Abstract
Introduction and aims
The burden of treatment toxicities in breast cancer requires longitudinal assessment of patient-centered outcomes. The current study aimed to assess the feasibility of collecting general and breast cancer-specific quality of life (QoL), ongoing symptoms and unmet needs, in patients identified from a population-based cancer registry, and to assess the contribution of demographic, disease, and care-related factors.
Methods
Eligible patients were identified from the Victorian Cancer Registry (Victoria, Australia) using the ICD-10 code C50, diagnosed during 2013, 2011, and 2009. Data included age, area of residence, cancer diagnosis, date of diagnosis, treatment modality, and staging. Patients completed a number of validated tools including the EQ-5D-5L and FACT-B, symptom items, and unmet needs.
Results
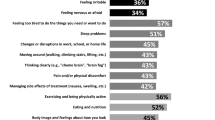
Of 1006 eligible patients, the overall response rate was 45.6%. Survivors 1 year post-diagnosis had significantly greater problems with pain or discomfort (59.2%) and with anxiety or depression (51.3%) compared with survivors 5 years post-diagnosis (45.1% with pain or discomfort, p < 0.05, and 32.7% with anxiety or depression, p < 0.01). For the 5 years group, pain or discomfort and anxiety or depression were significantly higher than for the general population (32.2% and 21.6%, respectively). Improved quality of life was found in those who did not receive chemotherapy (coefficient = 0.2269, p = 0.0409) and those who did not have a longstanding health condition (coefficient = 0.6342, p < 0.001). Poorer quality of life was associated with those who were not certain what was happening with their breast cancer (coefficient = − 0.3674, p = 0.0094) and those whose cancer had not been treated, had been treated but was still present, or had returned after treatment (coefficient = − 0.5314, p = 0.0136). Across the total cohort, women were bothered by changes in weight (21.3%) and concerned about the effects of stress on their cancer (19.6%). Fear of cancer recurrence was commonly reported and did not diminish over time (60.7%, 52.2%, and 56.9% at 1, 3, and 5 years, respectively).
Conclusion
Collecting patient-reported outcomes from a population-based sample of breast cancer survivors was feasible. Physical symptoms and psychosocial issues are common and are persistent. Use of chemotherapy was the only treatment modality that significantly impacted on QoL.
Similar content being viewed by others
Data availability
The data are stored in a deidentified database in the VCCC.
References
Australian Institute of Health and Welfare. (2017). Cancer in Australia 2017. Cancer series no.101. Cat. no. CAN 100. Canberra: AIHW.
EuroQol Group. (2013). EQ-5D-5L user guide. The Netherlands. Retrieved from https://euroqol.org/eq-5d-instruments/eq-5d-5l-about/.
Ong, W. L., Schouwenburg, M. G., van Bommel, A. C. M., Stowell, C., Allison, K. H., Benn, K. E., et al. (2017). A standard set of value-based patient-centered outcomes for breast cancer: The international consortium for health outcomes measurement (ICHOM) initiative. JAMA Oncology,3(5), 677–685.
Lipscomb, J., Gotay, C. C., & Snyder, C. F. (2007). Patient-reported outcomes in cancer: A review of recent research and policy initiatives. CA: A Cancer Journal for Clinicians,57(5), 278–300.
Richards, M., Corner, J., & Maher, J. (2011). The national cancer survivorship initiative: New and emerging evidence on the ongoing needs of cancer survivors. British Journal of Cancer,105(Suppl 1), S1–4.
Glaser, A. W., Fraser, L. K., Corner, J., Feltbower, R., Morris, E. J., Hartwell, G., et al. (2013). Patient-reported outcomes of cancer survivors in England 1–5 years after diagnosis: A cross-sectional survey. British Medical Journal Open,3(4), e002317.
Downing, A., Morris, E. J., Richards, M., Corner, J., Wright, P., Sebag-Montefiore, D., et al. (2015). Health-related quality of life after colorectal cancer in England: A patient-reported outcomes study of individuals 12 to 36 months after diagnosis. Journal of Clinical Oncology,33(6), 616–624.
Downing, A., Wright, P., Hounsome, L., Selby, P., Wilding, S., Watson, E., et al. (2019). Quality of life in men living with advanced and localised prostate cancer in the UK: A population-based study. The lancet Oncology,20(3), 436–447.
Carlsson, M., & Hamrin, E. (1996). Measurement of quality of life in women with breast cancer. Development of a Life Satisfaction Questionnaire (LSQ-32) and a comparison with the EORTC QLQ-C30. Quality of Life Research,5(2), 265–274.
Brady, M. J., Cella, D. F., Mo, F., Bonomi, A. E., Tulsky, D. S., Lloyd, S. R., Deasy, S., Cobleigh, M., Shiomoto, G. (2020). FACT-B. Retrieved from https://www.facit.org/FACITOrg/Questionnaires.
Montazeri, A. (2008). Health-related quality of life in breast cancer patients: A bibliographic review of the literature from 1974 to 2007. Journal of Experimental & Clinical Cancer Research,27(1), 32.
Mols, F., Vingerhoets, A. J., Coebergh, J. W., & van de Poll-Franse, L. V. (2005). Quality of life among long-term breast cancer survivors: A systematic review. European Journal of Cancer,41(17), 2613–2619.
Mertz, B. G., Dunn-Henriksen, A. K., Kroman, N., Johansen, C., Andersen, K. G., Andersson, M., et al. (2017). The effects of individually tailored nurse navigation for patients with newly diagnosed breast cancer: A randomized pilot study. Acta Oncologica,56(12), 1682–1689.
Centre, S. R. (2012). Dual-Frame Omnibus Survey: Technical and Methodological Summary Report.
Foundation, E. R. (2020). ED-5D. Retrieved from https://euroqol.org/eq-5d-instruments/eq-5d-5l-about/.
Viney, R., Norman, R., King, M. T., Cronin, P., Street, D. J., Knox, S., et al. (2011). Time trade-off derived EQ-5D weights for Australia. Value Health,14(6), 928–936.
Norman, R., Cronin, P., & Viney, R. (2013). A pilot discrete choice experiment to explore preferences for EQ-5D-5L health states. Applied Health Economics and Health Policy,11(3), 287–298.
Brady, M. J., Cella, D. F., Mo, F., Bonomi, A. E., Tulsky, D. S., Lloyd, S. R., et al. (1997). Reliability and validity of the functional assessment of cancer therapy-breast quality-of-life instrument. Journal of Clinical Oncology,15(3), 974–986.
Heeringa, S., West, B. T., & Berglund, P. A. (2010). Applied Survey Data Analysis (1st ed.). Boca Raton, FL: CRC Press, Taylor and Francis Group.
Hochberg, Y. (1998). A sharper Bonferroni procedure for multiple tests of significance. Biometrika,75(4), 800–802.
Brown, A. W. W., Kabir, M., Sherman, K. A., Meybodi, F., French, J. R., & Elder, E. B. (2017). Patient reported outcomes of autologous fat grafting after breast cancer surgery. Breast,35, 14–20.
Mercieca-Bebber, R., King, M. T., Boxer, M. M., Spillane, A., Winters, Z. E., Butow, P. N., et al. (2017). What quality-of-life issues do women with ductal carcinoma in situ (DCIS) consider important when making treatment decisions? Breast Cancer,24, 720–729.
Jefford, M., Ward, A. C., Lisy, K., Lacey, K., Emery, J. D., Glaser, A. W., et al. (2017). Patient-reported outcomes in cancer survivors: A population-wide cross-sectional study. Supportive Care in Cancer,25, 3171–3179.
Dindo, D., Demartines, N., & Clavien, P. A. (2004). Classification of surgical complications: A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Annals of Surgery,240(2), 205–213.
National Institutes of Health, US Department of Health and Human Services, Common Terminology Criteria for Adverse Events (CTCAE) Version 4.0, https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03/Archive/CTCAE_4.0_2009-05-29_QuickReference_8.5x11.pdf.
Sprangers, M. A., Groenvold, M., Arraras, J. I., Franklin, J., te Velde, A., Muller, M., et al. (1996). The European Organization for research and treatment of cancer breast cancer-specific quality-of-life questionnaire module: First results from a three-country field study. Journal of Clinical Oncology,14(10), 2756–2768.
Pusic, A. L., Klassen, A. F., Scott, A. M., Klok, J. A., Cordeiro, P. G., & Cano, S. J. (2009). Development of a new patient-reported outcome measure for breast surgery: The BREAST-Q. Plastic and Reconstructive Surgery,124(2), 345–353.
Fallowfield, L. J., Leaity, S. K., Howell, A., Benson, S., & Cella, D. (1999). Assessment of quality of life in women undergoing hormonal therapy for breast cancer: Validation of an endocrine symptom subscale for the FACT-B. Breast Cancer Research and Treatment,55(2), 189–199.
Funding
The Victorian Comprehensive Cancer Center supported the work.
Author information
Authors and Affiliations
Contributions
All authors have contributed at each stage of the project.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest/competing interests.
Ethical approval
Ethical approval to conduct the study was granted on September 30, 2013, by the Cancer Council Victoria Human Research Ethics Committee (Project No: HREC 1307).
Informed consent
All patients consented to participate as per PROJECT HREC1307). All authors have consented for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Skandarajah, A.R., Lisy, K., Ward, A. et al. Patient-reported outcomes in survivors of breast cancer one, three, and five years post-diagnosis: a cancer registry-based feasibility study. Qual Life Res 30, 385–394 (2021). https://doi.org/10.1007/s11136-020-02652-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-020-02652-w