overheating Failure Decomposition. Plasma Chem Plasma Process 43, 67–80 (2023). https://doi.org/10.1007/s11090-022-10305-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-022-10305-8