ABSTRACT
Purpose
The aim of this work was to develop anesthetic bioadhesive films containing benzocaine and study their in vitro skin permeation and in vivo performance, in comparison with commercial formulations.
Methods
Films containing 3% and 5% w/w of benzocaine were prepared and characterized by weight, drug content, thickness and morphology. In vitro permeation assays were performed in vertical diffusion cells using full-thickness pig ear skin as barrier. Intensity and duration of analgesia were evaluated in rats by tail-flick test, and skin histological analysis was carried out.
Results
Tail-flick test showed that the duration of benzocaine-induced analgesia was significantly prolonged with the films compared to commercial creams, in agreement with the higher in vitro permeation. Histological analysis of the rat tail skin did not reveal morphological tissue changes nor cell infiltration signs after application of the commercial creams or films.
Conclusions
Results from our study indicate that the films developed in this work can be considered as innovative dermal/transdermal therapeutic systems for benzocaine local delivery.






Similar content being viewed by others
REFERENCES
Harmatz A. Local anesthetics: uses and toxicities. Surg Clin North Am. 2009;89:587–98.
Butterworth JF, Strichartz GR. Molecular mechanisms of local anesthesia: a review. Anesthesiology. 1990;72:711–34.
Yanagidate F, Strichartz GR. Local anesthetics. Handb Exp Pharmacol. 2007;177:95–127.
Al-Melh MA, Anderson L. Comparison of topical anesthetics (EMLA/Oraqix vs. benzocaine) on pain experienced during palatal needle injection. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;103:16–20.
Roos TC, Merk HF. Allergic contact dermatitis from benzocaine ointment during tretment of herpes zoster. Contact Dermatitis. 2001;44:97–130.
de Padua CA, Schnuch A, Nink K, Pfahlberg A, Uter W. Allergic contact dermatitis to topical drugs-epidemiological risk assessment. Pharmacoepidemiol Drug Saf. 2008;17:813–21.
Young KD. What’s new in topical anesthesia. Clin Pediatr Emerg Med. 2007;8:232–9.
Shin SC, Lee KW, Yang CH. Preparation and evaluation of bioadhesive benzocaine gels for enhancement local anesthetic effects. Int J Pharm. 2003;260:77–81.
Mura P, Maestrelli F, Gonzalez-Rodriguez ML, Michelacci I, Ghelardini C, Rabasco AM. Development, characterization of benzocaine-loaded liposomes. Eur J Pharm Biopharm. 2007;67:86–95.
Maestrelli F, Capasso G, Gonzalez-Rodriguez ML, Rabasco AM, Mura P. Effect of preparation technique on the properties and in vivo efficacy of benzocaine-loaded ethosomes. J Liposome Res. 2009;4:1–8.
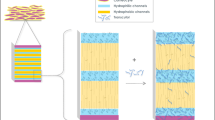
Padula C, Colombo G, Nicoli S, Catellani PL, Massimo G, Santi P. Bioadhesive film for the transdermal delivery of lidocaine: in vitro and in vivo behavior. J Control Release. 2003;88:277–85.
Padula C, Nicoli S, Aversa V, Colombo P, Falson F, Pirot F, et al. Bioadhesive film for dermal and transdermal drug delivery. Eur J Dermatol. 2007;17:309–12.
Nussinovitch A, Gal A, Padula C, Santi P. Physical characterization of a new skin bioadhesive film. AAPS PharmSciTech. 2008;9:458–63.
Padula C, Nicoli S, Colombo P, Santi P. Single-layer transdermal film containing lidocaine: modulation of drug release. Eur J Pharm Biopharm. 2007;66:422–8.
Nicoli S, Colombo P, Santi P. Release and permeation kinetics of caffeine from bioadhesive transdermal films. AAPS J. 2005;7:E218–23.
Nicoli S, Penna E, Padula C, Colombo P, Santi P. New transdermal bioadhesive film containing oxybutynin: in vitro permeation across rabbit ear skin. Int J Pharm. 2006;325:2–7.
Femenia-Font A, Padula C, Marra F, Balaguer-Fernandez C, Merino V, Lopez-Castellano A, et al. Bioadhesive monolayer film for the in vitro transdermal delivery of sumatriptan. J Pharm Sci. 2006;95:1561–9.
Nicoli S, Cella S, Aversa V, Santi P. Transdermal film containing nicotine and bupropion for combined smoking cessation therapy. Pharm Technol Eur. 2008;20:613–21.
Padula C, Nicoli S, Santi P. Innovative formulations for the delivery of levothyroxine to the skin. Int J Pharm. 2009;372:12–6.
Narang PK, Bird G, Crouthamel WG. High-performance liquid chromatographic assay for the benzocaine an p-aminobenzoic acid including preliminary stability data. J Pharm Sci. 1980;69:1384–7.
Nagorsen DW, Peterson RL. Mammal collectors’ manual: a guide for collecting, documenting and preparing mammal specimens for scientific research, Royal Ontario Museum; 1980.
Zar JH. Biostatistical analysis. New Jersey: Prentice Hall; 1996.
Nathan D, Sakr A, Lichtin JL, Bronaugh RL. In vitro skin absorption and metabolism of benzoic acid, p-aminobenzoic acid, and benzocaine in the hairless guinea pig. Pharm Res. 1990;7:1147–51.
Godwin D, Kim N. Influence of Transcutol® CG on the skin accumulation and transdermal permeation of ultraviolet absorbers. Eur J Pharm Biopharm. 2002;53:23–7.
Mura P, Faucci M, Bramanti G, Corti P. Evaluation of transcutol as a clonazepam transdermal permeation enhancer from hydrophilic gel formulations. Eur J Pharm Sci. 2000;9:365–72.
Narishetty ST, Panchagnula R. Transdermal delivery system for zidovuline: in vitro, ex vivo and in vivo evaluation. Biopharm Drug Dispos. 2004;25:9–20.
Catz P, Friend D. Transdermal delivery of levonorgestrel VIII. Effects of enhancers on rat skin, hairless mouse skin, hairless guinea pig skin, and human skin. Int J Pharm. 1990;58:93–102.
Schmook FP, Maingassner JG, Billich A. Comparison of human skin or epidermis models with human and animal skin in in-vitro percutaneous absorption. Int J Pharm. 2001;215:51–6.
Surber C, Smith E, Schwarb F, Maibach H. Drug concentration in the skin. In: Bronaugh R, Maibach H, editors. Percutaneous absorption. Drugs and the pharmaceutical sciences. New York: Marcel Dekker; 1999. p. 347–74.
Rougier A, Dupuis D, Lotte C, Maibach H. Stripping method for measuring percutaneous absorption in vivo. In: Bronaugh R, Maibach H, editors. Percutaneous absorption. Drugs and the pharmaceutical sciences. New York: Marcel Dekker; 1999. p. 375–94.
ACKNOWLEDGEMENTS
The authors are grateful to Comissão de Aperfeiçoamento de Pessoal de Ensino Superior (CAPES, Brazil, protocol 0115-070) and Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP, Brazil, protocol 06/00121-9).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
de Araujo, D.R., Padula, C., Cereda, C.M.S. et al. Bioadhesive Films Containing Benzocaine: Correlation Between In Vitro Permeation and In Vivo Local Anesthetic Effect. Pharm Res 27, 1677–1686 (2010). https://doi.org/10.1007/s11095-010-0151-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-010-0151-5