Abstract
Cellulose-supported Schiff base Pd(II) complex [(Cell-Sc-Pd(II))] has been designed and synthesized by multistep approach. (Cell-Sc-Pd(II)) was characterized by techniques, viz. FT-IR, XRD, XPS, EDS, FE-SEM and TGA. Catalytic activity of (Cell-Sc-Pd(II)) is explored for Suzuki Miyaura cross-coupling of phenyl boronic acid and aryl halides. Special features of present method are high TONs and TOFs, low catalyst loading (0.0016 mol %), reusability of the catalyst at least for five times and synthesis of novel biphenyl derivatives of xanthenes.
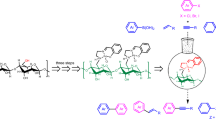
Graphic abstract














Similar content being viewed by others
References
J. Lemo, K. Heuzé, D. Astruc, Org. Lett. 7, 2253 (2005)
G. Fusini, F. Rizzo, G. Angelici, E. Pitzalis, C. Evangelisti and A. Carpita, Catalysts 10, (2020)
L. Yin, J. Liebscher, Chem. Rev. 107, 133 (2007)
K. Sonogashira, Y. Tohda, N. Hagihara, Tetrahedron Lett. 16, 4467 (1975)
A. Monfared, R. Mohammadi, S. Ahmadi, M. Nikpassand, A. Hosseinian, RSC Adv. 9, 3185 (2019)
D. Milstein, J.K. Stiile, J. Am. Chem. Soc. 100, 3636 (1978)
D.Y. Wang, M. Kawahata, Z.K. Yang, K. Miyamoto, S. Komagawa, K. Yamaguchi, C. Wang, M. Uchiyama, Nat. Commun. 7, 1 (2016)
D.S. Gaikwad, D.M. Pore, Synlett 23, 2631 (2012)
T. Baran, J. Mol. Struct. 1141, 535 (2017)
A. Suzuki, Angew. Chemie - Int. Ed. 50, 6722 (2011)
N. Miyaura, A. Suzuki, Chem. Rev. 95, 2457 (1995)
A. Suzuki, J. Organomet. Chem. 576, 147 (1999)
C. Baillie, W. Chen, J. Xiao, Tetrahedron Lett. 42, 9085 (2001)
A.F. Littke, C. Dai, G.C. Fu, J. Am. Chem. Soc. 122, 4020 (2000)
R. Dey, B. Sreedhar, B.C. Ranu, Tetrahedron 66, 2301 (2010)
Z. Xin, H. Peng, A. Zhang, T. Talreja, G. Kumaravel, L. Xu, E. Rohde, M. Y. Jung, M. N. Shackett, D. Kocisko, S. Chollate, A. W. Dunah, P. A. Snodgrass-Belt, H. Moore Arnold, A. G. Taveras, K. J. Rhodes and R. H. Scannevin, Bioorganic Med. Chem. Lett. 21, 7277 (2011)
K.W. Quasdorf, M. Riener, K.V. Petrova, N.K. Garg, J. Am. Chem. Soc. 131, 17748 (2009)
A. Taheri Kal Koshvandi, M. M. Heravi and T. Momeni, Appl. Organomet. Chem. 32 (2018)
M.A. Selepe, F.R. Van Heerden, Molecules 18, 4739 (2013)
C. del Pozo, A. Corma, M. Iglesias, F. Sánchez, Green Chem. 13, 2471 (2011)
I. Hussain, J. Capricho, M.A. Yawer, Adv. Synth. Catal. 358, 3320 (2016)
T. Baran, A. Menteş, J. Mol. Struct. 1122, 111 (2016)
T. Baran, I. Sargin, M. Kaya, A. Menteş, J. Mol. Catal. A Chem. 420, 216 (2016)
T. Baran and M. Nasrollahzadeh, Carbohydr. Polym. 222, 115029 (2019)
M. Nasrollahzadeh, S.M. Sajadi, A. Rostami-Vartooni, M. Khalaj, RSC Adv. 4, 43477 (2014)
P.R. Boruah, P.S. Gehlot, A. Kumar, D. Sarma, Mol. Catal. 461, 54 (2018)
R. Sayah, K. Glegoła, E. Framery, V. Dufaud, Adv. Synth. Catal. 349, 373 (2007)
B.W. Glasspoole, J.D. Webb, C.M. Crudden, J. Catal. 265, 148 (2009)
X. Feng, M. Yan, T. Zhang, Y. Liu, M. Bao, Green Chem. 12, 1758 (2010)
J. Zhi, D. Song, Z. Li, X. Lei, A. Hu, Chem. Commun. 47, 10707 (2011)
Q. Du, Y. Li, Beilstein J. Org. Chem. 7, 378 (2011)
S. Keshipour, S. Shojaei, A. Shaabani, Cellulose 20, 973 (2013)
P. Mhaldar, S. Vibhute, G. Rashinkar and D. Pore, React. Funct. Polym. 152 (2020)
T. Baran, N. Yılmaz Baran and A. Menteş, Appl. Organomet. Chem. 32, 1 (2018)
T. Baran, Catal. Letters (2019)
T. Baran, A. Menteş, J. Organomet. Chem. 803, 30 (2016)
N. Y. Baran, T. Baran, M. Nasrollahzadeh and R. S. Varma, J. Organomet. Chem. 900 (2019)
T. Baran and M. Nasrollahzadeh, J. Phys. Chem. Solids 146 (2020)
T. Baran, Carbohydr. Polym. 195, 45 (2018)
T. Baran, M. Nasrollahzadeh, Int. J. Biol. Macromol. 148, 565 (2020)
M. Nasrollahzadeh, N.S.S. Bidgoli, Z. Issaabadi, Z. Ghavamifar, T. Baran, R. Luque, Int. J. Biol. Macromol. 148, 265 (2020)
K. Hong, M. Sajjadi, J.M. Suh, K. Zhang, M. Nasrollahzadeh, H.W. Jang, R.S. Varma, M. Shokouhimehr, A.C.S. Appl, Nano Mater. 3, 2070 (2020)
M. Nasrollahzadeh, Z. Issaabadi, M. M. Tohidi and S. Mohammad Sajadi, Chem. Rec. 18, 165 (2018)
V. Polshettiwar, C. Len, A. Fihri, Coord. Chem. Rev. 253, 2599 (2009)
J. Chen, J. Zhang, D. Zhu, T. Li, Appl. Organomet. Chem. 32, 1 (2018)
Q. Sun, Z. Dai, X. Meng, F.S. Xiao, Chem. Soc. Rev. 44, 6018 (2015)
D. Klemm, B. Heublein, H.P. Fink, A. Bohn, Angew. Chemie - Int. Ed. 44, 3358 (2005)
X. Wang, P. Hu, F. Xue, Y. Wei, Carbohydr. Polym. 114, 476 (2014)
T. Baran, N. Yılmaz Baran and A. Menteş, J. Mol. Struct. 1160, 154 (2018)
Y. Dong, X. Wu, X. Chen, Y. Wei, Carbohydr. Polym. 160, 106 (2017)
M. Ibnu Abdulwahab, A. Khamkeaw, B. Jongsomjit and M. Phisalaphong, Catal. Letters 147, 2462 (2017)
N. Yılmaz Baran, T. Baran and A. Menteş, Appl. Catal. A Gen. 531, 36 (2017)
M. Boroumand Jazi, M. Arshadi, M. J. Amiri and A. Gil, J. Colloid Interface Sci. 422, 16 (2014)
S. Radi, Y. Toubi, M. El-Massaoudi, M. Bacquet, S. Degoutin, Y.N. Mabkhot, J. Mol. Liq. 223, 112 (2016)
X. Li, Y. Tang, X. Cao, D. Lu, F. Luo, W. Shao, Colloids Surfaces A Physicochem. Eng. Asp. 317, 512 (2008)
X. Qi, H. Yoon, S.H. Lee, J. Yoon, S.J. Kim, J. Ind. Eng. Chem. 14, 136 (2008)
R. Vafazadeh, B. Khaledi, A.C. Willis, M. Namazian, Polyhedron 30, 1815 (2011)
Y. Li, L. Xu, B. Xu, Z. Mao, H. Xu, Y. Zhong, L. Zhang, B. Wang, X. Sui, A.C.S. Appl, Mater. Interfaces 9, 17155 (2017)
K. Dhara, K. Sarkar, D. Srimani, S.K. Saha, P. Chattopadhyay, A. Bhaumik, Dalt. Trans. 39, 6395 (2010)
D.M. Pore, T.S. Shaikh, N.G. Patil, S.B. Dongare, U.V. Desai, Synth. Commun. 40, 2215 (2010)
P. Cotugno, M. Casiello, A. Nacci, P. Mastrorilli, M.M. Dell’Anna, A. Monopoli, J. Organomet. Chem. 752, 1 (2014)
M. Kempasiddaiah, V. Kandathil, R.B. Dateer, B.S. Sasidhar, S.A. Patil, S.A. Patil, Cellulose 27, 3335 (2020)
D. Kale, G. Rashinkar, A. Kumbhar, R. Salunkhe, React. Funct. Polym. 116, 9 (2017)
N. Shang, C. Feng, H. Zhang, S. Gao, R. Tang, C. Wang, Z. Wang, Catal. Commun. 40, 111 (2013)
A. Zarnegaryan, Z. Dehbanipour, D. Elhamifar, Polyhedron 170, 530 (2019)
M. Shaker, D. Elhamifar, New J. Chem. 44, 3445 (2020)
Acknowledgements
One of the authors, Pharande P. S., is grateful to the Council of Scientific and Industrial Research (CSIR), New Delhi, Government of India, for the award of the Junior Research Fellowship (File no. 09/816(0044)/2019-EMR-I).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pharande, P.S., Rashinkar, G.S. & Pore, D.M. Cellulose Schiff base-supported Pd(II): An efficient heterogeneous catalyst for Suzuki Miyaura cross-coupling. Res Chem Intermed 47, 4457–4476 (2021). https://doi.org/10.1007/s11164-021-04528-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-021-04528-1