Abstract
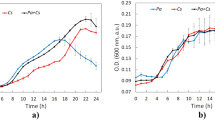
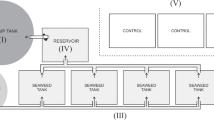
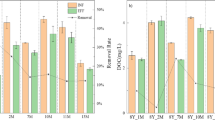
A continuous flow filtration system was designed to identify and quantify the removal mechanisms of Cyanobacteria (Microcystis aeruginosa) by hydroponic biofilters of Phalaris arundinacea compared to synthetic filters. The filtration units were continuously fed under plug-flow conditions with Microcystis grown in photobioreactors. Microcystis cells decreased at the two flow rates studied (1.2 ± 0.2 and 54 ± 3 cm3 minˉ1) and results suggested physical and chemical/biological removal mechanisms were involved. Physical interception and deposition was the main removal mechanism with packing density of the media driving the extent of cell removal at high flow, whilst physical and chemical/biological mechanisms were involved at low flow. At low flow, the biofilters decreased Microcystis cell numbers by 70% compared to the controls. The decrease in cell numbers in the biofilters was accompanied by a chlorotic process (loss of green colour), suggesting oxidative processes by the release of allelochemicals from the biofilters.






Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Alcocer DJR, Vallejos GG, Champagne P (2012) Assessment of the plug flow and dead volume ratios in a sub-surface horizontal-flow packed-bed reactor as a representative model of a sub-surface horizontal constructed wetland. Ecol Eng 40:18–26
Baier A, Winkler W, Korte T, Lockau W, Karradt A (2014) Degradation of Phycobilisomes in Synechocystis sp. PCC6803 evidence for essential formation of a NblA1/NblA2 heterodimer and its codegradation by a Clp protease comples. J Biol Chem 289(17):11755–11766
Benton TG, Solan M, Travis JM, Sait SM (2007) Microcosm experiments can inform global ecological problems. Trends Ecol Evol 22:516–521
Campos LC, Su MFJ, Graham NJD, Smith SR (2002) Biomass development in slow sand filters. Water Res 36:4543–4551
Castro-Castellon AT, Chipps MJ, Hankins NP, Hughes JMR (2016) Lessons from the “Living-Filter”: an in-reservoir floating treatment wetland for phytoplankton reduction prior to a water treatment works intake. Ecol Eng 95:839–851
Chang DW, Hsieh ML, Chen YM, Lie TF, Chang JS (2011) Kinetics of cell lysis for Microcystis aeruginosa and Nitzschia palea in the exposure to beta-cyclocitral. J Hazard Mater 185:1214–1220
Chorus I, Bartram J (1999) Toxic cyanobateria in water: a guide to their public health consequences, monitoring and management. World Health Organization, 440 p., E&FN Spon, London
Collier JL, Grossman AR (1992) Chlorosis induced by nutrient deprivation in Synechococcus sp. strain PCC 7942: not all bleaching is the same. J Bacteriol 174(14):4718–4726
Conn SJ, Hocking B, Dayod M, Xu B, Atham A, Henderson S, Aukett L, Conn V, Shearer MK, Fuentes S, Tyerman SD, Gilliham M (2013) Protocol: optimising hydroponic growth systems for nutritional and physiological analysis of Arabidopsis thaliana and other plants. Plant Methods 9(1):1–11
Dagnino D, De Abreu Meireles D, De Aquino Almeida JC (2006) Growth of nutrient-replete Microcystis PCC 7806 cultures is inhibited by an extracellular signal produced by chlorotic cultures. Environ Microbiol 8(1):30–36
Davis ML, Cornwell DA (2013) Introduction to Environmental Engineering. New York (US)
Dubelaar GBJ, Onker RR (2000) Flow cytometry as a tool for the study of phytoplankton. Sci Mar 64(2):135–156
Dushenkov V, Kumar PN, Motto H, Raskin I (1995) Rhizofiltration: the use of plants to remove heavy metals from aqueous streams. Environ Sci Technol 29:1239–1245
Fan J, Ho L, Hobson P, Brookes J (2013) Evaluating the effectiveness of copper sulphate, chlorine, potassium permanganate, hydrogen peroxide and ozone on cyanobacterial cell integrity. Water Res 47:5153–5164
Faulwetter JL, Gagnon V, Sundberg C, Chazarenc F, Burr MD, Brisson J, Camper AK, Stein OR (2009) Microbial processes influencing performance of treatment wetlands: a review. Ecol Eng 35(6):987–1004
Flörke M, Schneider C, McDonald RI (2018) Water competition between cities and agriculture driven by climate change and urban growth. Nat Sustain 1(1):51
Ger KA, Hansson L-A, Lürling M (2014) Understanding cyanobacteria-zooplankton interactions in a more eutrophic world. Freshw Biol 59:1783–1798
Gross EM (2003) Allelopathy of aquatic autotrophs. Crit Rev Plant Sci 22:313–339
Headley TR, Tanner CC (2012) Constructed wetlands with floating emergent macrophytes: an innovative stormwater treatment technology. Crit Rev Environ Sci Technol 42:2261–2310
Hilt S, Gross EM (2008) Can allelopathically active submerged macrophytes stabilise clear-water states in shallow lakes? Basic Appl Ecol 9:422–432
Hong Y, Hu HY, Xie X, Sakoda A, Sagehashi M, Li FM (2009) Gramine-induced growth inhibition, oxidative damage and antioxidant responses in freshwater cyanobacterium Microcystis aeruginosa. Aquat Toxicol 91:262–269
Hudnell KH (2010) The state of US freshwater harmful algal blooms assessments, policy and legislation. Toxicon, 55 (5), SI, 1024-1034
Huisman J, Codd GA, Paerl HW, Ibelings BW, Verspagen JM, Visser PM (2018) Cyanobacterial blooms. Nat Rev Microbiol 16(8):471
Imai H, Chang KH, Kusaba M, Nakano SI (2008) Temperature-dependent dominance of Microcystis (Cyanophyceae) species: M. aeruginosa and M. wesenbergii. J Plankton Res 31:171–178
Jancula D, Marsalek B (2011) Critical review of actually available chemical compounds for prevention and management of cyanobacterial blooms. Chemosphere 85:1415–1422
Körner S, Nicklisch A (2002) Allelopathic growth inhibition of selected phytoplankton species by submerged macrophytes1. J Phycol 38(5):862–871
Krasikov V, Aguirre von Wobeser E, Dekker HL, Huisman J, Matthijs HC (2012) Time-series resolution of gradual nitrogen starvation and its impact on photosynthesis in the cyanobacterium Synechocystis PCC 6803. Physiol Plant 145(3):426–439
Kurzbaum E, Kirzhener F, Armon R (2012) Improvement of water quality using constructed wetland systems. Rev Environ Health 27(1):59–64
Laue P, Bahrs H, Chakrabarti S, Steinberg CE (2014) Natural xenobiotics to prevent cyanobacterial and algal growth in freshwater: contrasting efficacy of tannic acid, gallic acid, and gramine. Chemosphere 104:212–220
Le Moal M, Gascuel-Odoux C, Ménesguen A, Souchon Y, Étrillard C, Levain A, Moatar F, Pannard A, Souchu P, Lefebvre A, Pinay G (2019) Eutrophication: a new wine in an old bottle? Sci Total Environ 651:1–11
Lu H, Yuan Y, Campbell DE, Qin P, Cui L (2014) Integrated water quality, energy and economic evaluation of three bioremediation treatment systems for eutrophic water. Ecol Eng 69:244–254
Lürling M, De Senerpont Domis LN (2013) Predictability of plankton communities in an unpredictable world. Freshwat Biol 58:455–462
Lürling M, Va Oosterhout F (2013) Controlling eutrophication by combined bloom precipitation and sediment phosphorus inactivation. Water Res 47:6527–6537
Marchand L, Nsanganwimana F, Oustriere N, Grebenshchykova Z, Lizama-Allende K, Mench M (2014) Copper removal from water using a bio-rack system either unplanted or planted with Phragmites australis, Juncus articulatus and Phalaris arundinacea. Ecol Eng 64:291–300
Martinez Hernandez J, Lopez-Rodas V, Costas E (2009) Microcystins from tap water could be a risk factor for liver and colorectal cancer: a risk intensified by global change. Med Hypotheses 72(5):539–540
Mulderij G, Van Nes EH, Van Donk E (2007) Macrophyte–phytoplankton interactions: the relative importance of allelopathy versus other factors. Ecol Model 204(1):85–92
Murray D, Jefferson B, Jarvis P, Parsons SA (2010) Inhibition of three algae species using chemicals released from barley straw. Environ Technol 31(4):455–466
O’Neil JM, Davis TW, Burford MA, Gobler CJ (2012) The rise of harmful cyanobacteria blooms: the potential roles of eutrophication and climate change. Harmful Algae 14:313–334
Otten TG, Paerl HW (2015) Health effects of toxic cyanobacteria in US drinking and recreational waters: our current understanding and proposed direction. Curr Environ Health Rep 2(1):75–84
Potier O, Leclerc JP, Pons MN (2005) Influence of geometrical and operational parameters on the axial dispersion in an aerated channel reactor. Water Res 39(18):4454–4462
Qin B, Paerl HW, Brookes JD, Liu J, Jeppesen E, Zhu G, Zhang Y, Xu H, Shi K, Deng J (2019) Why Lake Taihu continues to be plagued with cyanobacterial blooms through 10 years (2007-2017) efforts? Sci Bull 64:354–356. https://doi.org/10.1016/j.scib.2019.02.008
Raskin I, Ensley BD (2000) Phytoremediation of toxic metals: using plants to clean up the environment. John Wiley & Sons, Chichester, p 304
Rippka R, Deruelles J, Waterbutry JB, Herdman M, Staner RY (1979) Generic assignments, strain histories, and properties of pure cultures of cyanobacteria. J Gen Microbiol 11(1):1–61
Rojo C, Segura M, Rodrigo MA (2013) The allelopathic capacity of submerged macrophytes shapes the microalgal assemblages from a recently restored coastal wetland. Ecol Eng 58:149–155
Ruggeri B, Sassi G (1993) On the modelling approaches of biomass behaviour in bioreactor. Chem Eng Commun 122:1–56
Shao J, LIi R, Lepo JE Gu J-D (2013) Potential for control of harmful cyanobacterial blooms using biologically derived substances: Problems and prospects. J Environ Manag 125:149–155
U.S. EPA. 2005. Membrane filtration guidance manual, EPA 815-R-06-009. USEPA, Washington, DC
Vézie C, Rapala J, Vaitomaa J, Seitsonen J, Sivonen K (2002) Effect of nitrogen and phosphorus on growth of toxic and nontoxic microcystis strains and on intracellular microcystin concentrations. Microb Ecol 43(4):443–454
Weiss P, Westbrook A, Weiss J, Gulliver J, Bierboer D (2014) Effect of water velocity on hydroponic phytoremediation of metals. Int J Phytoremediation 16:203–217
Wolfe GV (2000) The chemical defense ecology of marine unicellular plankton: constraints, mechanisms, and impacts. Biol Bull 198(2):225–244
Xie L, Hanyu T, Futatsugi N, Komatsu M, Steinman AD Park HD (2014) Inhibitory effect of naringin on microcystin-LR uptake in the freshwater snail Sinotaia histrica. Environ Toxicol Pharmacol 38(2):430–437
Acknowledgements
From Thames Water, the authors thank specially Dr. Paul Rutter for his support and Nick Sutherland for assisting with the experimental set-up logistics. Thanks to Helen Shapland and Kevin O’Reilly, Trace Organics laboratory, Scientific Centre, Wessex Water Ltd., for allowing us the use of their HPLC. The authors are especially grateful to Toni Hall, Alexander T. Jennings and Gary Hunt for their assistance with developing the HPLC allelochemicals method.
Funding
This project was fully sponsored by Thames Water Utilities Ltd. The design of the study presented in this manuscript, collection, analysis, and interpretation of data, and writing the manuscript are the views of the authors. Lady Margaret Hall, University of Oxford sponsored attendance to conferences where aspects of this work were presented.
Author information
Authors and Affiliations
Contributions
Conceptualization, experimental design, methodology, material preparation, data collection, formal analysis, investigation, writing original draft, and reviewing and editing were performed by Ana T. Castro Castellon. Resources, experimental design, data analysis, and validation were performed by Daniel S. Read. Conceptualization and writing original draft: Yaldah Azimi and Jocelyne M.R. Hughes. Supervision, project administration and resources: Jocelyne M.R. Hughes, Nicholas P. Hankins, and Michael J. Chipps. Funding acquisition by Michael J. Chipps. All authors reviewed, commented, and edited on previous versions of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Responsible editor: Vitor Manuel Oliveira Vasconcelos
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Castro-Castellon, A.T., Hughes, J.M.R., Read, D.S. et al. The role of rhizofiltration and allelopathy on the removal of cyanobacteria in a continuous flow system. Environ Sci Pollut Res 28, 27731–27741 (2021). https://doi.org/10.1007/s11356-021-12343-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-12343-9