Abstract
Purpose
Methanotrophs are an important group of methane (CH4)-oxidizing bacteria in the soil, which act as a major sink for the greenhouse gas, CH4. In grazed grassland, one of the ecologically most sensitive areas is the animal urine patch soil, which is a major source of both nitrate (NO3 −) leaching and nitrous oxide (N2O) emissions. Nitrification inhibitors, such as dicyandiamide (DCD), have been used to mitigate NO3 − leaching and N2O emissions in grazed pastures. However, it is not clear if the high nitrogen loading rate in the animal urine patch soil and the use of nitrification inhibitors would have an impact on the abundance of methanotrophs in grazed grassland soils. The purpose of this study was to determine the effect of animal urine and DCD on methanotroph abundance in grazed grassland soils.
Materials and methods
A laboratory incubation study was conducted to determine the effect of urine and DCD applications on the abundance of methanotrophs in six grazed grassland soils sampled from across New Zealand, using real-time PCR targeting the functional pmoA gene.
Results and discussion
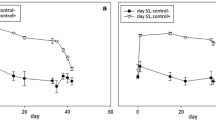
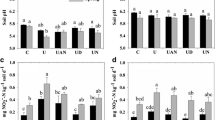
Results showed that the pmoA gene copy numbers were low in these soils, mostly below 2.36 × 104 g−1 soil except in the West Coast soil where pmoA gene copy number reached 8.95 × 105 g−1 soil. Most of the clones identified were aligned to the type II methanotrophs. There was no significant effect (P < 0.05) on the abundance of methanotrophs by the applications of urine at 1,000 kg N ha−1 or DCD at 10 kg ha−1.
Conclusions
These results suggest that the abundance of methanotrophs is not affected by urine deposition or the application of DCD to mitigate NO3 − leaching and N2O emissions in grazed grassland soils.



Similar content being viewed by others
References
Ball BC, Hunter R (1988) The determination of water release characteristics of soil cores at low suctions. Geoderma 43:195–212
Bedard C, Knowles R (1989) Physiology, biochemistry and specific inhibitors of CH4, NH4 and CO oxidation by methanotrophs and nitrifiers. Microbiol Rev 53:68–84
Bodelier PLE, Laanbroek HJ (2004) Nitrogen as a regulatory factor of methane oxidation in soils and sediments. FEMS Microbiol Ecol 47:265–277
Bodelier PLE, Roslev P, Henckel T, Frenzel P (2000) Stimulation by ammonium-based fertilizers of methane oxidation in soil around rice roots. Nature 403:421–424
Costello AM, Lidstrom ME (1999) Molecular characterization of functional and phylogenetic genes from natural populations of methanotrophs in lake sediments. Appl Environ Microbiol 65:5066–5074
De Klein CAM, Barton L, Sherlock RR, Li Z, Littlejohn RP (2003) Estimating a nitrous oxide emission factor for animal urine from some New Zealand pastoral soils. Aust J Soil 41:381–399
Di HJ, Cameron KC (2002a) Nitrate leaching in temperate agroecosystems: sources, factors and mitigating strategies. Nutr Cycl Agroecosyst 64:237–256
Di HJ, Cameron KC (2002b) The use of a nitrification inhibitor, dicyandiamide (DCD), to reduce nitrate leaching and nitrous oxide emissions in a simulated grazed and irrigated grassland. Soil Use Manage 18:395–403
Di HJ, Cameron KC (2005) Reducing environmental impacts of agriculture by using a fine particle suspension nitrification inhibitor to decrease nitrate leaching from grazed pastures. Agric Ecosyst Environ 109:202–212
Di HJ, Cameron KC, Sherlock RR (2007) Comparison of the effectiveness of a nitrification inhibitor, dicyandiamide (DCD), in reducing nitrous oxide emissions in four different soils under different climatic and management conditions. Soil Use Manage 23:1–9
Di HJ, Cameron KC, Shen JP, Winefield CS, O’Callaghan M, Bowatte S, He JZ (2009a) Nitrification driven by bacteria and not archaea in nitrogen rich grassland soils. Nat Geosci 2:621–624
Di HJ, Cameron KC, Shen JP, He JZ, Winefield CS (2009b) Nitrate leaching from grazed grassland as affected by a nitrification inhibitor, dicyandiamide, and relationships with ammonia oxidizing bacteria and archaea. Soil Use Manage 25:454–461
Di HJ, Cameron KC, Sherlock RR, Shen JP, He JZ, Winefield CS (2010a) Nitrous oxide emissions from grazed grassland as affected by a nitrification inhibitor, dicyandiamide, and relationships with ammonia oxidizing bacteria and archaea. J Soils Sediments 10:943–954
Di HJ, Cameron KC, Shen JP, Winefield CS, O’Callaghan M, Bowatte S, He JZ (2010b) Ammonia oxidizing bacteria and archaea grow under contrasting soil nitrogen conditions. FEMS Microbiol Ecol 72:386–394
Gilbert B, McDonald IR, Finch R, Stafford GP, Nielsen AK, Murrell JC (2000) Molecular analysis of the pmo (particulate methane monooxygenase) operons from two type II methanotrophs. Appl Environ Microbiol 66:966–975
Hanson HS, Hanson TE (1996) Methanotrophic bacteria. Microbiol Rev 60:439–471
Hewitt AE (1998) New Zealand soil classification, 2nd edn. Manaaki Whenua, Lincoln, Canterbury, New Zealand
Horz HP, Rich V, Avrahami S, Bohannan BJM (2005) Methane-oxidizing bacteria in a California upland grassland soil: diversity and response to simulated global change. Appl Environ Microbiol 71:2642–2652
Jarvis SC (1997) Emission processes and their interactions in grassland soils. In: Jarvis SC, Pain BF (eds) Gaseous nitrogen emissions from grasslands. CAB International, Wellingford, pp 1–17
Kowalchuk GA, Stephen JR (2001) Ammonia-oxidizing bacteria: a model for molecular microbial ecology. Annu Rev Microbiol 55:485–529
Kumar S, Tamura K, Nei M (2004) MEGA3: integrated software for molecular evolutionary genetics analysis and sequence alignment. Brief Bioinform 5:150–163
McDonald IR, Bodrossy L, Chen Y, Murrell JC (2008) Molecular ecology techniques for the study of aerobic methanotrophs. Appl Environ Microbiol 74:1305–1315
MFE (2009) New Zealand’s greenhouse gas inventory 1990–2007. Ministry for the Environment, Wellington
Mosier AR, Schimel DS, Valentine DW, Bronson K, Parton WJ (1991) Methane and nitrous oxide fluxes in native fertilized and cultivated grasslands. Nature 350:330–332
Purkhold U, Pommerening-Röser A, Juretschko S, Schmid MC, Koops HJ, Wagner M (2000) Phylogeny of all recognized species of ammonia oxidizers based on comparative 16S rRNA and amoA sequence analysis: implications for molecular diversity surveys. Appl Environ Microbiol 66:5368–5382
Saggar S, Tate R, Giltrap DL, Singh J (2008) Soil–atmosphere exchange of nitrous oxide and methane in New Zealand terrestrial ecosystems and their mitigation options: a review. Plant Soil 309:25–42
Semrau JD, Chistoserdov A, Lebron J, Costello AM, Davagnino J, Kenna EM, Holmes AJ, Finch R, Murrell JC, Lidstrom ME (1995) Particulate methane monooxygenase genes in methanotrophs. J Bacteriol 177:3071–3079
Singh BK, Tate KR, Ross DJ, Singh J, Dando J, Thomas N, Millard P, Murrell C (2009) Soil methane oxidation and methanotroph responses to afforestation of pastures with Pinus radiata stands. Soil Biol Biochem 41:2196–2205
Smith KS, Costello AM, Lidstrom ME (1997) Methane and trichloroethylene oxidation by an estuarine methanotroph, Methylobacter sp. strain BB5.1. Appl Environ Microbiol 63:4617–4620
Smith KA, Dobbie KE, Ball BC, Bakken LR, Sitaula BK, Hansen S, Brumme R, Borken W, Christensen S, Priemé A, Fowler D, Macdonald JA, Skiba U, Klemedtsson L, Kasimir-Klemedtsson A, Degórska A, Orlanski P (2000) Oxidation of atmospheric methane in Northern European soils, comparison with other ecosystems, and uncertainties in the global terrestrial sink. Glob Chang Biol 6:791–803
Soil Survey Staff (1998) Keys to soil taxonomy, 8th edn. United States Department of Agriculture, Washington, DC
Steudler PA, Bowden RD, Mellilo JM, Aber JD (1989) Influence of nitrogen fertilization on methane uptake in temperate forest soil. Nature 341:314–316
Stolyar S, Costello AM, Peeples TL, Lidstrom ME (1999) Role of multiple gene copies in particulate methane monooxygenase activity in the methane-oxidizing bacterium Methylococcus capsulatus Bath. Microbiology 145:1235–1244
Tate KR, Ross DJ, Saggar S, Hedley CB, Dando J, Singh BK (2007) Methane uptake in soils from Pinus radiata plantations, a reverting shrubland and adjacent pastures: effects of landuse change, and soil texture, water and mineral nitrogen. Soil Biol Biochem 39:1437–1448
Willison TW, Webster CP, Goulding KWT, Powlson DS (1995) Methane oxidation in temperate soils: effects of land use and the chemical form of nitrogen fertilizer. Chemosphere 30:539–546
Zheng Y, Zhang LM, Zheng YM, Di HJ, He JZ (2008) Abundance and community composition of methanotrophs in a Chinese paddy soil under long-term fertilization practices. J Soils Sediments 8:406–414
Acknowledgments
We would like to thank the New Zealand Foundation for Research, Science and Technology (FRST) for the funding; Drs. Ross Monaghan, Stewart Ledgard, and Mark Sheppard of AgResearch and Drs. Bruce Thorrold and Deanne Waugh of Dairy NZ for their assistance with soil sampling and Emily Gerard and Shona Brock of AgResearch and Jie Lei, Steve Moore, Carole Barlow, Trevor Hendry, and Neil Smith of Lincoln University for their technical support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Chengrong Chen
Rights and permissions
About this article
Cite this article
Di, H.J., Cameron, K.C., Shen, JP. et al. Methanotroph abundance not affected by applications of animal urine and a nitrification inhibitor, dicyandiamide, in six grazed grassland soils. J Soils Sediments 11, 432–439 (2011). https://doi.org/10.1007/s11368-010-0318-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-010-0318-z