Abstract
Objective
To examine the relationship between Edmonton Obesity Staging System (EOSS) and perioperative complications as well as surgical procedure.
Background
The application of EOSS for the selection of patients with obesity is a more comprehensive measure of obesity-related diseases and a predictor of mortality than body mass index (BMI).
Methods
This was a nationwide cohort study using prospectively inserted data from the German register for obesity and metabolic surgery StuDoQ|MBE. All patients undergoing sleeve gastrectomy (SG), Roux-en Y gastric bypass (RYGB), and one-anastomosis gastric bypass (OAGB) between February 2015 and July 2017 as a primary treatment for severe obesity were included. Data included gender, age, BMI, ASA score, EOSS, early postoperative complications next to the Clavien-Dindo grading system, readmission, and 30-day mortality.
Results
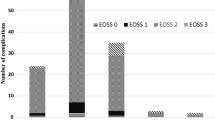
A total of 9437 patients were included. The mean BMI was 49.5 kg/m2 ± 7.8 (range 35–103.5). The total postoperative complication rate was 5.3%, with the highest rate in EOSS 3 (7.8%) and 4 (6.8%). Thirty-day mortality was 0.2% with the highest mortality after SG in EOSS 3 (1.16%) and EOSS 4 (0.92%) (p = 0.0068). Crosstabs showed a prevalence of Clavien-Dindo III and IV complications of 3.4% (SG), 3.6% (RYGB), and 1.6% (OAGB) in EOSS 2 (p = 0.0032) and 3.5% (SG), 5.1% (RYGB), and 5.6% (OAGB) in EOSS 3.
Conclusion
The highest postoperative complications and mortality occurred in patients with EOSS ≥ 3. SG and OAGB could be the procedure of choice to reduce perioperative morbidity; nevertheless, it has to be in mind that in EOSS ≥ 3, SG has the highest mortality.
Trial Registration
ClinicalTrials.gov Identifier NCT03556059.

Similar content being viewed by others
Abbreviations
- BMI:
-
Body mass index
- EOSS:
-
Edmonton Obesity Staging System
- T2DM:
-
Type 2 diabetes mellitus
- SG:
-
Sleeve gastrectomy
- RYGB:
-
Roux-en-Y gastric bypass
- OAGB:
-
One-anastomosis gastric bypass
- SD:
-
Standard deviation
- GERD:
-
Gastroesophageal reflux disease
- ASA:
-
American Society of Anesthesiologists
- MAS:
-
Metabolic acuity score
- OSAS:
-
Obstructive sleep apnea syndrome
References
De Luca M, Angrisani L, Himpens J, et al. Indications for surgery for obesity and weight-related diseases: position statements from the International Federation for the Surgery of Obesity and Metabolic Disorders (IFSO). Obes Surg. 2016;26(8):1659–96.
Sharma AM, Kushner RF. A proposed clinical staging system for obesity. Int J Obes. 2009;33(3):289–95.
Kuk JL, Ardern CI, Church TS, et al. Edmonton Obesity Staging System: association with weight history and mortality risk. Appl Physiol Nutr Metab. 2011;36(4):570–6.
Padwal RS, Pajewski NM, Allison DB, et al. Using the Edmonton obesity staging system to predict mortality in a population-representative cohort of people with overweight and obesity. CMAJ. 2011;183(14):E1059–66.
Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric-metabolic surgery versus conventional medical treatment in obese patients with type 2 diabetes: 5 year follow-up of an open-label, single-centre, randomised controlled trial. Lancet. 2015;386(9997):964–73.
Schauer PR, Bhatt DL, Kashyap SR. Bariatric surgery or intensive medical therapy for diabetes after 5 years. N Engl J Med. 2017;376(20):1997.
Angrisani L, Santonicola A, Iovino P, et al. IFSO worldwide survey 2016: primary, endoluminal, and revisional procedures. Obes Surg. 2018;28(12):3783–94.
Chiappetta S, Stier C, Squillante S, et al. The importance of the Edmonton Obesity Staging System in predicting postoperative outcome and 30-day mortality after metabolic surgery. Surg Obes Relat Dis. 2016;12(10):1847–55.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240(2):205–13.
DeMaria EJ, Murr M, Byrne TK, et al. Validation of the obesity surgery mortality risk score in a multicenter study proves it stratifies mortality risk in patients undergoing gastric bypass for morbid obesity. Ann Surg. 2007;246(4):578–82. discussion 83-4
Blackstone RP, Cortes MC. Metabolic acuity score: effect on major complications after bariatric surgery. Surg Obes Relat Dis. 2010;6(3):267–73.
Fischer L, Nickel F, Sander J, et al. Patient expectations of bariatric surgery are gender specific--a prospective, multicenter cohort study. Surg Obes Relat Dis. 2014;10(3):516–23.
Seetharamaiah S, Tantia O, Goyal G, et al. LSG vs OAGB-1 year follow-up data-a randomized control trial. Obes Surg. 2017;27(4):948–54.
Quan Y, Huang A, Ye M, et al. Efficacy of laparoscopic mini gastric bypass for obesity and type 2 diabetes mellitus: a systematic review and meta-analysis. Gastroenterol Res Pract. 2015;2015:152852.
Lee WJ, Chong K, Lin YH, et al. Laparoscopic sleeve gastrectomy versus single anastomosis (mini-) gastric bypass for the treatment of type 2 diabetes mellitus: 5-year results of a randomized trial and study of incretin effect. Obes Surg. 2014;24(9):1552–62.
Taha O, Abdelaal M, Abozeid M, et al. Outcomes of omega loop gastric bypass, 6-years experience of 1520 cases. Obes Surg. 2017;27(8):1952–60.
Zhu M, Wang Q, Luo Z, et al. Development and validation of a prognostic signature for preoperative prediction of overall survival in gastric cancer patients. Oncol Targets Ther. 2018;11:8711–22.
Aminian A, Brethauer SA, Andalib A, et al. Individualized metabolic surgery score: procedure selection based on diabetes severity. Ann Surg. 2017;266(4):650–7.
Stroh C, Kockerling F, Volker L, et al. Results of more than 11,800 sleeve gastrectomies: data analysis of the German bariatric surgery registry. Ann Surg. 2016;263(5):949–55.
El-Sayes IA, Frenken M, Weiner RA. Management of leakage and stenosis after sleeve gastrectomy. Surgery. 2017;162(3):652–61.
Tolone S, Cristiano S, Savarino E, et al. Effects of omega-loop bypass on esophagogastric junction function. Surg Obes Relat Dis. 2016;12(1):62–9.
Peterli R, Wolnerhanssen BK, Peters T, et al. Effect of laparoscopic sleeve gastrectomy vs laparoscopic roux-en-Y gastric bypass on weight loss in patients with morbid obesity: the SM-BOSS randomized clinical trial. JAMA. 2018;319(3):255–65.
Nguyen NT, Nguyen B, Nguyen VQ, et al. Outcomes of bariatric surgery performed at accredited vs nonaccredited centers. J Am Coll Surg. 2012;215(4):467–74.
Nguyen NT, Paya M, Stevens CM, et al. The relationship between hospital volume and outcome in bariatric surgery at academic medical centers. Ann Surg. 2004;240(4):586–93. discussion 93-4
Daigle CR, Brethauer SA, Tu C, et al. Which postoperative complications matter most after bariatric surgery? Prioritizing quality improvement efforts to improve national outcomes. Surg Obes Relat Dis. 2018;14(5):652–7.
Zafar SN, Miller K, Felton J, et al. Postoperative bleeding after laparoscopic Roux en Y gastric bypass: predictors and consequences. Surg Endosc. 2019;33(1):272–80.
Quirante FP, Montorfano L, Rammohan R, et al. Is bariatric surgery safe in the elderly population? Surg Endosc. 2017;31(4):1538–43.
Khidir N, El-Matbouly M, Al Kuwari M, et al. Incidence, indications, and predictive factors for ICU admission in elderly, high-risk patients undergoing laparoscopic sleeve gastrectomy. Obes Surg. 2018;28(9):2603–8.
Felsenreich DM, Ladinig LM, Beckerhinn P, et al. Update: 10 years of sleeve gastrectomy-the first 103 patients. Obes Surg. 2018;28(11):3586–94.
Sebastianelli L, Benois M, Vanbiervliet G, et al. Systematic endoscopy 5 years after sleeve gastrectomy results in a high rate of Barrett’s esophagus: results of a multicenter study. Obes Surg. 2019;29(5):1462–9.
Musella M, Susa A, Manno E, et al. Complications following the mini/one anastomosis gastric bypass (MGB/OAGB): a multi-institutional survey on 2678 patients with a mid-term (5 years) follow-up. Obes Surg. 2017;27(11):2956–67.
Baig SJ, Priya P, Mahawar KK, et al. Indian bariatric surgery outcome reporting G. weight regain after bariatric surgery-a multicentre study of 9617 patients from Indian bariatric surgery outcome reporting group. Obes Surg. 2019;29(5):1583–92.
Acknowledgments
This work has been conducted using the StuDoQ|MBE registry provided by the Study, Documentation and Quality Center (Studien-, Dokumentations- und Qualitätszentrum, StuDoQ) of the German Society for General Surgery (Deutsche Gesellschaft für Allgemein- und Viszeralchirurgie, DGAV) with the ID StuDoQ-2017-0019.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Statement of Human and Animal Rights
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Statement of Informed Consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chiappetta, S., Stier, C., Weiner, R.A. et al. The Edmonton Obesity Staging System Predicts Perioperative Complications and Procedure Choice in Obesity and Metabolic Surgery—a German Nationwide Register-Based Cohort Study (StuDoQ|MBE). OBES SURG 29, 3791–3799 (2019). https://doi.org/10.1007/s11695-019-04015-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-019-04015-y