Abstract
Purpose
Nitrous oxide (N2O) is a trace gas and is mainly produced through the microbial-mediated nitrification and denitrification processes in agricultural soils. Dicyandiamide (DCD) is a powerful nitrification inhibitor (NI) which can mitigate N2O production from soil. However, it remains poorly understood how N2O emission and associated microbes are affected by DCD application across different agricultural soils.
Materials and methods
Here, we carried out a 21-day laboratory microcosm incubation experiment to examine the impact of ammonium sulfate and DCD applications on N2O production and the abundance of nitrifiers and denitrifiers in two contrasting agricultural soils. The abundance of the functional genes was quantified by quantitative PCR to investigate the population changes in response to ammonium sulfate and DCD addition.
Results and discussion
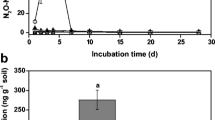
Higher N2O production emitted followed the application of ammonium sulfate (287.9 ng N2O–N g−1 soil) in the alluvial soil was obtained compared to that (84.9 ng N2O–N g−1 soil) in the red soil. The average N2O emission rate in the NH4 + treatment from the alluvial soil was significantly higher than that from the red soil, and DCD addition remarkably reduced N2O emission rates in both soils during the 21-day incubation. We also observed that the abundance of ammonia-oxidizing bacteria (AOB) amoA gene and the nirK gene was positively correlated with total N2O emissions in the alluvial soil, but only AOB amoA gene abundance was significantly related to the total N2O emissions in the red soil.
Conclusions
These results suggested that DCD was more effective in inhibiting N2O production in the alluvial soil than in the red soil. DCD could effectively reduce N2O emission by suppressing the growth of microorganisms containing AOB amoA gene and the nirK gene in the alluvial soil.





Similar content being viewed by others
References
Arp DJ, Stein LY (2003) Metabolism of inorganic N compounds by ammonia-oxidizing bacteria. Crit Rev Biochem Mol 38:471–495
Bakken LR, Bergaust L, Liu BB, Frostegard A (2012) Regulation of denitrification at the cellular level: a clue to the understanding of N2O emissions from soils. Philos T R Soc B 367:1226–1234
Čuhel J, Miloslav Š, Ronnie JL, David B (2010) Insights into the effect of soil pH on N2O and N2 emissions and denitrifier community size and activity. Appl Environ Microbiol 76:1870–1878
Cui PY, Fan FL, Yin C, Li ZJ, Song AL, Wan YF, Liang YC (2013) Urea- and nitrapyrin-affected N2O emission is coupled mainly with ammonia oxidizing bacteria growth in microcosms of three typical Chinese arable soils. Soil Biol Biochem 66:214–221
Dai Y, Di HJ, Cameron KC, He JZ (2013) Effects of nitrogen application rate and a nitrification inhibitor dicyandiamide on ammonia oxidizers and N2O emissions in a grazed pasture soil. Sci Total Environ 465:125–135
Davidson EA (2009) The contribution of manure and fertilizer nitrogen to atmospheric nitrous oxide since 1860. Nat Geosci 2:659–662
Davidson EA, Kanter D (2014) Inventories and scenarios of nitrous oxide emissions. Environ Res Lett 9:105012
Del Grosso SJ, Parton WJ, Mosier AR, Walsh MK, Ojima DS, Thornton PE (2006) DAYCENT national-scale simulations of nitrous oxide emissions from cropped soils in the United States. J Environ Qual 35:1451–1460
Di HJ, Cameron KC (2008) Sources of nitrous oxide from 15N-labelled animal urine and urea fertiliser with and without a nitrification inhibitor, dicyandimide (DCD). Aust J Soil Res 46:76–82
Di HJ, Cameron KC, Sherlock RR, Shen JP, He JZ, Winefield CS (2010) Nitrous oxide emissions from grazed grassland as affected by a nitrification inhibitor, dicyandiamide, and relationships with ammonia-oxidizing bacteria and archaea. J Soils Sediments 10:943–954
Di HJ, Cameron KC, Podolyan A, Robinson A (2014) Effect of soil moisture status and a nitrification inhibitor, dicyandiamide, on ammonia oxidizer and denitrifier growth and nitrous oxide emissions in a grassland soil. Soil Biol Biochem 73:59–68
Firestone MK, Davidson EA (1989) Microbiological basis of NO and N2O production and consumption in soils. In: Andreae MO, Schimel DS (eds) Exchange of trace gases between terrestrial ecosystems and the atmosphere. John Wiley & Sons, Chichester, pp 7–21
Galloway JN, Townsend AR, Erisman JW, Bekunda M, Cai ZC, Freney JR, Martinelli LA, Seitzinger SP, Sutton MA (2008) Transformation of the nitrogen cycle: recent trends, questions, and potential solutions. Science 320:889–892
Hallin S, Lindgren PE (1999) PCR detection of genes encoding nitrite reductase in denitrifying bacteria. Appl Environ Microbiol 65:1652–1657
Henry S, Bru D, Stres B, Hallet S, Philippot L (2006) Quantitative detection of the nosZ gene, encoding nitrous oxide reductase, and comparison of the abundances of 16S rRNA, narG, nirK, and nosZ genes in soils. Appl Environ Microbiol 72:5181–5189
Hu HW, Chen DL, He JZ (2015) Microbial regulation of terrestrial nitrous oxide formation: understanding the biological pathways for prediction of emission rates. FEMS Microbiol Rev 39:729–749
Huang T, Gao B, Hu XK, Lu X, Well R, Christie P, Bakken LR, Ju XT (2014a) Ammonia-oxidation as an engine to generate nitrous oxide in an intensively managed calcareous fluvo-aquic soil. Sci Rep 4:3950
Huang Y, Li YY, Yao HY (2014b) Nitrate enhances N2O emission more than ammonium in a highly acidic soil. J Soils Sediments 14:146–154
IPCC (2007) Mitigation of climate change. In: Merz B et al (eds) Contribution of working group I to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge, UK
Jia Z, Conrad R (2009) Bacteria rather than archaea dominate microbial ammonia oxidation in an agricultural soil. Environ Microbiol 11:1658–1671
Kim SW, Miyahara M, Fushinobu S, Wakagi T, Shoun H (2010) Nitrous oxide emission from nitrifying activated sludge dependent on denitrification by ammonia-oxidizing bacteria. Bioresource Technol 101:3958–3963
Kloos K, Mergel A, Rosch C, Bothe H (2001) Denitrification within the genus Azospirillum and other associative bacteria. Aust J Plant Physiol 28:991–998
Lan T, Han Y, Roelcke M, Nieder R, Cai Z (2013) Effects of the nitrification inhibitor dicyandiamide (DCD) on gross N transformation rates and mitigating N2O emission in paddy soils. Soil Biol Biochem 67:174–182
Leviĕnik-Höfferle S, Nicol GW, Ausec L, Mandic-Mulec I, Prosser JI (2012) Stimulation of thaumarchaeal ammonia oxidation by ammonia derived from organic nitrogen but not added inorganic nitrogen. FEMS Microbiol Ecol 80:114–123
Merino P, Estavillo JM, Besga G, Pinto M, Gonzalez-Murua C (2001) Nitrification and denitrification derived N2O production from a grassland soil under application of DCD and Actilith F2. Nutr Cycl Agroecosys 60:9–14
O’Callaghan M, Gerard EM, Carter PE, Lardner R, Sarathchandra U, Burch G, Ghani A, Bell N (2010) Effect of the nitrification inhibitor dicyandiamide (DCD) on microbial communities in a pasture soil amended with bovine urine. Soil Biol Biochem 42:1425–1436
Rotthauwe JH, Witzel KP, Liesack W (1997) The ammonia monooxygenase structural gene amoA as a functional marker: molecular fine-scale analysis of natural ammonia-oxidizing populations. Appl Environ Microbiol 63:4704–4712
Ruser R, Schulz R (2015) The effect of nitrification inhibitors on the nitrous oxide (N2O) release from agricultural soils—a review. J plant Nutri. Soil Sci 178:171–188
Shaw LJ, Nicol GW, Smith Z, Fear J, Prosser JI, Baggs EM (2006) Nitrosospira spp. can produce nitrous oxide via a nitrifier denitrification pathway. Environ Microbiol 8:214–222
Stieglmeier M, Mooshammer M, Kitzler B, Wanek W, Zechmeister-Boltenstern S, Richter A, Schleper C (2014) Aerobic nitrous oxide production through N-nitrosating hybrid formation in ammonia-oxidizing archaea. ISME J 8:1135–1146
Subbarao GV, Ito O, Sahrawat KL, Berry WL, Nakahara K, Ishikawa T, Watanabe T, Suenaga K, Rondon M, Rao IM (2006) Scope and strategies for regulation of nitrification in agricultural systems-challenges and opportunities. Crit Rev Plant Sci 25:303–335
Tourna M, Freitag TE, Nicol GW, Prosser JI (2008) Growth, activity and temperature responses of ammonia-oxidizing archaea and bacteria in soil microcosms. Environ Microbiol 10:1357–1364
USDA (1994) Keys to soil taxonomy, 6th edn. United States Department of Agriculture, Soil Conservation Service, USA, p. 306
Verhamme DT, Prosser JI, Nicol GW (2011) Ammonia concentration determines differential growth of ammonia-oxidising archaea and bacteria in soil microcosms. ISME J 5:1067–1071
Wang Q, Zhang LM, Shen JP, Du S, Han LL, He JZ (2016) Nitrogen fertiliser-induced changes in N2O emissions are attributed more to ammonia-oxidising bacteria rather than archaea as revealed using 1-octyne and acetylene inhibitors in two arable soils. Biol Fertil Soils 52:1163–1171
Wrage N, Velthof GL, van Beusichem ML, Oenema O (2001) Role of nitrifier denitrification in the production of nitrous oxide. Soil Biol Biochem 33:1723–1732
Zhang LM, HW H, Shen JP, He JZ (2012) Ammonia-oxidizing archaea have more important role than ammonia-oxidizing bacteria in ammonia oxidation of strongly acidic soils. ISME J 6:1032–1045
Acknowledgements
This work was supported by the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB15020200) and by the Natural Science Foundation of China (41322007, 41371265, and 41301265).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Weijin Wang
Rights and permissions
About this article
Cite this article
Wang, Q., Hu, HW., Shen, JP. et al. Effects of the nitrification inhibitor dicyandiamide (DCD) on N2Oemissions and the abundance of nitrifiers and denitrifiers in two contrasting agricultural soils. J Soils Sediments 17, 1635–1643 (2017). https://doi.org/10.1007/s11368-016-1633-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-016-1633-9