Abstract
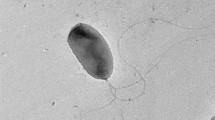
The present study has been conducted towards isolation of bacteria capable of producing heliotropin via microbial conversion. Strain ZMT-1 capable of synthesizing heliotropin efficiently was obtained by enrichment culture of soil samples and a high-throughput screening method, and identified as Serratia liquefaciens. Heliotropin was identified by gas chromatography and gas chromatography–mass spectrometry analysis. In addition, the culture medium was optimized to improve heliotropin yield by experimental designs. The application of a Plackett–Burman design found that NH4NO3 and K2HPO4•3H2O have significant effects on heliotropin production. Central composite design experiments were further used to predict the optimal concentrations of NH4NO3 and K2HPO4•3H2O, which were 1.0 and 0.5 g/l, respectively. After the optimization of cultural medium, heliotropin yield was increased by 4.52-fold when compared with the unoptimized minimal medium. This study is the first to report the biosynthesis of heliotropin by S. liquefaciens. S. liquefaciens ZMT-1 can produce heliotropin efficiently, indicating its potential as one heliotropin-producing strain.






Similar content being viewed by others
References
Gupta, C., Prakash, D., & Gupta, S. (2015). A biotechnological approach to microbial based perfumes and flavours. Journal of Microbiology and Experimentation, 2, 1–8.
Akacha, N. B., & Gargouri, M. (2015). Microbial and enzymatic technologies used for the production of natural aroma compounds: synthesis, recovery modeling, and bioprocesses. Food and Bioproducts Processing, 94, 675–706.
Krings, U., & Berger, R. G. (1998). Biotechnological production of flavours and fragrances. Applied Microbiology and Biotechnology, 49, 1–8.
Han, D. F., Ryu, J. Y., Lee, H., & Hur, H. G. (2013). Bacterial biotransformation of phenylpropanoid compounds for producing flavor and fragrance compounds. Journal of Korean Society for Applied Biological Chemistry, 56, 125–133.
Yamagishi, R., Yokomaku, A., Omoto, F., Misa, K., Takada, K., Yoshimatsu, S., ABE, A., & Hayashi, M. (2010). Sleep-improving effects of the aromatic compound heliotropin. Sleep and Biological Rhythms, 8, 254–260.
Frosch, P. J., Johansen, J. D., Menne, T., Pirker, C., Rastogi, S. C., Andersen, K. E., Bruze, M., Goossens, A., Lepoittevin, J. P., & White, I. R. (2002). Further important sensitizers in patients sensitive to fragrances. Contact Dermatitis, 47, 279–287.
Cortés-Salazar, F., Avella-Moreno, E., Cortés, M. T., & Suárez-Herrera, M. F. (2007). Study of the electrochemical oxidation process of 3,4-(methylenedioxy)phenylmethanol in non-aqueous solvents. Journal of Electroanalytical Chemistry, 606, 1–7.
Bellardita, M., Loddo, V., Palmisano, G., Pibiri, I., Palmisano, L., & Augugliaro, V. (2014). Photocatalytic green synthesis of piperonal in aqueous TiO2 suspension. Applied Catalysis B: Environmental, 144, 607–613.
Alvarez, H. M., Andrade, L. D., Pereira Jr., N., Muri, E. M. F., Horn Jr., A., Barbosa, D. P., & Antunes, O. A. C. (2007). Catalytic oxidation of isosafrol by vanadium complexes. Catalysis Communications, 8, 1336–1340.
Li, X., Choi, Y., Yanakawa, Y., & Park, T. (2014). Piperonal prevents high-fat diet-induced hepatic steatosis and insulin resistance in mice via activation of adiponectin/AMPK pathway. International Journal of Obesity, 38, 140–147.
Grimshaw, J., & Cheng, H. (1994). The conversion of isosafrole to piperonal and anethole to anisaldehyde electrochemical active manganese dioxide. Electrochimica Acta, 39, 497–499.
María, A. L., & María, A. S. (2006). Production of food aroma compounds: microbial and enzymatic methodologies. Food Technology and Biotechnology, 44, 335–353.
Santos, A. S., Pereira Jr., N., Silva, I. I. D., & Antunes, O. A. C. (2003). Microbiological oxidation of isosafrole into piperonal. Applied Biochemistry and Biotechnology, 107, 649–658.
Santos, A. S., Pereira Jr., N., Silva, I. I. D., & Antunes, O. A. C. (2004). Peroxidase catalyzed microbiological oxidation of isosafrol into piperonal. Process Biochemistry, 39, 2269–2275.
Shimoni, E., Baasov, T., Ravid, U., & Shoham, Y. (2003). Biotransformations of propenylbenzenes by an Arthrobacter sp. and its t-anethole blocked mutants. Journal of Biotechnology, 105, 61–70.
Doi, S., Hashimoto, Y., Tomita, C., Kumano, T., & Kobayashi, M. (2016). Discovery of piperonal-converting oxidase involved in the metabolism of a botanical aromatic aldehyde[J]. Scientific Reports-UK, 2016, 6. doi:10.1038/srep38021.
Shimoni, E., Ravid, U., & Shoham, Y. (2000). Isolation of a Bacillus sp. capable of transforming isoeugenol to vanillin. Journal of Biotechnology, 78, 1–9.
Krebs, K. G., Heusser, D., & Wimmer, H. (1969). In E. Stahl (Ed.), Thin-layer chromatography, a laboratory handbook (pp. 854–909). Berlin: Springer.
Shimoni, E., Baasov, T., Ravid, U., & Shoham, Y. (2002). The trans-anethole degradation pathway in an Arthrobacter sp.*. The Journal of Biological Chemistry, 277, 11866–11872.
Holt, J. G., Krieg, N. R., Sneath, P. H. A., Staley, J. T., & Williams, S. T. (1994). Bergey’s manual of determinative bacteriology (9th ed.). Baltimore: Williams and Wilkins.
Tamura, K., Stecher, G., Peterson, D., Filipski, A., & Kumar, S. (2013). MEGA6: molecular evolutionary genetics analysis version 6.0. Molecular Biology and Evolution, 30, 2725–2729.
Faveri, D. D., Torre, P., Aliakbarian, B., Domínguez, J. M., Perego, P., & Converti, A. (2007). Response surface modeling of vanillin production by Escherichia coli JM109pBB1. Biochemical Engineering Journal, 36, 268–275.
Shieh, C. J., & Chang, S. W. (2001). Optimized synthesis of lipase-catalyzed hexyl acetate in n-hexane by response surface methodology. Journal of Agricultural and Food Chemistry, 49, 1203–1207.
Rabenhorst, J. & Hopp, R. (1991). Verfahren zur herstellung von naturalichem vanillin. Deutsches Patentampt DE 39 20 A1.
Hopp, R. (1993). Some highlights of H & R research. A review of nearly 120 years of research at Haarmann & Reimer. In R. Hopp & K. Mori (Eds.), Recent developments in flavor and fragrance Chemistry. New York: VCH.
Yao, R. S., Sun, M., Wang, C. L., & Deng, S. S. (2006). Degradation of phenolic compounds with hydrogen peroxide catalyzed by enzyme from Serratia Marcescens AB 90027. Water Research, 40, 3091–3098.
Delaforge, M., Jaouen, M., & Bouille, G. (1999). Inhibitory metabolite complex formation of methylenedioxymethamphetamine with rat and human cytochrome P450. Particular involvement of CYP 2D. Environmental Toxicology and Pharmacology, 7, 153–158.
Kakko, I., Toimela, T., & Tahti, H. (2000). Piperonyl butoxide potentiates the synaptosome ATPase inhibiting effect of pyretrin. Chemosphere, 40, 301–305.
Acknowledgements
The authors are grateful to the financial support from the Fundamental Research Funds for the Central Universities (Nos. JUSRP 51504 and JUSRP 116031).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Zhao, M., Zheng, P., Chen, P. et al. Biosynthesis of Heliotropin by a Novel Strain of Serratia liquefaciens . Appl Biochem Biotechnol 183, 1282–1294 (2017). https://doi.org/10.1007/s12010-017-2497-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-017-2497-1