Abstract
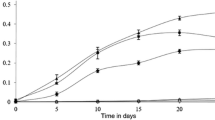
This study investigates the ability of Rhodococcus sp. strain p52, a dioxin degrader, to biodegrade petroleum hydrocarbons. Strain p52 can use linear alkanes (tetradecane, tetracosane, and dotriacontane), branched alkane (pristane), and aromatic hydrocarbons (naphthalene and phenanthrene) as sole carbon and energy sources. Specifically, the strain removes 85.7 % of tetradecane within 48 h at a degradation rate of 3.8 mg h−1 g−1 dry cells, and 79.4 % of tetracosane, 66.4 % of dotriacontane, and 63.9 % of pristane within 9–11 days at degradation rates of 20.5, 14.7, and 20.3 mg day−1 g−1 dry cells, respectively. Moreover, strain p52 consumes 100 % naphthalene and 55.3 % phenanthrene within 9–11 days at respective degradation rates of 16 and 12.9 mg day−1 g−1 dry cells. Metabolites of the petroleum hydrocarbons by strain p52 were analyzed. Genes encoding alkane-hydroxylating enzymes, including cytochrome P450 (CYP450) enzyme (CYP185) and two alkane-1-monooxygenases, were amplified by polymerase chain reaction. The transcriptional activities of these genes in the presence of petroleum hydrocarbons were detected by reverse transcription-polymerase chain reaction. The results revealed potential of strain p52 to degrade petroleum hydrocarbons.




Similar content being viewed by others
References
Auffret M, Labbe D, Thouand G, Greer C, Fayolle-Guichard F (2009) Degradation of a mixture of hydrocarbons, gasoline, and diesel oil additives by Rhodococcus aetherivorans and Rhodococcus wratislaviensis. Appl Environ Microbiol 75:7774–7782
Broadway NM, Dickinson FM, Ratledge C (1993) The enzymology of dicarboxylic acid formation by Corynebacterium sp. strain 7E1C grown on n-alkanes. J Gen Microbiol 139:1337–1344
Cardini G, Jurtshuk P (1970) The enzymatic hydroxylation of n-octane by Corynebacterium sp. strain 7E1C. J Biol Chem 245:2789–2796
Coleman NV, Bui NB, Holmes AJ (2006) Soluble di-iron monooxygenase gene diversity in soils, sediments and ethene enrichments. Environ Microbiol 8:1228–1239
Funhoff EG, Bauer U, Garcia-Rubio I, Witholt B, van Beilen JB (2006) CYP153A6, a soluble P450 oxygenase catalyzing terminal-alkane hydroxylation. J Bacteriol 188:5220–5227
Keum YS, Seo JS, Hu Y, Li QX (2006) Degradation pathways of phenanthrene by Sinorhizobium sp. C4. Appl Microbiol Biotechnol 71:935–941
Li L, Li Q, Li F, Shi Q, Yu B, Liu F, Xu P (2006) Degradation of carbazole and its derivatives by a Pseudomonas sp. Appl Microbiol Biotechnol 73:941–948
Ludwig B, Akundi A, Kendall K (1995) A long-chain secondary alcohol dehydrogenase from Rhodococcus erythropolis ATCC 4277. Appl Environ Microbiol 61:3729–3733
Maier T, Forster HH, Asperger O, Hahn U (2001) Molecular characterization of the 56-kDa CYP153 from Acinetobacter sp. EB104. Biochem Biophys Res Commun 286:652–658
Marquez-Rocha FJ, Hernández-Rodrí V, Lamela MT (2001) Biodegradation of diesel oil in soil by a microbial consortium. Water Air Soil Pollut 128:313–320
Muller R, Asperger O, Kleber HP (1989) Purification of cytochrome P-450 from n-hexadecane-grown Acinetobacter calcoaceticus. Biomed Biochim Acta 48:243–254
Nhi-Cong LT, Mikolasch A, Klenk H-P, Schauer F (2009) Degradation of the multiple branched alkane 2,6,10,14-tetramethyl-pentadecane (pristane) in Rhodococcus ruber and Mycobacterium neoaurum. Int Biodeterior Biodegrad 63:201–207
Östberg TL, Jonsson AP, Lundström US (2006) Accelerated biodegradation of n-alkanes in aqueous solution by the addition of fermented whey. Int Biodeterior Biodegrad 57:190–194
Peng P, Yang HY, Jia RB, Li L (2013) Biodegradation of dioxin by a newly isolated Rhodococcus sp. with the involvement of self-transmissible plasmids. Appl Microbiol Biotechnol 97:5585–5595
Pirnik MP, Atlas RM, Bartha R (1974) Hydrocarbon metabolism by Brevibacterium erythrogenes: normal and branched alkanes. J Bacteriol 119:868–878
Rapp P, Gabriel-Jurgens LH (2003) Degradation of alkanes and highly chlorinated benzenes, and production of biosurfactants, by a psychrophilic Rhodococcus sp. and genetic characterization of its chlorobenzene dioxygenase. Microbiology 149:2879–2890
Rojo F (2009) Degradation of alkanes by bacteria. Environ Microbiol 11:2477–2490
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press. Cold Spring Harbor, New York
Sayavedra-Soto LA, Chang WN, Lin TK, Ho CL, Liu HS (2006) Alkane utilization by Rhodococcus strain NTU-1 alone and in its natural association with Bacillus fusiformis L-1 and Ochrobactrum sp. Biotechnol Prog 22:1368–1373
Schaeffer TL, Cantwell SG, Brown JL, Watt DS, Fall RR (1979) Microbial growth on hydrocarbons: terminal branching inhibits biodegradation. Appl Environ Microbiol 38:742–746
Sorkhoh NA, Ghannoum MA, Ibrahim AS, Stretton RJ, Radwan SS (1990) Crude oil and hydrocarbon-degrading strains of Rhodococcus rhodochrous isolated from soil and marine environments in Kuwait. Environ Pollut 65:1–17
Takei D, Washio K, Morikawa M (2008) Identification of alkane hydroxylase genes in Rhodococcus sp. strain TMP2 that degrades a branched alkane. Biotechnol Lett 30:1447–1452
Tang J, Wang M, Wang F, Sun Q, Zhou Q (2011) Eco-toxicity of petroleum hydrocarbon contaminated soil. J Environ Sci (China) 23:845–851
van Beilen JB, Funhoff EG (2007) Alkane hydroxylases involved in microbial alkane degradation. Appl Microbiol Biotechnol 74:13–21
van Beilen JB, Wubbolts MG, Witholt B (1994) Genetics of alkane oxidation by Pseudomonas oleovorans. Biodegradation 5:161–174
van Beilen JB, Li Z, Duetz WA, Smits TH, Witholt B (2003) Diversity of alkane hydroxylase systems in the environment. Oil Gas Sci Technol 58:427–440
van Beilen JB, Marin MM, Smits TH, Röthlisberger M, Franchini AG, Witholt B, Rojo F (2004) Characterization of two alkane hydroxylase genes from the marine hydrocarbonoclastic bacterium Alcanivorax borkumensis. Environ Microbiol 6:264–273
Wang XB, Chi CQ, Nie Y, Tang YQ, Tan Y, Wu G, Wu XL (2011) Degradation of petroleum hydrocarbons (C6-C40) and crude oil by a novel Dietzia strain. Bioresour Technol 102:7755–7761
Warhurst AM, Fewson CA (1994) Biotransformations catalyzed by the genus Rhodococcus. Crit Rev Biotechnol 14:29–73
Whyte LG, Bourbonniere L, Greer CW (1997) Biodegradation of petroleum hydrocarbons by psychrotrophic Pseudomonas strains possessing both alkane (alk) and naphthalene (nah) catabolic pathways. Appl Environ Microbiol 63:3719–3723
Whyte LG, Hawari J, Zhou E, Bourbonniere L, Inniss WE, Greer CW (1998) Biodegradation of variable-chain-length alkanes at low temperatures by a psychrotrophic Rhodococcus sp. Appl Environ Microbiol 64:2578–2584
Whyte LG, Slagman SJ, Pietrantonio F, Bourbonniere L, Koval SF, Lawrence JR, Inniss WE, Greer CW (1999) Physiological adaptations involved in alkane assimilation at a low temperature by Rhodococcus sp. strain Q15. Appl Environ Microbiol 65:2961–2968
Whyte LG, Smits THM, Labbe D, Witholt B, Greer CW, van Beilen JB (2002) Gene cloning and characterization of multiple alkane hydroxylase systems in Rhodococcus strains Q15 and NRRL B-16531. Appl Environ Microbiol 68:5933–5942
Acknowledgments
This work was supported by Jinan Science and Technology Development Foundation for Young Scientist (no. 20080408), Shandong Province Young and Middle-Aged Scientists Research Awards Fund (no. BS2009SW020), and Natural Science Foundation of China (no. 21377069).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Robert Duran
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig5
Fig. SM-1 Proposed metabolic pathway responsible for the identified metabolites of tetradecane by Rhodococcus sp. strain p52. Compounds pointed by solid arrows represent the detected metabolites (in box) in this study, while broken arrows point to the undetected metabolites which were speculated according to the referenced pathway (GIF 18 kb)
Fig6
Fig. SM-2 Proposed metabolic pathway responsible for the identified metabolites of pristane by Rhodococcus sp. strain p52. Compounds pointed by solid arrows represent the detected metabolites in this study, while broken arrows point to the undetected metabolites which were speculated according to the referenced pathway (GIF 6 kb)
Rights and permissions
About this article
Cite this article
Yang, HY., Jia, RB., Chen, B. et al. Degradation of recalcitrant aliphatic and aromatic hydrocarbons by a dioxin-degrader Rhodococcus sp. strain p52. Environ Sci Pollut Res 21, 11086–11093 (2014). https://doi.org/10.1007/s11356-014-3027-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-3027-0