Abstract
Purpose
Global climate change, in particular temperature variation, is likely to alter soil microbial abundance and composition, with consequent impacts on soil biogeochemical cycling and ecosystem functioning. However, responses of belowground nitrogen transformation microorganisms to temperature changes in high-elevation terrestrial ecosystems are not well understood.
Materials and methods
Here, the effects of simulated cooling and warming on the abundance and community composition of ammonia-oxidizing archaea (AOA) and bacteria (AOB), as well as the abundance of denitrifiers, were investigated using quantitative polymerase chain reaction and clone library approaches, on the basis of a 2-year reciprocal elevation translocation experiment along an elevation gradient from 3,200 to 3,800 m above sea level on the Tibetan Plateau.
Results and discussion
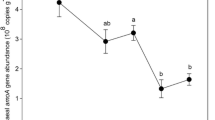
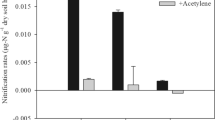
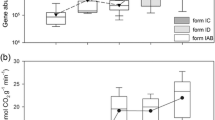
We found that, compared with the temperature variations caused by elevation translocation, the soil origin exerted a much stronger influence on AOA abundance. There were significant effects of both soil origin and elevation translocation on AOB abundance, which was particularly decreased by elevation-enhanced (simulated cooling) and increased by elevation-decreased (simulated warming) treatments. Altered temperature affected the abundance of nirK rather than nirS and nosZ genes, and the latter two seemed to be associated tightly with the soil origin. Furthermore, the results showed that temperature changes had obvious influences on the community structure and diversity of AOB, but not AOA. More apparent response of AOB to warming than in other studies on grassland and forest ecosystems may be attributed to higher elevation and lower mean annual temperature in this study.
Conclusions
Our findings thus suggest that, in comparison with AOA and denitrifying populations, AOB may respond more sensitively to natural temperature variation caused by elevation translocation in this alpine grassland ecosystem on the Tibetan Plateau.






Similar content being viewed by others
References
Abell GCJ, Revill AT, Smith C, Bissett AP, Volkman JK, Robert SS (2010) Archaeal ammonia oxidizers and nirS-type denitrifiers dominate sediment nitrifying and denitrifying populations in a subtropical macrotidal estuary. ISME J 4:286–300
Ai C, Liang GQ, Sun JW, Wang XB, He P, Zhou W (2013) Different roles of rhizosphere effect and long-term fertilization in the activity and community structure of ammonia oxidizers in a calcareous fluvo-aquic soil. Soil Biol Biochem 57:30–42
Avrahami S, Liesack W, Conrad R (2003) Effects of temperature and fertilizer on activity and community structure of soil ammonia oxidizers. Environ Microbiol 5:691–705
Bertrand R, Lenoir J, Piedallu C, Riofrío-Dillon G, de Ruffray P, Vidal C, Pierrat JC, Gégout JC (2011) Changes in plant community composition lag behind climate warming in lowland forests. Nature 479:517–520
Cantarel AAM, Bloor JMG, Pommier T, Guillaumaud N, Moirot C, Soussana JF, Poly F (2012) Four years of experimental climate change modifies the microbial drivers of N2O fluxes in an upland grassland ecosystem. Glob Chang Biol 18:2520–2531
Chen X, Zhang LM, Shen JP, Xu ZH, He JZ (2010a) Soil type determines the abundance and community structure of ammonia-oxidizing bacteria and archaea in flooded paddy soils. J Soils Sediments 10:1510–1516
Chen Z, Luo XQ, Hu RG, Wu MN, Wu JS, Wei WX (2010b) Impact of long-term fertilization on the composition of denitrifier communities based on nitrite reductase analyses in a paddy soil. Microb Ecol 60:850–861
Chen YL, Xu ZW, Hu HW, Hu YJ, Hao ZP, Jiang Y, Chen BD (2013) Responses of ammonia-oxidizing bacteria and archaea to nitrogen fertilization and precipitation increment in a typical temperate steppe in Inner Mongolia. Appl Soil Ecol 68:36–45
Clark IM, Buchkina N, Jhurreea D, Goulding KWT, Hirsch PR (2012) Impacts of nitrogen application rates on the activity and diversity of denitrifying bacteria in the Broadbalk Wheat Experiment. Philos Trans R Soc B Biol Sci 367:1235–1244
Daebeler A, Abell GCJ, Bodelier PLE, Bodrossy L, Frampton DMF, Hefting MM, Laanbroek HJ (2012) Archaeal dominated ammonia-oxidizing communities in Icelandic grassland soils are moderately affected by long-term N fertilization and geothermal heating. Front Microbiol 3:352
Dandie CE, Burton DL, Zebarth BJ, Henderson SL, Trevors JT, Goyer C (2008) Changes in bacterial denitrifier community abundance over time in an agricultural field and their relationship with denitrification activity. Appl Environ Microbiol 74:5997–6005
Dijkstra FA, Blumenthal D, Morgan JA, Pendall E, Carrillo Y, Follett RF (2010) Contrasting effects of elevated CO2 and warming on nitrogen cycling in a semiarid grassland. New Phytol 187:426–437
Duan JC, Wang SP, Zhang ZH, Xu GP, Luo CY, Chang XF, Zhu XX, Cui SJ, Zhao XQ, Wang WY, Du MY (2013) Non-additive effect of species diversity and temperature sensitivity of mixed litter decomposition in the alpine meadow on Tibetan Plateau. Soil Biol Biochem 57:841–847
Enwall K, Philippot L, Hallin S (2005) Activity and composition of the denitrifying bacterial community respond differently to long-term fertilization. Appl Environ Microbiol 71:8335–8343
Fierer N, Carney KM, Horner-Devine MC, Megonigal JP (2009) The biogeography of ammonia-oxidizing bacterial communities in soil. Microb Ecol 58:435–445
He JS, Wang ZH, Wang XP, Schmid B, Zuo WY, Zhou M, Zheng CY, Wang MF, Fang JY (2006) A test of the generality of leaf trait relationships on the Tibetan Plateau. New Phytol 170:835–848
Horz HP, Barbrook A, Field CB, Bohannan BJM (2004) Ammonia-oxidizing bacteria respond to multifactorial global change. Proc Natl Acad Sci U S A 101:15136–15141
IPCC (2007) In: Solomon S, Qin D, Manning M, Chen Z, Marquis M, Averyt KB, Tignor M, Miller HL (eds) Summary for policymakers. Climate change 2007: the physical science basis. Contribution of working group I to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge, pp 539–543
Jung J, Yeom J, Kim J, Han J, Lim HS, Park H, Hyun S, Park W (2011) Change in gene abundance in the nitrogen biogeochemical cycle with temperature and nitrogen addition in Antarctic soils. Res Microbiol 162:1018–1026
Long X, Chen CR, Xu ZH, Linder S, He JZ (2012a) Abundance and community structure of ammonia oxidizing bacteria and archaea in a Sweden boreal forest soil under 19-year fertilization and 12-year warming. J Soils Sediments 12:1124–1133
Long X, Chen CR, Xu ZH, Oren R, He JZ (2012b) Abundance and community structure of ammonia-oxidizing bacteria and archaea in a temperate forest ecosystem under ten-years elevated CO2. Soil Biol Biochem 46:163–171
Luo YQ, Sherry R, Zhou XH, Wan SQ (2009) Terrestrial carbon-cycle feedback to climate warming: experimental evidence on plant regulation and impacts of biofuel feedstock harvest. GCB Bioenergy 1:62–74
Luo CY, Xu GP, Chao ZG, Wang SP, Lin XW, Hu YG, Zhang ZH, Duan JC, Chang XF, Su AL, Li YN, Zhao XQ, Du MY, Tang YH, Kimball B (2010) Effect of warming and grazing on litter mass loss and temperature sensitivity of litter and dung mass loss on the Tibetan plateau. Glob Chang Biol 16:1606–1617
Malchair S, De Boeck HJ, Lemmens CMHM, Ceulemans R, Merckx R, Nijs I, Carnol M (2010) Diversity–function relationship of ammonia-oxidizing bacteria in soils among functional groups of grassland species under climate warming. Appl Soil Ecol 44:15–23
McAleece N, Gage JDG, Lambshead PJD, Paterson GLJ (1997) BioDiversity Professional statistics analysis software. Jointly developed by the Scottish Association for Marine Science and the Natural History Museum London. Available at: http://www.sams.ac.uk
Mertens J, Broos K, Wakelin SA, Kowalchuk GA, Springael D, Smolders E (2009) Bacteria, not archaea, restore nitrification in a zinc-contaminated soil. ISME J 3:916–923
Ollivier J, Towe S, Bannert A, Hai B, Kastl EM, Meyer A, Su MX, Kleineidam K, Schloter M (2011) Nitrogen turnover in soil and global change. FEMS Microbiol Ecol 78:3–16
Paranychianakis NV, Tsiknia M, Giannakis G, Nikolaidis NP, Kalogerakis N (2013) Nitrogen cycling and relationships between ammonia oxidizers and denitrifiers in a clay-loam soil. Appl Microbiol Biotechnol 97:5507–5515
Rui YC, Wang SP, Xu ZH, Wang YF, Chen CR, Zhou XQ, Kang XM, Lu SB, Hu YG, Lin QY, Luo CY (2011) Warming and grazing affect soil labile carbon and nitrogen pools differently in an alpine meadow of the Qinghai-Tibet Plateau in China. J Soils Sediments 11:903–914
Rustad LE, Campbell JL, Marion GM, Norby RJ, Mitchell MJ, Hartley AE, Cornelissen JHC, Gurevitch J, GCTE-NEWS (2001) A meta-analysis of the response of soil respiration, net nitrogen mineralization, and aboveground plant growth to experimental ecosystem warming. Oecologia 126:543–562
Schloss PD, Handelsman J (2005) Introducing DOTUR, a computer program for defining operational taxonomic units and estimating species richness. Appl Environ Microbiol 71:1501–1506
Shen JP, Zhang LM, Zhu YG, Zhang JB, He JZ (2008) Abundance and composition of ammonia-oxidizing bacteria and ammonia-oxidizing archaea communities of an alkaline sandy loam. Environ Microbiol 10:1601–1611
Sims A, Horton J, Gajaraj S, McIntosh S, Miles RJ, Mueller R, Reed R, Hu ZQ (2012) Temporal and spatial distributions of ammonia-oxidizing archaea and bacteria and their ratio as an indicator of oligotrophic conditions in natural wetlands. Water Res 46:4121–4129
Singh BK, Bardgett RD, Smith P, Reay DS (2010) Microorganisms and climate change: terrestrial feedbacks and mitigation options. Nat Rev Microbiol 8:779–790
Singh D, Takahashi K, Adams JM (2012) Elevational patterns in archaeal diversity on Mt. Fuji. PLoS One 7:e44494
Singleton DR, Furlong MA, Rathbun SL, Whitman WB (2001) Quantitative comparisons of 16S rRNA gene sequence libraries from environmental samples. Appl Environ Microbiol 67:4374–4376
Szukics U, Abell GCJ, Hödl V, Mitter B, Sessitsch A, Hackl E, Zechmeister-Boltenstern S (2010) Nitrifiers and denitrifiers respond rapidly to changed moisture and increasing temperature in a pristine forest soil. FEMS Microbiol Ecol 72:395–406
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Tian XF, Hu HW, Ding Q, Song MH, Xu XL, Zheng Y, Guo LD (2013) Influence of nitrogen fertilization on soil ammonia-oxidizers and denitrifiers abundance, microbial biomass and enzyme activities in an alpine meadow. Biol Fertil Soils. doi:10.1007/s00374-013-0889-0
Walker JKM, Egger KN, Henry GHR (2008) Long-term experimental warming alters nitrogen-cycling communities but site factors remain the primary drivers of community structure in high arctic tundra soils. ISME J 2:982–995
Wan SQ, Xia JY, Liu WX, Niu SL (2009) Photosynthetic overcompensation under nocturnal warming enhances grassland carbon sequestration. Ecology 90:2700–2710
Weedon JT, Kowalchuk GA, Aerts R, van Hal J, van Logtestijn R, Tas N, Röling WFM, van Bodegom PM (2012) Summer warming accelerates sub-arctic peatland nitrogen cycling without changing enzyme pools or microbial community structure. Glob Chang Biol 18:138–150
Wertz S, Goyer C, Zebarth BJ, Burton DL, Tatti E, Chantigny MH, Filion M (2013) Effects of temperatures near the freezing point on N2O emissions, denitrification and on the abundance and structure of nitrifying and denitrifying soil communities. FEMS Microbiol Ecol 83:242–254
Wu YC, Lu L, Wang BZ, Lin XG, Zhu JG, Cai ZC, Yan XY, Jia ZJ (2011) Long-term field fertilization significantly alters community structure of ammonia-oxidizing bacteria rather than archaea in a paddy soil. Soil Sci Soc Am J 75:1431–1439
Xu ZH, Chen CR, He JZ, Liu JX (2009) Trends and challenges in soil research 2009: linking global climate change to local long-term forest productivity. J Soils Sediments 9:83–88
Xu GP, Hu YG, Wang SP, Zhang ZH, Chang XF, Duan JC, Luo CY, Chao ZG, Su AL, Lin QY, Li YN, Du MY (2010) Effects of litter quality and climate change along an elevation gradient on litter mass loss in an alpine meadow ecosystem on the Tibetan plateau. Plant Ecol 209:257–268
Yang W, Zheng Y, Gao C, He XH, Ding Q, Kim YC, Rui YC, Wang SP, Guo LD (2013) The arbuscular mycorrhizal fungal community response to warming and grazing differs between soil and roots on the Qinghai-Tibetan Plateau. PLoS One 8:e76447
Yao HY, Gao YM, Nicol GW, Campbell CD, Prosser JI, Zhang LM, Han WY, Singh BK (2011) Links between ammonia oxidizer community structure, abundance, and nitrification potential in acidic soils. Appl Environ Microbiol 77:4618–4625
Yvon-Durocher G, Jones JI, Trimmer M, Woodward G, Montoya JM (2010) Warming alters the metabolic balance of ecosystems. Philos Trans R Soc B Biol Sci 365:2117–2126
Zhang LM, Wang M, Prosser JI, Zheng YM, He JZ (2009) Altitude ammonia-oxidizing bacteria and archaea in soils of Mount Everest. FEMS Microbiol Ecol 70:208–217
Zheng Y, Yang W, Sun X, Wang SP, Rui YC, Luo CY, Guo LD (2012) Methanotrophic community structure and activity under warming and grazing of alpine meadow on the Tibetan Plateau. Appl Microbiol Biotechnol 93:2193–2203
Zhou ZF, Zheng YM, Shen JP, Zhang LM, He JZ (2011) Response of denitrification genes nirS, nirK, and nosZ to irrigation water quality in a Chinese agricultural soil. Environ Sci Pollut Res 18:1644–1652
Zhou ZF, Shi XJ, Zheng Y, Qin ZX, Xie DT, Li ZL, Guo T (2014) Abundance and community structure of ammonia-oxidizing bacteria and archaea in purple soil under long-term fertilization. Eur J Soil Biol 60:24–33
Acknowledgments
We thank Dr. Qiong Ding, College of Horticulture and Landscape Architecture, Hainan University for the helpful comments on data analyses. We also appreciate two anonymous referees for their invaluable suggestions and constructive comments. This study was supported by the grants of the National Natural Science Foundation of China (nos. 41001149, 31070434, 41230750) and Strategic Priority Research Program (B) of the Chinese Academy of Sciences (no. XDB03030403). Y.Z. received a fellowship as a visiting scholar from the China Scholarship Council (no. 2011491255).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Yanfen Wang
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure S1
Rank of the abundance (sequence numbers) of AOA (A) and AOB (B) OTUs at 97 % level of similarity. The diagram trend is analogous for both AOA and AOB: the top 5 abundant OTUs cover more than 56 % and 70 % of all sequences of AOA and AOB, respectively; and a high number of singletons represent the ‘long tail’ of the diagram. (DOC 41 kb)
Figure S2
Rarefaction curves of the amoA gene sequences of AOA (A) and AOB (B) obtained from four treatments using software aRarefactWin version 1.3. (DOC 86 kb)
Rights and permissions
About this article
Cite this article
Zheng, Y., Yang, W., Hu, HW. et al. Ammonia oxidizers and denitrifiers in response to reciprocal elevation translocation in an alpine meadow on the Tibetan Plateau. J Soils Sediments 14, 1189–1199 (2014). https://doi.org/10.1007/s11368-014-0867-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-014-0867-7