Abstract
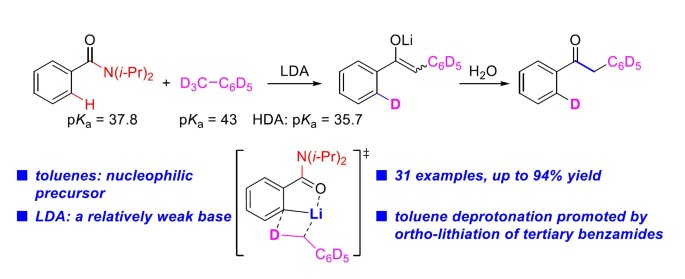
The deprotonative functionalization of toluenes, for their weak acidity, generally needs strong bases, thus leading to the requirement of harsh conditions and the generation of by-products. Direct nucleophilic acyl substitution reaction of amides with organometallic reagents could provide an ideal solution for ketone synthesis. However, the inert amides and highly reactive organometallic reagents bring great challenges for an efficient and selective synthetic approach. Herein, we reported an lithium diisopropylamide (LDA)-promoted benzylic aroylation of toluenes with unactivated tertiary benzamides, providing a direct and efficient synthesis of various aryl benzyl ketones. This process features a kinetic deprotonative functionalization of toluenes with a readily available base LDA. Mechanism studies revealed that the directed ortho-lithiation of the tertiary benzamide with LDA promoted the benzylic kinetic deprotonation of toluene and triggered the nucleophilic acyl substitution reaction with the amide.
Similar content being viewed by others
References
Vanjari R, Singh KN. Chem Soc Rev, 2015, 44: 8062–8096
Yi H, Zhang G, Wang H, Huang Z, Wang J, Singh AK, Lei A. Chem Rev, 2017, 117: 9016–9085
Bordwell FG, Algrim D, Vanier NR. J Org Chem, 1977, 42: 1817–1819
Eberhardt GG, Peterson HJ. J Org Chem, 1965, 30: 82–84
Broaddus CD. J Am Chem Soc, 1966, 88: 4174–4178
Broaddus CD. J Org Chem, 1970, 35: 10–15
Pines H, Stalick WM, Holford TG, Golab J, Lazar H, Simonik J. J Org Chem, 1971, 36: 2299–2303
Hage M, Ogle CA, Rathman TL, Hubbard JL. Main Group Metal Chem, 1998, 21: 777–781
Steele BR, Screttas CG. J Am Chem Soc, 2000, 122: 2391–2392
Dettenrieder N, Aramaki Y, Wolf BM, Maichle-Mössmer C, Zhao X, Yamashita M, Nozaki K, Anwander R. Angew Chem Int Ed, 2014, 53: 6259–6262
Blangetti M, Fleming P, O’Shea DF. J Org Chem, 2012, 77: 2870–2877
Unkelbach C, O’Shea DF, Strohmann C. Angew Chem Int Ed, 2014, 53: 553–556
Manvar A, Fleming P, O’Shea DF. J Org Chem, 2015, 80: 8727–8738
Yamashita Y, Suzuki H, Sato I, Hirata T, Kobayashi S. Angew Chem Int Ed, 2018, 57: 6896–6900
Sato I, Yamashita Y, Kobayashi S. Synthesis, 2019, 51: 240–250
Wang Z, Zheng Z, Xu X, Mao J, Walsh PJ. Nat Commun, 2018, 9: 3365
Mao J, Wang Z, Xu X, Liu G, Jiang R, Guan H, Zheng Z, Walsh PJ. Angew Chem Int Ed, 2019, 58: 11033–11038
Liu G, Walsh PJ, Mao J. Org Lett, 2019, 21: 8514–8518
Takemoto S, Shibata E, Nakajima M, Yumoto Y, Shimamoto M, Matsuzaka H. J Am Chem Soc, 2016, 138: 14836–14839
McGrew GI, Temaismithi J, Carroll PJ, Walsh PJ. Angew Chem Int Ed, 2010, 49: 5541–5544
Zhang J, Stanciu C, Wang B, Hussain MM, Da CS, Carroll PJ, Dreher SD, Walsh PJ. J Am Chem Soc, 2011, 133: 20552–20560
Mao J, Zhang J, Jiang H, Bellomo A, Zhang M, Gao Z, Dreher SD, Walsh PJ. Angew Chem Int Ed, 2016, 55: 2526–2530
Sha SC, Tcyrulnikov S, Li M, Hu B, Fu Y, Kozlowski MC, Walsh PJ. J Am Chem Soc, 2018, 140: 12415–12423
Jiang H, Sha SC, Jeong SA, Manor BC, Walsh PJ. Org Lett, 2019, 21: 1735–1739
Nahm S, Weinreb SM. Tetrahedron Lett, 1981, 22: 3815–3818
Balasubramaniam S, Aidhen I. Synthesis, 2008, 2008: 3707–3738
Evans DA, Borg G, Scheidt KA. Angew Chem Int Ed, 2002, 41:3188–3191
Heller ST, Newton JN, Fu T, Sarpong R. Angew Chem Int Ed, 2015, 54: 9839–9843
Sureshbabu P, Azeez S, Muniyappan N, Sabiah S, Kandasamy J. J Org Chem, 2019, 84: 11823–11838
Szostak M, Aubé J. Org Biomol Chem, 2011, 9: 27–35
Szostak M, Aubé J. Chem Rev, 2013, 113: 5701–5765
Adachi S, Kumagai N, Shibasaki M. Tetrahedron Lett, 2018, 59: 1147–1158
Liu C, Achtenhagen M, Szostak M. Org Lett, 2016, 18: 2375–2378
Pace V, Holzer W, Olofsson B. Adv Synth Catal, 2014, 356: 3697–3736
Bechara WS, Pelletier G, Charette AB. Nat Chem, 2012, 4: 228–234
Xiao KJ, Wang AE, Huang YH, Huang PQ. Asian J Org Chem, 2012, 1: 130–132
Huang PQ, Wang Y, Xiao KJ, Huang YH. Tetrahedron, 2015, 71: 4248–4254
Chen C, Liu P, Luo M, Zeng X. ACS Catal, 2018, 8: 5864–5868
Huang PQ, Huang YH, Geng H, Ye JL. Sci Rep, 2016, 6: 28801
Huang PQ, Huang YH, Xiao KJ. J Org Chem, 2016, 81: 9020–9027
Wang S, Huang P. Chin J Chem, 2019, 37: 887–891
Geng H, Huang P. Chin J Chem, 2019, 37: 811–816
Wu D, He Q, Chen D, Ye J, Huang P. Chin J Chem, 2019, 37: 315–322
Ou W, Huang PQ. Sci China Chem, 2020, 63: 11–15
Ouyang K, Hao W, Zhang WX, Xi Z. Chem Rev, 2015, 115: 12045–12090
Takise R, Muto K, Yamaguchi J. Chem Soc Rev, 2017, 46: 5864–5888
Chaudhari MB, Gnanaprakasam B. Chem Asian J, 2019, 14: 76–93
Simmons BJ, Weires NA, Dander JE, Garg NK. ACS Catal, 2016, 6: 3176–3179
Liu X, Hsiao CC, Guo L, Rueping M. Org Lett, 2018, 20: 2976–2979
Zhai DD, Zhang XY, Liu YF, Zheng L, Guan BT. Angew Chem Int Ed, 2018, 57: 1650–1653
Liu YF, Zhai DD, Zhang XY, Guan BT. Angew Chem Int Ed, 2018, 57: 8245–8249
Liu YF, Zheng L, Zhai DD, Zhang XY, Guan BT. Org Lett, 2019, 21: 5351–5356
Guan BT, Shi ZJ. Sci Sin Chim, 2021, 51: 201–212
Fraser RR, Bresse M, Mansour TS. J Am Chem Soc, 1983, 105: 7790–7791
Clayden J. Organolithiums: Selectivity for Synthesis. Oxford: Elsevier, 2002. 9–109
Petragnani N, Yonashiro M. Synthesis, 1982, 1982: 521–578
Beak P, Snieckus V. Acc Chem Res, 1982, 15: 306–312
Clayden J, Davies RP, Hendy MA, Snaith, R, Wheatley AEH. Angew Chem Int Ed, 2001, 40: 1238–1240
Kondo Y, Morey JV, Morgan JC, Naka H, Nobuto D, Raithby PR, Uchiyama M, Wheatley AEH. J Am Chem Soc, 2007, 129: 12734–12738
Campbell Smith A, Donnard M, Haywood J, McPartlin M, Vincent MA, Hillier IH, Clayden J, Wheatley AEH. Chem Eur J, 2011, 17: 8078–8084
Armstrong DR, Garden JA, Kennedy AR, Leenhouts SM, Mulvey RE, O’Keefe P, O’Hara CT, Steven A. Chem Eur J, 2013, 19: 13492–13503
Garden JA, Kennedy AR, Mulvey RE, Robertson SD. Chem Commun, 2012, 48: 5265–5267
Yang F, Zou D, Chen S, Wang H, Zhao Y, Zhao L, Li L, Li J, Walsh PJ. Adv Synth Catal, 2020, 362: 3423–3430
Acknowledgements
We gratefully acknowledge the support from the Fudan University. We thank Prof. Bi-Jie Li (Tsinghua Univ.) and Prof. Qian Peng (Nankai Univ.) for their helpful discussions. This work was supported by the Natural Science Foundation of Tianjin (19JCYBJC20100).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Bao, CC., Luo, YL., Du, HZ. et al. Benzylic aroylation of toluenes with unactivated tertiary benzamides promoted by directed ortho-lithiation. Sci. China Chem. 64, 1349–1354 (2021). https://doi.org/10.1007/s11426-021-1035-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-021-1035-5