Abstract
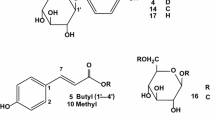
A methanol extract from the rhizomes of Kaempferia parviflora Wall. ex Baker (Zingiberaceae) has shown inhibitory effects against melanogenesis in theophylline-stimulated murine B16 melanoma 4A5 cells (IC50 = 9.6 μg/mL). Among 25 flavonoids and three acetophenones isolated previously (1–28), several constituents including 5-hydroxy-7,3′,4′-trimethoxyflavone (6, IC50 = 8.8 μM), 5,7,3′,4′-tetramethoxyflavone (7, 8.6 μM), 5,3′-dihydroxy-3,7,4′-trimethoxyflavone (12, 2.9 μM), and 5-hydroxy-3,7,3′,4′-tetramethoxyflavone (13, 3.5 μM) showed inhibitory effects without notable cytotoxicity at the effective concentrations. Compounds 6, 7, 12, and 13 inhibited the expression of tyrosinase, tyrosine-related protein (TRP)-1, and TRP-2 mRNA, which could be the mechanism of their melanogenesis inhibitory activity. In addition, a quantitative analytical method for 12 methoxyflavones (1, 2, 4–11, 13, and 14) in the extract was developed using HPLC. The optimal condition for separation and detection of these constituents were achieved on an ODS column (3 μm particle size, 2.1 mm i.d. × 100 mm) with MeOH–0.1 % aqueous acetic acid solvent systems as the mobile phase, and the detection and quantitation limits of the method were estimated to be 0.08–0.66 ng and 0.22–2.00 ng, respectively. The relative standard deviation values of intra- and interday precision were lower than 0.95 and 1.08 %, respectively, overall mean recoveries of all flavonoids were 97.9–102.9 %, and the correlation coefficients of all the calibration curves showed good linearity within the test ranges. For validation of the protocol, extracts of three kinds of the plant’s rhizomes collected from different regions in Thailand (Leoi, Phetchabun, and Chiang Mai provinces) were evaluated. The results indicated that the assay was reproducible, precise, and could be readily utilized for the quality evaluation of the plant materials.


Similar content being viewed by others
References
Grotewold E (ed) (2006) The science of flavonoids. Springer, Berlin
Andersen ØM, Markham KR (eds) (2006) Flavonoids: chemistry, biochemistry and applications. CRC, Boca Raton
Agrawal PK (ed) (1986) Carbon-13 NMR of flavonoids. Elsevier, Amsterdam
Amić D, Davidović-Amić D, Bešlo D, Rastija V, Lučić B, Trinajstić N (2007) SAR and QSAR of the antioxidant activity of flavonoids. Curr Med Chem 14:827–845
Yao LH, Jiang YM, Shi J, Tomás-Barberán FA, Datta N, Singanusong R, Chen SS (2004) Flavonoids in food and their health benefits. Plant Foods Human Nutr 59:113–122
Cook NC, Samman S (1996) Flavonoids—chemistry, metabolism, cardioprotective effects, and dietary sources. J Nutr Biochem 7:66–76
Rice-Evans CA, Miller NJ, Paganga G (1996) Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Rad Biol Med 20:933–956
Evans M, Sharma P, Guthrie N (2012) Bioavailability of citrus polymethoxylated flavones and their biological role in metabolic syndrome and hyperlipidemia. In: Noreddin A (ed) Readings in advanced phamacokinetics—theory, methods and applications. InTech, Rijeka, pp 267–284
Chaipech S, Morikawa T, Ninomiya K, Yoshikawa M, Pongpiriyadacha Y, Hayakawa T, Muraoka O (2012) Structures of two new phenolic glycosides, kaempferiaosides A and B, and hepatoprotective constitunets from the rhizomes of Kaempferia parviflora. Chem Pharm Bull 60:62–69
Chaipech S, Morikawa T, Ninomiya K, Yoshikawa M, Pongpiriyadacha Y, Hayakawa T, Muraoka O (2012) New flav-3-en-3-ol glycosides, kaempferiaosides C and D, and acetophenone glycosides, kaempferiaosides E and F, from the rhizomes of Kaempferia parviflora. J Nat Med 66:486–492
Hu F, Lesney PF (1964) The isolation and cytology of two pigment cell strains from B-16 mouse melanomas. Cancer Res 24:1634–1643
Tanabe G, Sugano Y, Shirato M, Sonoda N, Tsutsui N, Morikawa T, Ninomiya K, Yoshikawa M, Muraoka O (2015) Total synthesis of 4,5-didehydroguadiscine: a potent melanogenesis inhibitor from the Brazilian medicinal herb, Hornschuchia oblique. J Nat Prod 78:1536–1542
Morikawa T, Ninomiya K, Kuramoto H, Kamei I, Yoshikawa M, Muraoka O (2016) Phenylethanoid and phenylpropanoid glycosides with melanogenesis inhibitory activity from the flowers of Narcissus tazetta var. chinensis. J Nat Med 70:89–101
Morikawa T, Nakanishi Y, Ninomiya K, Matsuda H, Nakashima S, Miki H, Miyashita Y, Yoshikawa M, Hayakawa T, Muraoka O (2014) Dimeric pyrrolidinoindoline-type alkaloids with melanogenesis inhibitory activity in flower buds of Chimonanthus praecox. J Nat Med 68:539—549
Prota G (1988) Progress in the chemistry of melanins and related metabolites. Med Res Rev 8:525–556
Kim YJ, Uyama H (2005) Tyrosinase inhibitors from natural and synthetic sources: structure, inhibition mechanism and perspective for the future. Cell Mol Life Sci 62:1707–1723
Hearing VJ, Korner AM, Pawelek JM (1982) New regulators of melanogenesis are associated with purified tyrosinase isozymes. J Invest Dermatol 79:16–18
Hearing VJ, Jiménez M (1987) Mammalian tyrosinase–the critical regulatory control point in melanocyte pigmentation. Int J Biochem 19:1141–1147
Kuzumaki T, Matsuda A, Wakamatsu K, Ito S, Ishikawa K (1993) Eumelanin biosynthesis is regulated by coordinate expression of tyrosinase and tyrosinase-related protein-1 genes. Exp Cell Res 207:33–40
Friedmann PS, Gilchrest BA (1987) Ultraviolet radiation directly induces pigment production by cultured human melanocytes. J Cell Physiol 133:88–94
Hunt G, Todd C, Cresswell JE, Thody AJ (1994) Alpha-melanocyte stimulating hormone and its analogue Nle4DPhe7 alpha-MSH affect morphology, tyrosinase activity and melanogenesis in cultured human melanocytes. J Cell Sci 107:205–211
Steinberg ML, Whittaker JR (1976) Stimulation of melanotic expression in a melanoma cell line by theophylline. J Cell Physiol 87:265–275
Matsuda H, Nakashima S, Oda Y, Nakamura S, Yoshikawa M (2009) Melanogenesis inhibitors from the rhizomes of Alpinia officinarum in B16 melanoma cells. Bioorg Med Chem 17:6048–6053
Parvez S, Kang M, Chung HS, Bae H (2007) Naturally occurring tyrosinase inhibitors: mechanism and applications in skin health, cosmetics and agriculture industries. Phytother Res 21:805–816
Bao K, Dai Y, Zhu Z-B, Tu F-J, Zhang W-G, Yao X-S (2010) Design and synthesis of biphenyl derivatives as mushroom tyrosinase inhibitors. Bioorg Med Chem 18:6708–6714
Parvez S, Kang M, Chung H-S, Cho C, Hong M-C, Shin M-K, Bae H (2006) Survey and mechanism of skin depigmentating and lightening agents. Phytother Res 20:921–934
Chang T-S (2009) An updated review of tyrosinase inhibitors. Int J Mol Sci 10:2440–2475
Bertolotto C, Buscá R, Abbe P, Bille K, Aberdam E, Ortonne J-P, Ballotti R (1998) Different cis-acting elements are involved in the regulation of TRP1 and TRP2 promoter activities by cyclic AMP: pivotal role of M boxes (GTCATGTGCT) and of microphthalmia. Mol Cell Biol 18:694–702
Azuma T, Kayano S, Matsumura Y, Konishi Y, Tanaka Y, Kikuzaki H (2011) Antimutagenic and α-glucosidase inhibitory effects of constituents from Kaempferia parviflora. Food Chem 125:471–475
Acknowledgments
This work was supported by MEXT-Supported Program for the Strategic Research Foundation at Private Universities, 2014–2018. Thanks are also due to Kobayashi International Scholarship Foundation for the financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ninomiya, K., Matsumoto, T., Chaipech, S. et al. Simultaneous quantitative analysis of 12 methoxyflavones with melanogenesis inhibitory activity from the rhizomes of Kaempferia parviflora . J Nat Med 70, 179–189 (2016). https://doi.org/10.1007/s11418-015-0955-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-015-0955-z