Abstract
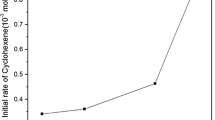
2-(2’-quinolyl) benzimidazole (QBIMH) anchored on to the crosslinked polymer beads of chloromethylated poly (styrene-co-divinyl benzene) (PSDVB) was reacted with sodium tetrachloropalladate (Na2PdCl4) to form polymer-supported palladium complex. The immobilized palladium complex was treated with sodium borohydride and activated. Multifarious approach supported the characterization of polymer-anchored [2-(2’-quinolyl) benzimidazole]palladium complex (PSDVB-QBIM-PdCl2) and its activated form. The optimal activated complex catalyzed the hydrogenation of olefins, Schiff bases and nitro compounds under ambient conditions. The kinetic studies were carried out for the hydrogenation of certain olefins. The recycling ability of the supported and the unsupported catalysts was showcased.
















Similar content being viewed by others
References
Alexander S, Udayakumar V, Nararaju N, Gayathri V (2010) Hydrogenation of substituted nitroarenes by a polymer-bound palladium(II) Schiff base catalyst. Trans Met Chem 35:427–435. https://doi.org/10.1007/s11243-009-9320-y
Alexander S, Udayakumar V, Gayathri V (2012) Catalytic activity of a supported palladium–benzimidazole complex toward alkene hydrogenation. Transition Met Chem 37:367–372. https://doi.org/10.1007/s11243-012-9597-0
Allan JC (1992) Development of organopalladium(IV) chemistry: fundamental aspects and systems for studies of mechanism in organometallic chemistry and catalysis. Acc Chem Res 25:83–90. https://doi.org/10.1021/ar00014a005
Antony R, Tembe GL, Ravindranathan M, Ram RN (2003) Polymer-supported palladium(II) complexes and their catalytic study. J App Polym Sci 90:370–378. https://doi.org/10.1002/app.12587
Blaser HU (2003) Asymmetric catalysis on industrial scale. Weinheim, Schmidt E (ed).Wiley, Berlin
Brown JM, Cooley NA (1988) Carbon–carbon bond formation through organometallic elimination reactions. Chem Rev 88:1031–1046. https://doi.org/10.1021/cr00089a003
Brown HC, Rothberg I, Van der Jagt DL (1972) Convenient synthetic routes to the 5,6-trimethylenenorbornanones. J Org Chem 37:4098–4103. https://doi.org/10.1021/jo00798a027
Chen X, Wang W, Zhu H, Yang W, Ding Y (2018) Pd0-PyPPh2@porous organic polymer: Efficient heterogeneous nanoparticle catalyst for dehydrogenation of 3-methyl-2-cyclohexen-1-one without extra oxidants and hydrogen acceptors. Mol Catal 456:49–56. https://doi.org/10.1016/j.mcat.2018.07.007
David G, Simionescu BC, Simionescu CI (2007) Synthesis of polymer supported transition metal catalysts for olefin epoxidation. Rev Roum Chem 52:875–882
Ferraro JR (1971) Low-Frequency vibrations of inorganic and coordination compounds. Plenum Press, New York, p 321
Hisano T, Ichikawa M, Tsumoto M, Tasaki M (1982) Synthesis of benzoxazoles, benzothiazoles and benzimidazoles and evaluation of their antifungal, insecticidal and herbicidal activities. Chem Pharm Bull 30:2996–3004. https://doi.org/10.1248/cpb.30.2996
Hodge PP, Sherrington DC (1980) Polymer supported: reactions in organic synthesis. Wiley, New York
Islam M, Bose A, Mal D, Saha CR (1998) Zeolites and catalysis: synthesis reactions and applications. J Chem Res (s) 1:44–45. https://doi.org/10.1039/A703426A
Jainamma M, Mahadevan V (1990) Transition metal ions supported on hydrogels as hydrogenation catalysts. J Mol Catal 60:189–207. https://doi.org/10.1016/0304-5102(90)85269-N
L’Argentiere PC, Cagnola EA, Quiroga ME, Liprandi DA (2002) A palladium tetra-coordinated complex as catalyst in the selective hydrogenation of 1-heptyne. Appl Catal A Gen 226:253–263. https://doi.org/10.1016/S0926-860X(01)00911-5
Lautsch W, Broser W, Rothkegel W, Biedermann W, Doering U, Zoschke H (1952) Some types of organic high polymers and permutoids provided with active groups: their synthesis, optical properties, and reactivity. J Polym Sci 8:191–213. https://doi.org/10.1002/pol.1952.120080206
Li W, Binglin He (1990) The study of hydrogenations of olefins catalyzed by polymer-supported palladium-lanthanoid bimetallic catalysts. React Polym 12:45–50. https://doi.org/10.1016/0923-1137(90)90060-H
Mani R, Mahadevan V, Srinivasan M (1990) Studies on the hydrogenation of olefins using polymer-bound palladium (II) species. Brit Poly J 22:177–184. https://doi.org/10.1002/pi.4980220302
Mukherjee D (2008) Potential application of palladium nanoparticles as selective recyclable hydrogenation catalysts. J Nanopart Res 10:429–436. https://doi.org/10.1007/s11051-007-9270-2
Papp M, Urbán B, Drotár E, Skoda-Földes R (2015) Mono- and double carbonylation of iodobenzene in the presence of reusable supported palladium catalysts. Green Process Synth 4:103–115. https://doi.org/10.1515/gps-2014-0093
Perrin DD, Armanego WCF, Perrin DR (1966) Purification of Laboratory chemicals, Chapter-3, I (ed) Pergamon Press, New York
Pomogailo AD (1992) Specific characteristics of catalysis by polymer-immobilised complexes Russ. Chem Rev 61:133–153. https://doi.org/10.1070/rc1992v061n02abeh000936
Pomogailo AD (2004) Catalysis by Heterogenized Metal Polymers: Advances and Prospects. Kinet Catal 45:61–103. https://doi.org/10.1023/B:KICA.0000016109.46262.ce
Randall Lee T, George Whitesides M (1992) Heterogeneous, platinum-catalyzed hydrogenations of (diolefin)dialkylplatinum(II) complexes. Acc Chem Res 25:266–272. https://doi.org/10.1021/ar00018a004
Roe A, Montgonery JA (1953) Kinetics of the catalytic hydrogenation of certain schiff bases. J Am Chem Soc 75:910–912. https://doi.org/10.1021/ja01100a040
Selvaraj PC, Mahadevan V (1997) Polymer-Supported Palladium and Rhodium Species as Hydrogenation Catalysts. J Polym Sci Part a: Polym Chem 35:105–122. https://doi.org/10.1002/(SICI)1099-0518(19970115)35:1%3C105::AID-POLA12%3E3.0.CO;2-O
Shilpa ML, Gayathri V (2013) Trans Met Chem 38:53–62. https://doi.org/10.1007/s11243-012-9660-x
Skala D, Hanika J (2003) Kinetics of dicyclopentadiene hydrogenation using Pd/C catalyst. Pet Coal 45:105–108
Trakarnpruk W, Kanjina W (2008) Polymer -Supported Cobalt Catalysts for Hydrogenation of Cyclooctene Thammasat. Int J Sc Tech 2:1–10
Udayakumar V, Alexander S, Gayathri V, Shivakumaraiah BV (2011) Hydrogenation of olefins catalyzed by polymer-supported palladium-imidazole. Chin J Catal 32:280–285. https://doi.org/10.1016/S1872-2067(10)60176-3
Wen H, Yao K, Zhang Y, Zhou Z, Kirsching A (2009) Catalytic transfer hydrogenation of aromatic nitro compounds in presence of polymer-supported nano-amorphous Ni–B catalyst. Catal Commun 10:1207–1211. https://doi.org/10.1016/j.catcom.2009.01.030
Zhang W, Peng L, Deng C, Zhang Y, Qian H (2018) A Simple Method for Preparing Imidazolium-Based Polymer as Solid Catalyst for Suzuki-Miyaura Reaction. Mol Catal 445:170–178. https://doi.org/10.1016/j.mcat.2017.11.036
Zhu Y, Wang WD, Sun X, Fan M, Xiwei H, Dong Z (2020) Palladium nanoclusters confined in MOF@COP as a novel nanoreactor for catalytic hydrogenation ACS. Appl Mater Interfaces 12:7285–7294. https://doi.org/10.1021/acsami.9b21802
Acknowledgements
Authors wish to thank UGC, New Delhi, India, for Major Research Project [F no. 39-741/2010(SR)]. Department of Chemistry, Bangalore University, Bengaluru, for instrumentation facilities and Thermax Ltd. for providing PSDVB are gratefully acknowledged. Roopashree B thanks Management and Principal, JSS Academy of Technical Education, Bengaluru, for the encouragement. Authors are grateful to Dr. M.S.Hegde, IISc., Bengaluru, for XPS studies and Prof. Puttaswamy, Dept. of Studies in Chemistry, Bangalore University, Bangalore, for his kind help in deriving rate law equation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, I, Dr. Gayathri Virupaiah, corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lakshminarayana, S.M., Boregowda, R. & Virupaiah, G. Reusable palladium–quinolyl benzimidazole complex immobilized on a polymer for the hydrogenation of organic substrates. Chem. Pap. 77, 3589–3601 (2023). https://doi.org/10.1007/s11696-023-02721-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-023-02721-7