Purpose
The aim of this study was to evaluate the utility of a parametric deconvolution method using a sum of inverse Gaussian functions (IG) to characterize the absorption and concentrations vs. time profile of drugs exhibiting complex absorption.
Methods
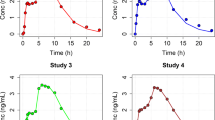
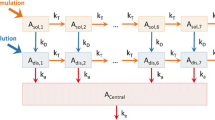
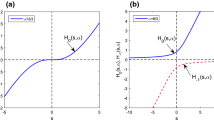
For a linear time-invariant system the response, Y(t), following an arbitrary input function I(t), is the convolution of I(t) with the disposition function, H(t), of the system: \(Y{\left( t \right)} = {\int\limits_0^t {I{\left( \tau \right)}H{\left( {t - \tau } \right)}d\tau } }\). The method proposed uses a sum of n inverse Gaussian functions to characterize I(t). The approach was compared with a standard nonparametric method using linear splines. Data were provided from previously published studies on two drugs (hydromorphone and veralipride) showing complex absorption and analyzed with NONMEM®.
Results
A satisfactory fit for hydromorphone and veralipride data following oral administration was achieved by fitting a sum of two or three IG functions. The predictions of the input functions were very similar to those using linear splines.
Conclusions
The use of a sum of IG as opposed to nonparametric functions, such as splines, offers a simpler implementation, a more intuitive interpretation of the results, a built-in extrapolation, and an easier implementation in a population context. Disadvantages are an apparent greater sensitivity to initial value estimates (when used with NONMEM®).





Similar content being viewed by others
References
L. F. Prescott (1974) ArticleTitleGastrointestinal absorption of drugs Med. Clin. N. Am. 58 5–17
D. Verotta (1996) ArticleTitleConcepts, properties and applications of linear systems to describe distribution, identify input, and control endogenous substances and drugs in biological systems Crit. Rev. Biomed. Eng. 24 73–139 Occurrence Handle9108983
C. DeBoor (1978) A Practical Guide to Spline Springer-Verlag New York
K. Ishii Y. Katayama S. Itai Y. Ito H. Hayashi (1995) ArticleTitleA new pharmacokinetic model including in vivo dissolution and gastrointestinal transit parameters Biol. Pharm. Bull. 18 IssueID6 882–886 Occurrence Handle7550125
Y. Plusquellec G. Campistron S. Staveris J. Barre L. Jung J. P. Tillement G. Houin (1987) ArticleTitleA double-peak phenomenon in the pharmacokinetics of veralipride after oral administration: a double-site model for drug absorption J. Pharmacokinet. Pharmacodyn. 15 IssueID3 225–239
R. Suverkrup (1979) ArticleTitleDiscontinuous absorption processes in pharmacokinetic models J. Pharm. Sci. 68 1395–1400 Occurrence Handle512886
K. Murata K. Noda K. Kohno M. Samejima (1987) ArticleTitlePharmacokinetic analysis of concentration data of drugs with irregular absorption profiles using multi-function absorption models J. Pharm. Sci. 76 109–113 Occurrence Handle3572746
K. Higaki S. Yamashita G. L. Amidon (2001) ArticleTitleTime-dependent oral absorption models J. Pharmacokinet. Pharmacodyn. 28 IssueID2 109–128 Occurrence Handle10.1023/A:1011573831444 Occurrence Handle11381566
R. L. Oberle G. L. Amidon (1987) ArticleTitleThe influence of variable gastric emptying and intestinal transit rates on the plasma level curve of cimetidine: an explanation for the double peak phenomenon J. Pharmacokinet. Biopharm. 15 539–544 Occurrence Handle10.1007/BF01061761
M. Weiss (1996) ArticleTitleA novel extravascular input function for the assessment of drug absorption in bioavailabilities studies Pharm. Res. 13 IssueID10 1547–1553 Occurrence Handle10.1023/A:1016039931663 Occurrence Handle8899849
K. E. Fattinger D. Verotta (1995) ArticleTitleA nonparametric subject-specific population method for deconvolution: I. Description, internal validation, and real data examples J. Pharmacokinet. Biopharm. 23 IssueID6 581–610 Occurrence Handle8733948
D. R. Drover M. S. Angst M. Valle B. Ramaswamy S. Naidu D. R. Stanski D. Verotta (2002) ArticleTitleInput characteristics and bioavailability after administration of immediate and a new extended-release formulation of hydromorphone in healthy volunteers Anesthesiology 97 827–836 Occurrence Handle10.1097/00000542-200210000-00013 Occurrence Handle12357147
A. J. Boeckmann S. L. Beal L. B. Sheiner (1992) NONMEM Users’ Guides NONMEM Project Group, University of California at San Francisco San Francisco
A. Akaike (1974) ArticleTitleA new look at the statistical model identification problem IEEE Trans. Automat. Contr. 19 716–723 Occurrence Handle10.1109/TAC.1974.1100705
E. J. Hannan (1987) ArticleTitleRational transfer function approximation Stat. Sci. 2 1029–1054
G. Schwartz (1978) ArticleTitleEstimating the dimension of a model Ann. Stat. 6 461–464
S-Plus Software, Statistical Sciences, Version 4.0 Release 2, 1997. http://www.statsci.org/splus.html.
P. Veng-Pedersen M. J. Berg D. D. Schottelius (1986) ArticleTitleLinear system approach for the analysis of an induced drug removal process. Phenobarbital removal by oral activated charcoal J. Pharmacokinet. Biopharm. 14 19–28 Occurrence Handle10.1007/BF01059201 Occurrence Handle3746631
H. Zhou (2003) ArticleTitlePharmacokinetic strategies in deciphering atyptical drug absorption profiles J. Clin. Pharmacol. 43 211–227 Occurrence Handle10.1177/0091270002250613 Occurrence Handle12638389
Acknowledgment
This study was supported by NIH Grant A150587.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Csajka, C., Drover, D. & Verotta, D. The Use of a Sum of Inverse Gaussian Functions to Describe the Absorption Profile of Drugs Exhibiting Complex Absorption. Pharm Res 22, 1227–1235 (2005). https://doi.org/10.1007/s11095-005-5266-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-005-5266-8