Abstract
Introduction
A great variety of bioactive natural products has been reported for different Palicourea and Psychotria species (Rubiaceae). However, few of them as well as few of species of these botanical genera have been evaluated for antiplasmodial activity.
Objective
To assess the antiplasmodial activity of 24 extracts from Palicourea and Psychotria genera, along with the targeted LC–MS metabolite profiling, as well as identification of the main metabolites in the bioactive extracts.
Methods
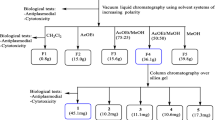
Twenty four ethanol and acid–base extracts from Palicourea and Psychotria genera collected in the Amazonia and Atlantic Forest, Brazil, were evaluated against chloroquine-resistant Plasmodium falciparum W2 strain by PfLDH. The metabolite profiling and putative identification of metabolites from bioactive extracts were determined by LC-DAD-ESI–MS and LC-HRMS, respectively.
Results
The ethanol extracts disclosed low antiplasmodial activity (% GI < 50%). High antiplasmodial effect was observed for the acid–base extracts from Psychotria apoda and Psychotria colorata with 100% inhibition of parasite growth inhibition. Fragment ions related to pyrrolidinoindoline alkaloids were observed by LC-DAD-ESI–MS mainly in the most bioactive extracts. The results of the in vitro screening associated with the LC-DAD-ESI–MS and LC-HRMSn data allowed to predict, for the first time, the pyrrolidinoindoline alkaloids as possible antiplasmodial representing, then, new potential natural antimalarial hits. In addition, other metabolite classes such as flavanones, lignans and chalcones were also putatively identified in the bioactive extracts of Psychotria apoda, Psychotria capitata, and Psychotria poeppigiana.
Conclusion
The present results point to Palicourea and Psychotria species as sources of new antimalarial hits.





Similar content being viewed by others
References
Amirka, V., & Heinrich, M. (2014). Alkaloids as drug leads—A predictive structural and biodiversity-based analysis. Phytochemistry Letters, 10, 48–53. https://doi.org/10.1016/j.phytol.2014.06.015
Antony, H. A., & Parija, S. C. (2016). Antimalarial drug resistance: An overview. Tropical Parasitolology, 6, 30–41. https://doi.org/10.4103/2229-5070.175081
Ayres, D. C., & Loike, J. D. (1990). Lignans: Chemical, Biological and Clinical Properties. Cambridge: Cambridge University Press. https://doi.org/10.1017/CBO9780511983665
Barreca, D., Bisignano, C., Ginestra, G., Bisignano, G., Bellocco, E., Leuzzi, U., et al. (2013). Polymethoxylated, C- and O-glycosyl flavonoids in tangelo (Citrus reticulata X Citrus paradisi) juice and their influence on antioxidant properties. Food Chemistry, 141, 1481–1488. https://doi.org/10.1016/j.foodchem.2013.03.095
Berger, A., Kostyan, M. K., Klose, S. I., Gastegger, M., Lorbeer, E., Brecker, L., et al. (2015). Loganin and secologanin derived tryptamine-iridoid alkaloids from Palicourea crocea and Palicourea padifolia (Rubiaceae). Phytochemistry, 116, 162–169. https://doi.org/10.1016/j.phytochem.2015.05.013
Berger, A., Tanuhadi, E., Brecker, L., Schinnerl, J., & Valant-Vetschera, K. (2017). Chemodiversity of tryptamine-derive alkaloids in six Costa Rican Palicourea species (Rubiaceae – Palicoureeae). Phytochemistry, 143, 124–131. https://doi.org/10.1016/j.phytochem.2017.07.016
Bertelli, P. R., Biegelmeyer, R., Rico, E. P., Klein-Júnior, L. C., Toson, N. S. B., Minetto, L., et al. (2017). Toxicological profile and acetylcholinesterase inhibitory potential of Palicourea deflexa, a source of β-carboline alkaloids. Comparative Biochemistry and Physiology C, 201, 44–50. https://doi.org/10.1016/j.cbpc.2017.09.003
Blasco, B., Leroy, D., & Fidock, D. A. (2017). Antimalarial drug resistance: Linking Plasmodium falciparum parasite biology to the clinic. Nature Medicine, 23, 917–928. https://doi.org/10.1038/nm.4381
Carvalho, A. R., Jr., Ferreira, R. O., Passos, M. S., Boeno, S. I. S., Virgens, L. L. G., Ventura, T. L. B., et al. (2019). Antimycobacterial and nitric oxide production inhibitory activities of triterpenes and alkaloids from Psychotria nuda (Cham. & Schltdl.) Wawra. Molecules, 24, 1026. https://doi.org/10.3390/molecules24061026
Calderón, A., Romero, L. I., Ortega-Barría, E., Brun, R., Correa, M. D., & Gupta, M. P. (2006). Evaluation of larvicidal and in vitro antiparasitic activities of plants in a biodiversity plot in the Altos de Campana National Park, Panama. Pharmaceutical Biology, 44, 487–498. https://doi.org/10.1080/13880200600878361
Calixto, N. O., Pinto, M. E. F., Ramalho, S. D., Burguer, M. C. M., Bobey, A. F., et al. (2016). The genus Psychotria: Phytochemistry, chemotaxonomy, ethnopharmacology and biological properties. Journal of the Brazilian Chemical Society, 27, 1355–1378. https://doi.org/10.5935/0103-5053.20160149
Chibli, L. A., Rosa, A. L., Nonato, M. C., & Da Costa, F. B. (2019). Untargeted LC-MS metabolomics studies of Asteraceae species to discover inhibitors of Leishmania major dihydroorotate dehydrogenase. Metabolomics, 15, 1–22. https://doi.org/10.1007/s11306-019-1520-7
Cho, N., Valenciano, A. L., Du, Y., Clement, J., Cassera, M. B., Goetz, M., et al. (2018). Antiplasmodial flavanones and a stilbene from Carpha glomerata. Bioorganic Medicinal Chemistry Letters, 28, 3368–3371. https://doi.org/10.1016/j.bmcl.2018.09.003
Cragg, G. M., Newmanm, D. J., & Yang, S. S. (2006). Natural product extracts of plant and marine origin having Antileukemia potential. The NCI experience. Journal of Natural Products, 69, 488–498. https://doi.org/10.1021/np0581216
Cragg, G. M., & Newman, D. J. (2013). Natural products: A continuing source of novel drug leads. Biochimica Et Biophysica Acta, 1830, 3670–3695. https://doi.org/10.1016/j.bbagen.2013.02.008
Formagio, A. S. N., Volobuff, C. R. F., Kassuya, C. A. L., Cardoso, C. A. L., Vieira, M. C., Pereira, Z. V., et al. (2019). Psychotria leiocarpa extract and vincosamide reduce chemically-induced inflammation in mice and inhibit the acetylcholinesterase activity. Inflammation, 42, 1561–1574. https://doi.org/10.1007/s10753-019-01018-w
Freire, R. T., Bero, J., Beaufay, C., Selegato, D. M., Coqueiro, A., Choi, Y. H., et al. (2019). Identification of antiplasmodial triterpenes from Keetia species using NMR-based metabolic profiling. Metabolomics, 15, 1–11. https://doi.org/10.1007/s11306-019-1487-4
Giang, P. M., & Otsuka, H. (2018). New compounds and potential candidates for drug discovery from medicinal plants of Vietnam. Chemical and Pharmaceutical Bulletin, 66, 493–505. https://doi.org/10.1248/cpb.c17-00628
Gontijo, D. C., Brandão, G. C., Gontijo, P. C., Oliveira, A. B., Diaz, M. A. N., Fietto, L. G., et al. (2017). Identification of phenolics compounds and biologically related activities from Ocotea odorifera aqueous extract leaves. Food Chemistry, 230, 618–626. https://doi.org/10.1016/j.foodchem.2017.03.087
Gontijo, D. C., Leite, J. P. V., Nascimento, M. F. A., Brandão, G. C., & Oliveira, A. B. (2019a). Bioprospection for antiplasmodial activity, and identification of bioactive metabolites of native plants species from the Atlantic Forest biome, Brazil. Natural Product Research, 24, 1–6. https://doi.org/10.1080/14786419.2019.1633645
Gontijo, D. C., Brandão, G. C., Nascimento, M. F. A., & Oliveira, A. B. (2019b). Antiplasmodial activity and cytotoxicity, isolation of active alkaloids, and dereplication of Xylopia sericea leaves ethanol extract by UPLC-DAD-ESI-MS/MS. Journal of Pharmacy and Pharmacology, 71, 260–269. https://doi.org/10.1111/jphp.13029
Hadi, S., Rahmawati, K. P., Asnawati, D., Ersalena, V. F., & Azwari, A. (2014). Characterization of Alkaloids from the leaves of Psychotria malayana Jack of Lombok Island on the basis of gas chromatography-mass spectroscopy. The Journal of Pure and Applied Chemistry Research, 3, 108–113. https://doi.org/10.21776/UB.JPACR.2014.003.03.176
Hanhineva, K., Rogachev, I., Aura, A. M., Aharoni, A., Poutnen, K., & Mykkänen, H. (2012). Identification of novel lignans in the whole grain rye bran by non-targeted LC-MS metabolite profiling. Metabolomics, 8, 399–409. https://doi.org/10.1007/s11306-011-0325-0
Jannic, V., Guéritte, F., Laprévote, O., Serani, L., Martin, M. T., Sévenet, T., et al. (1999). Pyrrolidinoindoline alkaloids from Psychotria oleides and Psychotria lyciiflora. Journal of Natural Products, 62, 838–843. https://doi.org/10.1021/np9805387
Katchborian-Neto, A., Santos, W. T., Nicácio, K. J., Corrêa, J. O., Murgu, M., Martins, T. M. M., et al. (2020). Neuroprotective potential of Ayahusca and untargeted metabolomics analyses: Applicability to Parkinson’s disease. Journal of Ethnopharmacology, 255, 112743. https://doi.org/10.1016/j.jep.2020.112743
Klein-Júnior, L. C., Cretton, S., Allard, P. M., Genta-Jouve, G., Passos, C. S., Salton, J., et al. (2017). Targeted isolation of monoterpene indole alkaloids from Palicourea sessilis. Journal of Natural Products, 80, 3032–3037. https://doi.org/10.1021/acs.jnatprod.7b00681
Klein-Júnior, L. C., Cretton, S., Heyden, Y. V., Gasper, A. L., Nejad-Ebrahimi, S., Christen, P., et al. (2020). Bioactive Azepine-Indole alkaloids from Psychotria nemorosa. Journal of Natural Products, 83, 852–863. https://doi.org/10.1021/acs.jnatprod.9b00469
Kornpointner, C., Berger, A., Fischer, I. M., Popl, L., Groher, C., Valant-Vetschera, K., et al. (2018). Revisiting costa rican Carapichea affinis (Rubiaceae: Palicoureeae): A source of bioactive dopamine-iridoid alkaloids. Phytochemistry Letters, 26, 164–169. https://doi.org/10.1016/j.phytol.2018.05.004
Kornpointner, C., Berger, A., Traxler, F., Hadziabdic, A., Massar, M., Matek, J., et al. (2020). Alkaloid and iridoid glucosides from Palicourea luxurians (Rubiaceae: Palicoureeae) indicate tryptamine- and tryptophan-iridoid alkaloid formation apart the strictosidine pathway. Phytochemistry, 173, 112296. https://doi.org/10.1016/j.phytochem.2020.112296
Lathrop, S., & Movassaghi, M. (2014). Application of diazene-directed fragment assembly to the total synthesis and stereochemical assignmet of (+)-meso-chimonanthine and related heterodimeric alkaloids. Chemical Science, 5, 333–340. https://doi.org/10.1039/C3SC52451E
Lu, Q., Wang, J., & Kong, L. (2014). Chemical constituents from Psychotria yunnanensis and its chemotaxonomic study. Biochemical Systematics and Ecology, 52, 20–22. https://doi.org/10.1016/j.bse.2013.11.002
Mabry, T. J., Markham, K. R., & Thomas, M. B. (1970). The Systematic Identification of Flavonoids. New York: Springer. https://doi.org/10.1007/978-3-642-88458-0
Magedans, Y. V. S., Matsuura, H. N., Tasca, R. A. J. C., Wairich, A., Junkes, C. F. O., Costa, F., et al. (2017). Accumulation of the antioxidant alkaloid brachycerine from Psychotria brachyceras Müll. Arg. is increased by heat and contributes to oxidative stress mitigation. Environmental and Experimental Botany, 143, 185–193. https://doi.org/10.1016/j.envexpbot.2017.09.008
Makler, M. T., Piper, R. C., & Milhous, W. K. (1998). Lactate dehydrogenase and the diagnosis of malaria. Parasitology Today, 14, 376–377. https://doi.org/10.1016/S0169-4758(98)01284-8
Moura, V. M., Ribeiro, M. A. S., Corrêa, J. G. S., Peixoto, M. A., Souza, G. K., Morais, D., et al. (2020). Minutifloroside, a new bis-iridoid glucoside with antifungal and antioxidant activities and other constituents from Palicourea minutiflora. Journal of the Brazilian Chemical Society, 31, 505–511. https://doi.org/10.21577/0103-5053.20190209
Muhammad, I., Dunbar, D. C., Khan, S. I., Tekwani, B. L., Bedir, E., Takamatsu, S., et al. (2003). Antiparasitic Alkaloids from Psychotria klugii. Journal of Natural Products, 66, 962–967. https://doi.org/10.1021/np030086k
Nakano, T., & Martín, A. (1976). Studies on the alkaloids of Palicourea fendleri. Planta Medica, 30, 185–188. https://doi.org/10.1055/s-0028-1097715
Nascimento, M. F. A., Borgati, T. F., Souza, L. C. R., Tagliati, C. A., & Oliveira, A. B. (2020). In silico, in vitro and in vivo evaluation of natural Bignoniaceous naphthoquinones in comparison with atovaquone targeting the selection of potential antimalarial candidates. Toxicology and Applied Pharmacology, 401, 115074. https://doi.org/10.1016/j.taap.2020.115074
Ohashi, L. H., Gontijo, D. C., Nascimento, M. F. A., Margalho, L. F., Brandão, G. C., & Oliveira, A. B. (2021). Extraction and fractionation effects on antiplasmodial activity and phytochemical composition of Palicourea hoffmansseggiana. Planta Medica International Open, 8, 1–9. https://doi.org/10.1055/a-1375-6456
Pinto, M. E. F., Chan, L. Y., Koehbach, J., Devi, S., Gründemann, C., Gruber, C. W., et al. (2021). Cyclotides from Brazilian Palicourea sessilis and their effects on Human lymphocytes. Journal of Natural Products, 84, 81–90. https://doi.org/10.1021/acs.jnatprod.0c01069
Portet, B., Fabre, N., Roumy, V., Gornitzka, H., Bourdy, G., Chevalley, S., et al. (2007). Activity-guided isolation of antiplasmodial dihydrochalcones and flavanones from Piper hostmannianum var. berbicense. Phytochemistry, 68, 1312–1320. https://doi.org/10.1016/j.phytochem.2007.02.006
Porto, D. D., Matsuura, H. N., Henriques, A. T., Rosa, L. M. G., Fett, J. P., & Fett-Neto, A. G. (2020). The alkaloid brachycerine contributes to protection against acute UV-B damage in Psychotria. Industrial Crops and Products, 147, 112216. https://doi.org/10.1016/j.indcrop.2020.112216
Sangster, A. W., & Stuart, K. L. (1964). Ultraviolet spectra of alkaloids. Chemical Reviews, 65, 69–130. https://doi.org/10.1021/cr60233a003
Solis, P. N., Lang’at, C., Gupta, M. P., Kirby, G. C., Warhust, D. C., & Phillipson, J. D. (1995). Bio-active compounds from Psychotria camponutans. Planta Medica, 61, 62–65. https://doi.org/10.1055/s-2006-958001
Systat. (2013). Software, San Jose, CA, USA. https://systatsoftware.com/
Uzor, P. F. (2020). Alkaloids from plants with antimalarials activity: A review of recent studies. Evidence-Based Complementary and Alternative Medicine. https://doi.org/10.1155/2020/8749083
Verotta, L., Pilati, T., Tatò, M., Elisabetsky, E., Amador, T. A., & Nunes, D. S. (1998). Pyrrolidinoindoline alkaloids from Psychotria colorata. Journal of Natural Products, 61, 392–396. https://doi.org/10.1021/np9701642
Verotta, L., Peterlongo, F., Elisabetsky, E., Amador, T. A., & Nunes, D. S. (1999). High-performance liquid chromatography-diode array detection-tandem, mass spectrometry analyses of the Alkaloids extract of Amazon Psychotria species. Journal of Chromatography A, 841, 165–176. https://doi.org/10.1016/S0021-9673(99)00298-8
Wang, X., Liu, J., Zhang, A., Sun, H., & Zhang, Y. (2017). Systematic characterization of the absorbed components of Acanthopanax senticosus stem. Serum Pharmacochemistry of Traditional Chinese Medicine, Chapter, 23, 313–336. https://doi.org/10.1016/B978-0-12-811147-5.00023-6
WHO, World Health Organization. (2020). World malaria report 2020. https://www.who.int/news-room/fact-sheets/detail/malaria. Accessed Feb 17, 2021.
Yang, H., Zhang, H., Yang, C., & Chen, Y. (2016). Chemical constituents of plants from the genus Psychotria. Chemistry & Biodiversity, 13, 1–14. https://doi.org/10.1002/cbdv.201500259
Acknowledgements
To the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – CAPES for Postdoctoral fellowship to DCG and to Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq for Senior Research fellowship to ABO. To the CRTI – Centro Regional para o Desenvolvimento Tecnológico e Inovação, UFG, on behalf of Prof. CMA Oliveira, for LC-HRMSn data aquisition.
Author information
Authors and Affiliations
Contributions
DCG and ABO wrote the manuscript. DCG performed the phytochemical work and data analyses. MFAN performed the in vitro antiplasmodial assays. HVSR performed the multivariate statistical analyses. RAM and LFM performed collection and taxonomic identification of the Palicourea and Psychotria species in the states of Minas Gerais and Pará, respectively. Acquisition of LC–MS was performed by DCG and GCB in the Laboratório de Fitoquímica, Faculdade de Farmácia, UFMG. ABO proposed the research project and supervised its implementation.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have influenced the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gontijo, D.C., do Nascimento, M.F.A., Rody, H.V.S. et al. In vitro antiplasmodial activity, targeted LC–MS metabolite profiling, and identification of major natural products in the bioactive extracts of Palicourea and Psychotria species from the Amazonia and Atlantic Forest biomes, Brazil. Metabolomics 17, 81 (2021). https://doi.org/10.1007/s11306-021-01833-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-021-01833-z