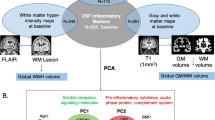
Abstract
While MRI brain changes have been related to mortality during ageing, the role of inflammation in this relationship remains poorly understood. Hence, this study aimed to investigate the impact of MRI changes on all-cause mortality and the mediating role of cytokines. All-cause mortality was evaluated in 268 community dwelling elderly (age 65–83 years) in the MEMO study (Memory and Morbidity in Augsburg elderly). MRI markers of brain atrophy and cerebral small vessel disease (SVD), C-reactive protein (CRP) and a panel of cytokines in serum were assessed. Cox proportional hazard models were used to estimate the association of MRI changes with survival over 9 years. Regression models were used to assess the hypothesis that inflammation is mediating the relationship between MRI-brain changes and mortality. In total, 77 (29 %) deaths occurred during a mean follow up of 9 years. After adjusting for confounders, the degree of global cortical atrophy and the level of the cytokines CRP, TNF-α and IL-8 were of higher significance in study participants who had died at follow-up in comparison to survivors. In Cox proportional hazard models, higher degrees of global cortical atrophy (HR 1.56, p = 0.003) and regional atrophy of the temporal lobe (HR 1.38, p = 0.011) were associated with a significantly increased risk of mortality. Mediation analyses revealed a partial mediation by IL-6 and IL-8 of the effects of global cortical atrophy on mortality. Global cortical brain atrophy is a significant indicator of survival in the elderly. Our study supports a possible role for inflammation in the atrophy pathogenesis. If replicated in other samples, IL-6 and IL-8 level assessment may improve risk prognosis for mortality.
Similar content being viewed by others
References
Amor S, Puentes F, Baker D, van der Valk P (2010) Inflammation in neurodegenerative diseases. Immunology 129:154–169. doi:10.1111/j.1365-2567.2009.03225.x
Baron RM, Kenny DA (1986) The moderator–mediator variable distinction in social psychological research: conceptual, strategic, and statistical considerations. J Pers Soc Psychol 51:1173–1182
Baune BT, Ponath G, Rothermundt M et al (2009) Association between cytokines and cerebral MRI changes in the aging brain. J Geriatr Psychiatry Neurol 22:23–34. doi:10.1177/0891988708328216
Baune BT, Rothermundt M, Ladwig KH et al (2010) Systemic inflammation (interleukin 6) predicts all-cause mortality in men: results from a 9-year follow-up of the MEMO study. Age 33:209–217. doi:10.1007/s11357-010-9165-5
Berger K, Hense HW, Rothdach A et al (2000) A single question about prior stroke versus a stroke questionnaire to assess stroke prevalence in populations. Neuroepidemiology 19:245–257
Berger K, Kase CS, Buring JE (1996) Interobserver agreement in the classification of stroke in the physicians' health study. Stroke 27:238–242
Blom JW, de Ruijter W, Witteman JCM et al (2013) Changing prediction of mortality by systolic blood pressure with increasing age: the Rotterdam study. Age 35:431–438. doi:10.1007/s11357-011-9349-7
Bokura H, Kobayashi S, Yamaguchi S et al (2006) Silent brain infarction and subcortical white matter lesions increase the risk of stroke and mortality: a prospective cohort study. J Stroke Cerebrovasc Dis 15:57–63. doi:10.1016/j.jstrokecerebrovasdis.2005.11.001
Briley DP, Haroon S, Sergent SM, Thomas S (2000) Does leukoaraiosis predict morbidity and mortality? Neurology 54:90–94
Bruunsgaard H, Ladelund S, Pedersen AN et al (2003) Predicting death from tumour necrosis factor-alpha and interleukin-6 in 80-year-old people. Clin Exp Immunol 132:24–31
Bruunsgaard H, Pedersen BK (2003) Age-related inflammatory cytokines and disease. Immunol Allergy Clin N Am 23:15–39
Cefalu WT, Wagner JD (1997) Aging and atherosclerosis in human and nonhuman primates. Age 20:15–28. doi:10.1007/s11357-997-0002-4
Conijn MMA, Kloppenborg RP, Algra A et al (2011) Cerebral small vessel disease and risk of death, ischemic stroke, and cardiac complications in patients with atherosclerotic disease: the second manifestations of ARTerial disease-magnetic resonance (SMART-MR) study. Stroke; a journal of cerebral circulation 42:3105–3109. doi:10.1161/STROKEAHA.110.594853
Davidson KW, Rieckmann N, Rapp MA (2005) Definitions and distinctions among depressive syndromes and symptoms: implications for a better understanding of the depression-cardiovascular disease association. Psychosom Med 67(Suppl 1):S6–S9. doi:10.1097/01.psy.0000162257.19266.fc
de Leeuw FE, de Groot JC, Achten E et al (2001) Prevalence of cerebral white matter lesions in elderly people: a population based magnetic resonance imaging study. The Rotterdam scan study. J Neurol Neurosurg Psychiatry 70:9–14
Frazier PA, Tix AP, Barron KE (2004) Testing Moderator and Mediator Effects in Counseling Psychology Research. 51:115–134.
Hassan A, Hunt BJ, O'Sullivan M et al (2003) Markers of endothelial dysfunction in lacunar infarction and ischaemic leukoaraiosis. Brain 126:424–432
Hauss-Wegrzyniak B, Vraniak PD, Wenk GL (2000) LPS-induced neuroinflammatory effects do not recover with time. Neuroreport 11:1759–1763
Ikram MA, Vernooij MW, Vrooman HA et al (2009) Brain tissue volumes and small vessel disease in relation to the risk of mortality. Neurobiol Aging 30:450–456. doi:10.1016/j.neurobiolaging.2007.07.009
Jefferson AL, Massaro JM, Wolf PA et al (2007) Inflammatory biomarkers are associated with total brain volume: the Framingham heart study. Neurology 68:1032–1038. doi:10.1212/01.wnl.0000257815.20548.df
Jochemsen HM, Muller M, Visseren FL et al (2013) Blood pressure and progression of brain atrophy: the SMART-MR study. JAMA neurology 70:1046–1053. doi:10.1001/jamaneurol.2013.217
Keil U, Liese AD, Hense HW et al (1998) Classical risk factors and their impact on incident non-fatal and fatal myocardial infarction and all-cause mortality in southern Germany. Results from the MONICA Augsburg cohort study 1984–1992. Monitoring trends and determinants in cardiovascular diseases. Eur Heart J 19:1197–1207
Kuller LH, Arnold AM, Longstreth WT et al (2007) White matter grade and ventricular volume on brain MRI as markers of longevity in the cardiovascular health study. Neurobiol Aging 28:1307–1315. doi:10.1016/j.neurobiolaging.2006.06.010
Launer LJ, Oudkerk M, Nilsson LG et al (2000) CASCADE: a European collaborative study on vascular determinants of brain lesions. Study design and objectives Neuroepidemiology 19:113–120
Melton LM, Keith AB, Davis S et al (2003) Chronic glial activation, neurodegeneration, and APP immunoreactive deposits following acute administration of double-stranded RNA. Glia 44:1–12. doi:10.1002/glia.10276
Mitaki S, Nagai A, Oguro H, Yamaguchi S (2016) C-reactive protein levels are associated with cerebral small vessel-related lesions. Acta Neurol Scand 133:68–74. doi:10.1111/ane.12440
Mraovitch S (2003) Isocortical hyperemia and allocortical inflammation and atrophy following generalized convulsive seizures of thalamic origin in the rat. Cell Mol Neurobiol 23:773–791
Olesen PJ, Guo X, Gustafson D et al (2011) A population-based study on the influence of brain atrophy on 20-year survival after age 85. Neurology 76:879–886. doi:10.1212/WNL.0b013e31820f2e26
Pantoni L (2010) Cerebral small vessel disease: from pathogenesis and clinical characteristics to therapeutic challenges. Lancet Neurol 9:689–701. doi:10.1016/S1474-4422(10)70104-6
Samaras K, Lutgers HL, Kochan NA et al (2014) The impact of glucose disorders on cognition and brain volumes in the elderly: the Sydney memory and ageing study. Age 36:977–993. doi:10.1007/s11357-013-9613-0
Scheltens P, Erkinjunti T, Leys D et al (1998) White matter changes on CT and MRI: an overview of visual rating scales. European task force on age-related white matter changes. Eur Neurol 39:80–89
Schmidt WP, Roesler A, Kretzschmar K et al (2004) Functional and cognitive consequences of silent stroke discovered using brain magnetic resonance imaging in an elderly population. J Am Geriatr Soc 52:1045–1050. doi:10.1111/j.1532-5415.2004.52300.x
Sexton CE, Storsve AB, Walhovd KB et al (2014) Poor sleep quality is associated with increased cortical atrophy in community-dwelling adults. Neurology 83:967–973. doi:10.1212/WNL.0000000000000774
Shrout PE, Bolger N (2002) Mediation in experimental and nonexperimental studies: new procedures and recommendations. Psychol Methods 7:422–445
Staff RT, Murray AD, Ahearn T et al (2010) Brain volume and survival from age 78 to 85: the contribution of Alzheimer-type magnetic resonance imaging findings. J Am Geriatr Soc 58:688–695. doi:10.1111/j.1532-5415.2010.02765.x
Störk S, Feelders RA, van den Beld AW et al (2006) Prediction of mortality risk in the elderly. Am J Med 119:519–525. doi:10.1016/j.amjmed.2005.10.062
Sullivan GW, Sarembock IJ, Linden J (2000) The role of inflammation in vascular diseases. J Leukoc Biol 67:591–602
Swan GE, DeCarli C, Miller BL et al (1998) Association of midlife blood pressure to late-life cognitive decline and brain morphology. Neurology 51:986–993
Tabarean I, Morrison B, Marcondes MC et al (2010) Hypothalamic and dietary control of temperature-mediated longevity. Ageing Res Rev 9:41–50. doi:10.1016/j.arr.2009.07.004
Trollor JN, Smith E, Agars E et al (2011) The association between systemic inflammation and cognitive performance in the elderly: the Sydney memory and ageing study. Age 34:1295–1308. doi:10.1007/s11357-011-9301-x
van Dijk EJ, Prins ND, Vermeer SE et al (2005) C-reactive protein and cerebral small-vessel disease: the Rotterdam scan study. Circulation 112:900–905. doi:10.1161/CIRCULATIONAHA.104.506337
Vermeer SE, Prins ND, Heijer den T et al (2003) Silent brain infarcts and the risk of dementia and cognitive decline. N Engl J Med 348:1215–1222. doi:10.1056/NEJMoa022066
Vila E, Salaices M (2005) Cytokines and vascular reactivity in resistance arteries. Am J Physiol Heart Circ Physiol 288:H1016–H1021. doi:10.1152/ajpheart.00779.2004
Ward MA, Carlsson CM, Trivedi MA et al (2005) The effect of body mass index on global brain volume in middle-aged adults: a cross sectional study. BMC Neurol 5:23. doi:10.1186/1471-2377-5-23
Wardlaw JM, Sandercock PA, Dennis MS, Starr J (2003) Is breakdown of the blood-brain barrier responsible for lacunar stroke, leukoaraiosis, and dementia? Stroke; a journal of cerebral circulation 34:806–812. doi:10.1161/01.STR.0000058480.77236.B3
Willette AA, Coe CL, Birdsill AC et al (2013) Interleukin-8 and interleukin-10, brain volume and microstructure, and the influence of calorie restriction in old rhesus macaques. Age 35:2215–2227. doi:10.1007/s11357-013-9518-y
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Source of funding
The MEMO study is supported by the German Research Society (DFG, grant: BE1996/1–1). Data assessment was carried out within the framework of the Cooperative Health Research in the Augsburg Region (KORA). This study was supported by the Federal German Ministry of Education and Research (BMBF, grant 01ER1205). Dr. U. Hanning receives a research grant of the Medical Faculty of the University of Muenster.
About this article
Cite this article
Hanning, U., Roesler, A., Peters, A. et al. Structural brain changes and all-cause mortality in the elderly population—the mediating role of inflammation. AGE 38, 455–464 (2016). https://doi.org/10.1007/s11357-016-9951-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-016-9951-9