Abstract
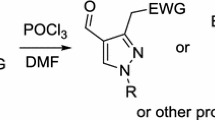
Formylation of ten accessible phenylhydrazones and semicarbazones derived from alkyl, benzyl, and cycloalkyl methyl ketones with the complex of POCl3 with dimethylformamide was studied. Depending on the electronic and steric structure of the substrates, the reaction yields 1-phenyl- or 1-unsubstituted 3,4-dialkyl-, 3-alkyl-4-aryl-, or 3-alkyl-4-formylpyrazoles. These compounds can be readily oxidized into the corresponding carboxylic acids.
Similar content being viewed by others
REFERENCES
Kira, M.A. and Bruckner-Wilhelm, A., Acta Chim. Acad. Sci. Hung., 1968, vol. 56, p. 47.
Kira, M.A., Bruckner-Wilhelm, A., and Ruff, F., Acta Chim. Acad. Sci. Hung., 1968, vol. 56, p. 189.
Kira, M.A., Abdel-Raeman, M.O., and Gadalla, K.Z., Tetrahedron Lett., 1969, no. 2, p. 109.
Kira, M.A., Aboul-Enein, M.N., and Korkor, M.I., J. Heterocyclic Chem., 1970, vol. 7, no.1, p. 25.
Author information
Authors and Affiliations
Additional information
__________
Translated from Zhurnal Obshchei Khimii, Vol. 75, No. 3, 2005, pp. 448–452.
Original Russian Text Copyright © 2005 by Lebedev, Lebedeva, Sheludyakov, Kovaleva, Ustinova, Kozhevnikov.
Rights and permissions
About this article
Cite this article
Lebedev, A.V., Lebedeva, A.B., Sheludyakov, V.D. et al. Vilsmeier Formylation of Hydrazones and Semicarbazones Derived from Alkyl, Benzyl, and Cycloalkyl Methyl Ketones. Russ J Gen Chem 75, 412–416 (2005). https://doi.org/10.1007/s11176-005-0241-y
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11176-005-0241-y