Abstract
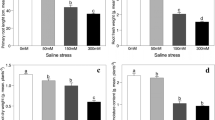
Alhagi sparsifolia is a leguminous perennial desert species that is plays an important role in dune stabilization and revegetation of degraded desert ecosystems. We investigated the effects of three different levels of salinity (50, 150, 250 mmol/L NaCl) on the growth, shoot photosynthetic parameters and salt distribution amongst different plant organs in one-year-old A. sparsifolia seedlings in a pot experiment over a 50 d period. The minimum (predawn) and maximum (midday) water potentials of A. sparsifolia seedlings decreased with the increase of external NaCl concentrations as a consequence of the osmotic or water deficit effect of saline solutions outside the roots. Salinity also reduced gas exchange parameters in A. sparsifolia, with seedlings subjected to salinity having lower photosynthesis rates and reduced stomatal conductances compared to the control. The reductions in photosynthetic rates in high salinity treatments of the A. sparsifolia seedlings were mainly caused by stomatal limitation. Consequently plants growing at greater external NaCl concentrations had significantly lower biomass accumulation compared to the control grown at 50 mmol/L. However, plants exposed to higher salinity were able to maintain growth throughout the experiment but allocated a greater proportion of biomass belowground. Plants exposed to higher external salinity levels had increased concentrations of Na+ and Cl− ions in shoots and roots, suggesting that A. sparsifolia seedlings were utilizing Na+ and Cl− as osmolytes to increase the cellular osmolality and decrease their water potential. We observed the greatest NaCl concentrations in the plants treated with 150 mmol/L NaCl indicating that there may be a threshold level of NaCl that can be tolerated by the plants. In conclusion our results indicate that A. sparsifolia seedlings are moderately salt tolerant. Photosynthetic gas exchange parameters were reduced by greater external salinity but the seedlings maintained substantial photosynthetic rates even under high salinity stress, were able to maintain growth over the 50 d experimental period and showed no signs of salinity toxicity or damage.
Similar content being viewed by others
References
Parida A K, Das A B. Salt tolerance and salinity effects on plants: a review. Ecotox Environ Safe, 2005, 60: 324–349
Munns R. Physiological processes limiting plant growth in saline soils: some dogmas and hypotheses. Plant Cell Environ, 1993, 16: 15–24
Mehari A, Ericsson T, Weih M. Effects of NaCl on seedling growth, biomass production and water status of Acacia nilotica and A. Tortilis. J Arid Environ, 2005, 62: 343–349
Guo S K, Zhao K F. The possible mechanisms of NaCl inhibit photosythesis of maize seedlings (in Chinese). Acta Phytophys Sin, 2001, 27(6): 461–466
Munns R. Comparative physiology of salt and water stress. Plant Cell Environ, 2002, 25: 239–250
Brown C E, Pezeshki S R, Delaune R D. The effects of salinity and soil drying on nutrient uptake and growth of Spartina alterniflora in a simulated tidal system. Environ Exp Bot, 2005, 1–9
Wang B S, Zhao K F. Effect of NaCl stress on the H+-extrusion and redox system of the plasma membrane of corn roots (in Chinese). Acta Bot Sin, 1997, 39(4): 341–346
Haseqawa P M, Bressan P A, Zhu J K, et al. Plant cellular and molecular response to high salinity. Anne Rex Plant Physiol Mol Biol, 2000, 51: 463–499
Yang Y H, Sun Q Y, Shen H. Salt tolerance and injury of plants (in Chinese). Biol Teaching, 2002, 27(11): 1–2
Tao J, Li T, Sun C B, et al. Review on salt stress in plants (in Chinese). Jilin Forest Sci Tech, 2003, 32(5): 1–6
Matthews M A, Boyer J S. Acclimation of photosynthesis to low leaf water potentials. Plant Physiol, 1984, 74: 161–166
Bethke P C, Drew M C. Stomatal and nonstomatal components to inhibition of photosynthesis in leaves of Capsicum annuum during progressive exposure to NaCl salinity. Plant Physiol, 1992, (99): 219–226
Heidari-Sharifabada H, Mirzaie-Nodoushan H. Salinity-induced growth and some metabolic changes in three Salsola species. J Arid Environ, 2006, (67): 715–720
Shin W, Katsumi K, Yu J, et al. Effects of saline and osmotic stress on proline and sugar accumulation in Populus euphratica in vitro. Plant Cell Tissue Org Cul, 2000, (63): 199–206
Liu J P, Zhu J K. Proline accumulation and salt-stress-induced gene expression in a salt-hypersensitive mutant of Arabidopsis. Plant Physiol, 1997, 114: 591–596
Zeng F J, Zhang X M, Li X M. Study on the characteristics of Alhagi sparsifolia and its impact on resoverce protection and development (in Chinese). Arid Land Geogr, 2002, 25: 286–288
Jin Q H. Population character of Alhagi sparsifolia and plant commol/Lunity succession (in Chinese). Acta Phytoecol Sin, 1995, 19(3): 255–260
Zhang L Y, Mai M T. The effects of summol/Ler irrigation on morphological morphological, ecological structure of commol/Lunity and natural reborm of Alhagi sparsifolia (in Chinese). Arid Zone Res, 1995, 12(4): 34–40
Zakeri A, Banihashemi Z. The role of weeds in cultivated and virgin soils on activity and perpetuation of Fusarium oxysporum f. sp. melonis in Fars Province. Iranian J Plant Pathol, 1996, 32(1): 28–39
Li X Y, Zhang X M, Zeng F J, et al. Water relations on Alhagi sparsifolia in the southern fringe of Taklimakan Desert. Acta Bot Sin, 2002, 44(10): 1219–1224
Zhu Y H, Wu Y Q. Water consumption of natural plant Alhagi sparsifolia in arid desert region. Bull Soil Water Conserv, 2003, 23(4): 43–65
Zhang X M, Michael R. The Durative Management of Basic Ecology in the Southern Margin of the Taklimakan Desert (in Chinese). Beijing: Science Press, 2006. 53–130
Berry J A, Downton W J S. Environmental regulation of photosynthesis. In: Govind J, ed. Photosynthesis (Vol.III). New York: Academic Press, 1982. 263–345
Wilson C, Liu X, Scott M, et al. Growth response of major USA cowpea cultivars II. Effect of salinity on leaf gas exchange. Clyde Plant Sci, 2006, 170: 1095–1101
Wang S P, Li J, Guo S R, et al. Effects of NaCl stress on growth and photosynthetic characteristics of cucumber (Cucumber sativus L.) seedlings NaCl (in Chinese). Acta Bot Boreal-Occident Sin, 2006, 26(3): 455–461
Yu W W, Cao B H, Wu L Y. Growths and mineral nutrient balance of black locust clones under salt stress (in Chinese). Acta Bot Boreal.-Occident Sin, 2005, 25(10): 2097–2102
Zeng F J, Zhang X M, Li X Y, et al. Seasonal variation of Tamarix ramosissima and Populus euphratica water potentials in southern fringe of Taklimakan Desert (in Chinese). Chin J Appl Ecol, 2005, 16(8): 1389–1393
Romero-Aranda R, Soria T, Cuartero J. Tomato plant-water uptake and plant-water relationships under saline growth conditions. Plant Sci, 2001, 160: 265–272
Matthews M A, Boyer J S. Acclimation of photosynthesis to low leaf water potentials. Plant Physiol, 1984, 74: 161–166
Hui H X, Xu X, Li S M. Possible mechanism of inhibition on photosynthesis of Lyeium barbarum under salt stress (in Chinese). Chin J Ecol, 2004, 23(1): 5–9
Zheng G Q, Xu X, Xu Z Z, et al. The effect of salt stress on the stomatal and non-stomatal limitation of photosynthesis of Lycium barbarum (in Chinese). Acta Bot Borea1-Occident Sin, 2002, 22(6): 1355–1359
Jiang Q Z, Roche D, Monaco T A, et al. Gas exchange, chlorophyll fluorescence parameters and carbon isotope discrimination of 14 barley genetic lines in response to salinity. Field Crops Res, 2006, 96: 269–278
Gao G L, Jiang W B, Yu K J, et al. A review of studies on effect of salt stress on photosynthesis in fruit crops (in Chinese). J Fruit Sci, 2003, 20(6): 493–497
Tan Y, Zhang H C, Fang S Z, et al. Distribution of K+, Na+, Ca2+ and Mg2+ in poplar rhizosphere system under NaCl stress (in Chinese). J Plant Resour Environ, 2003, 2(3): 11–15
Greenway H, Munns R, Wolfe J, et al. Interaction between growth, Cl− and Na+ uptake, and water relations of plants in saline environments. Plant Cell Environ, 1983, 6: 567–574
Arndt S K, Arampatsis C, Foetzki A, et al. Contrasting patterns of leaf solute accumulation and salt adaptation in four phreatophytic desert plants in a hyperarid desert with saline groundwater. J Arid Environ, 2004, 59: 259–270
Jin Q H. A study on the characters of content and distribution of K, Na, Ca, Mg elements in Alhagi sparsifolia (in Chinese). Acta Phytoecol Sin, 1996, 20(1): 80–84
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by National Natural Science Foundation of China (Grant No. 30670386), Station Foundation of Chinese Academy of Sciences, Science and Technology Key Project of Xinjiang (Grant No. 200633130) and Technology Key Project of Xinjiang (Grant No. 200733144-2)
About this article
Cite this article
Zeng, J., Zeng, F., Arndt, S.K. et al. Growth, physiological characteristics and ion distribution of NaCl stressed Alhagi sparsifolia seedlings. Chin. Sci. Bull. 53 (Suppl 2), 169–176 (2008). https://doi.org/10.1007/s11434-008-6020-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-008-6020-5