Summary
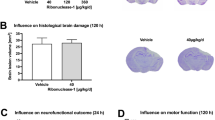
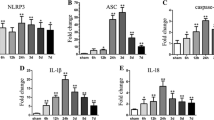
The aim of this study was to investigate the possible beneficial role of telmisartan in cerebral edema after traumatic brain injury (TBI) and the potential mechanisms related to the nucleotide-binding oligomerization domain (NOD)-like receptor (NLR) pyrin domain-containing 3 (NLRP3) inflammasome activation. TBI model was established by cold-induced brain injury. Male C57BL/6 mice were randomly assigned into 3, 6, 12, 24, 48 and 72 h survival groups to investigate cerebral edema development with time and received 0, 5, 10, 20 and 40 mg/kg telmisartan by oral gavage, 1 h prior to TBI to determine the efficient anti-edemic dose. The therapeutic window was identified by post-treating 30 min, 1 h, 2 h and 4 h after TBI. Blood-brain barrier (BBB) integrity, the neurological function and histological injury were assessed, at the same time, the mRNA and protein expression levels of NLRP3 inflammasome, IL-1β and IL-18 concentrations in peri-contused brain tissue were measured 24 h post TBI. The results showed that the traumatic cerebral edema occurred from 6 h, reached the peak at 24 h and recovered to the baseline 72 h after TBI. A single oral dose of 5, 10 and 20 mg/kg telmisartan could reduce cerebral edema. Post-treatment up to 2 h effectively limited the edema development. Furthermore, prophylactic administration of telmisartan markedly inhibited BBB impairment, NLRP3, apoptotic speck-containing protein (ASC) and Caspase-1 activation, as well as IL-1β and IL-18 maturation, subsequently improved the neurological outcomes. In conclusion, telmisartan can reduce traumatic cerebral edema by inhibiting the NLRP3 inflammasome-regulated IL-1β and IL-18 accumulation.
Similar content being viewed by others
References
Kimbler DE, Shields J, Yanasak N, et al. Activation of P2X7 promotes cerebral edema and neurological injury after traumatic brain injury in mice. PLoS One, 2012,7(7): e41229
Kahle KT, Walcott BP, Simard JM. Continuous hyperosmolar therapy for traumatic brain injury-associated cerebral edema: as good as it gets, or an iatrogenic secondary insult? J Clin Neurosci, 2013,20(1):30–31
Zhang B, Wang B, Cao S, et al. Epigallocatechin-3-Gallate (EGCG) attenuates traumatic brain injury by inhibition of edema formation and oxidative stress. Korean J Physiol Pharmacol, 2015,19(6):491–497
Alves JL. Blood-brain barrier and traumatic brain injury. J Neurosci Res, 2014,92(2):141–147
Huang LQ, Zhu GF, Deng YY, et al. Hypertonic saline alleviates cerebral edema by inhibiting microglia-derived TNF-a and IL-1β-induced Na-K-Cl Cotransporter up-regulation. J Neuroinflammation, 2014,11(11):102
Ma Q, Chen S, Hu Q, et al. NLRP3 inflammasome contributes to inflammation after intracerebral hemorrhage. Ann Neurol, 2014,75(2):209–219
Liu HD, Li W, Chen ZR, et al. Expression of the NLRP3 inflammasome in cerebral cortex after traumatic brain injury in a rat model. Neurochem Res, 2013,38(10):2072–2083
Fann DY, Lee SY, Manzanero S, et al. Intravenous immunoglobulin suppresses NLRP1 and NLRP3 inflammasome-mediated neuronal death in ischemic stroke. Cell Death Dis, 2013,4:e790
Timaru-Kast R, Luh C, Gotthardt P, et al. Influence of age on brain edema formation, secondary brain damage and inflammatory response after brain trauma in mice. PLoS One, 2012,7(8):e43829
Abdul-Muneer PM, Chandra N, Haorah J. Interactions of oxidative stress and neurovascular inflammation in the pathogenesis of traumatic brain injury. Mol Neurobiol, 2015,51(3):966–979
Shiozaki T, Hayakata T, Tasaki O, et al. Cerebrospinal fluid concentrations of anti-inflammatory mediators in early-phase severe traumatic brain injury. Shock, 2005,23(5):406–410
Hayakata T, Shiozaki T, Tasaki O, et al. Changes in CSF S100B and cytokine concentrations in early-phase severe traumatic brain injury. Shock, 2004,22(2):102–107
Chiaretti A, Genovese O, Aloe L, et al. Interleukin 1beta and interleukin 6 relationship with paediatric head trauma severity and outcome. Childs Nerv Syst, 2005,21(3):185–193
Clausen F, Hånell A, Björk M, et al. Neutralization of interleukin-1beta modifies the inflammatory response and improves histological and cognitive outcome following traumatic brain injury in mice. Eur J Neurosci, 2009,30(3):385–396
Jones NC, Prior MJ, Burden-Teh E, et al. Antagonism of the interleukin-1 receptor following traumatic brain injury in the mouse reduces the number of nitric oxide synthase-2-positive cells and improves anatomical and functional outcomes. Eur J Neurosci, 2005,22(1):72–78
Tehranian R, Andell-Jonsson S, Beni SM, et al. Improved recovery and delayed cytokine induction after closed head injury in mice with central overexpression of the secreted isoform of the interleukin-1 receptor antagonist. J Neurotrauma, 2002,19(8):939–951
Panahpour H, Nekooeian AA, Dehghani GA. Blockade of central angiotensin II AT1 receptor protects the brain from ischemia/reperfusion injury in normotensive rats. Iran J Med Sci, 2014,39(6):536–542
Villapol S, Balarezo MG, Affram K, et al. Neurorestoration after traumatic brain injury through angiotensin II receptor blockage. Brain, 2015,138(Pt 11):3299–3331
Panahpour H, Nekooeian AA, Dehghani GA. Candesartan attenuates ischemic brain edema and protects the blood-brain barrier integrity from ischemia/reperfusion injury in rats. Iran Biomed J, 2014,18(4):232–238
Kono S, Kurata T, Sato K, et al. Neurovascular protection by telmisartan via reducing neuroinflammation in stroke-resistant spontaneously hypertensive rat brain after ischemic stroke. J Stroke Cerebrovasc Dis, 2015,24(3):537–547
Jung KH, Chu K, Lee ST, et al. Blockade of AT1 receptor reduces apoptosis, inflammation, and oxidative stress in normotensive rats with intracerebral hemorrhage. J Pharmacol Exp Ther, 2007,322(3):1051–1058
Garrido-Gil P, Joglar B, Rodriguez-Perez AI, et al. Involvement of PPAR-κ in the neuroprotective and anti-inflammatory effects of angiotensin type 1 receptor inhibition: effects of the receptor antagonist telmisartan and receptor deletion in a mouse MPTP model of Parkinson's disease. J Neuronflammation, 2012,9:38
Pang T, Wang J, Benicky J, et al. Telmisartan directly ameliorates the neuronal inflammatory response to IL-1β partly through the JNK/c-Jun and NADPH oxidase pathways. J Neuroinflammation, 2012,9:102
Wang J, Pang T, Hafko R, et al. Telmisartan ameliorates glutamate-induced neurotoxicity: roles of AT(1) receptor blockade and PPAR-κ activation. Neuropharmacology, 2014,79:249–261
Morita-Fujimura Y, Fujimura M, Kawase M, et al. Early decrease in apurinic/apyrimidinic endonuclease is followed by DNA fragmentation after cold injury-induced brain trauma in mice. Neuroscience, 1999,93(4):1465–1473
Zeynalov E, Jones SM, Seo JW, et al. Arginine-vasopressin receptor blocker conivaptan reduces brain edema and blood-brain barrier disruption after experimental stroke in mice. PLoS One, 2015,10(8):e0136121
Timaru-Kast R, Wyschkon S, Luh C, et al. Delayed inhibition of angiotensin II receptor type 1 reduces secondary brain damage and improves functional recovery after experimental brain trauma. Crit Care Med, 2012,40(3):935–944
Hu X, Li P, Guo Y, et al. Microglia/macrophage polarization dynamics reveal novel mechanism of injury expansion after focal cerebral ischemia. Stroke, 2012,43(11): 3063–3070
Michinaga S, Koyama Y. Pathogenesis of brain edema and investigation into anti-edema drugs. Int J Mol Sci, 2015,16(5):9949–9975
Gohlke P, Weiss S, Jansen A, et al. AT1 receptor antagonist telmisartan administered peripherally inhibits central responses to angiotensin II in conscious rats. J Pharmacol Exp Ther, 2001,298(1):62–70
Billecke SS, Marcovitz PA. Long-term safety and efficacy of telmisartan/amlodipine single pill combination in the treatment of hypertension. Vasc Health Risk Manag, 2013,9:95–104
Pushpa VH, Shetty KP, Suresha RN, et al. Evaluation and comparison of anticonvulsant activity of telmisartan and olmesartan in experimentally induced animal models of epilepsy. J Clin Diagn Res, 2014,8(10):HC08–11
Yang Z, Zhong L, Xian R, et al. MicroRNA-223 regulates inflammation and brain injury via feedback to NLRP3 inflammasome after intracerebral hemorrhage. Mol Immunol, 2015,65(2):267–276
Tan CC, Zhang JG, Tan MS, et al. NLRP1 inflammasome is activated in patients with medial temporal lobe epilepsy and contributes to neuronal pyroptosis in amygdala kindling-induced rat model. J Neuroinflammation, 2015,12:18
Kaushal V, Dye R, Pakavathkumar P, et al. Neuronal NLRP1 inflammasome activation of Caspase-1 coordinately regulates inflammatory interleukin-1-beta production and axonal degeneration-associated Caspase-6 activation. Cell Death Differ, 2015,22(10):1676–1686
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was supported by grants from the National Natural Science Foundation of China (No. 81270239) and the Natural Science Foundation of Hubei Province of China (No. 2014CFB200).
Rights and permissions
About this article
Cite this article
Wei, X., Hu, Cc., Zhang, Yl. et al. Telmisartan reduced cerebral edema by inhibiting NLRP3 inflammasome in mice with cold brain injury. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 36, 576–583 (2016). https://doi.org/10.1007/s11596-016-1628-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-016-1628-1