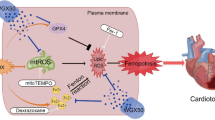
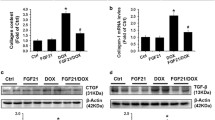
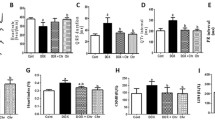
Abstract
As a widely used anti-tumor anthracycline, the accumulation of Doxorubicin (DOX) in body causes irreparable cardiomyocyte damage and therefore is limited in clinical application. Strategies to prevent from DOX-associated cardiotoxicity are urgent for patients who undergo DOX-based chemotherapy. Since oxidative stress injury being the major reason for myocardial toxicity of DOX, here we demonstrated that, Alpha-lipoic acid (ALA), which is a reductive agent, plays a cardioprotective role in attenuating DOX-induced cardiotoxicity by inhibiting pyruvate dehydrogenase kinase 4 (PDK4) expression. In vivo, the beneficial effect of ALA was evidenced by increased survival rate, mechanical contraction, and oxidative phosphorylation, while decreased reactive oxidative species (ROS) and apoptosis. In vitro, PDK4 overexpression remarkably increased DOX-induced apoptosis and ROS production in H9C2 cells. Notably, the protective effect of ALA was abrogated by PDK4 overexpression. We further used PDK4 knockout mice to identify the role of PDK4 in DOX-induced cardiotoxicity. Results elicited that PDK4 deficiency showed a consistent effect in protecting DOX cardiotoxicity as ALA treatment, which was evidenced by restored redox homeostasis and mitochondrial metabolism, finally inhibited myocardial injury. In conclusion, the cardioprotective role of ALA against DOX cardiotoxicity was dependent on PDK4-mediated regulation of oxidative stress and mitochondria metabolism.
Graphical abstract






Similar content being viewed by others
Data Availability
The datasets generated and used during current study are available from the corresponding author on reasonable request.
Change history
03 April 2023
This article has been retracted. Please see the Retraction Notice for more detail: https://doi.org/10.1007/s12012-023-09791-9
References
Carvalho, F. S., Burgeiro, A., Garcia, R., Moreno, A. J., Carvalho, R. A., & Oliveira, P. J. (2014). Doxorubicin-induced cardiotoxicity: From bioenergetic failure and cell death to cardiomyopathy. Medical Research Reviews,34(1), 106–135. https://doi.org/10.1002/med.21280
Lipshultz, S. E., Franco, V. I., Miller, T. L., Colan, S. D., & Sallan, S. E. (2015). Cardiovascular disease in adult survivors of childhood cancer. Annual Review Medicine,66, 161–176. https://doi.org/10.1146/annurev-med-070213-054849
Paulus, W. J., & Tschope, C. (2013). A novel paradigm for heart failure with preserved ejection fraction: Comorbidities drive myocardial dysfunction and remodeling through coronary microvascular endothelial inflammation. Journal of American College Cardiology,62(4), 263–271. https://doi.org/10.1016/j.jacc.2013.02.092
Wenningmann, N., Knapp, M., Ande, A., Vaidya, T. R., & Ait-Oudhia, S. (2019). Insights into doxorubicin-induced cardiotoxicity: molecular mechanisms, preventive strategies, and early monitoring. Molecular Pharmacology.,96(2), 219–232. https://doi.org/10.1124/mol.119.115725
Renu, K., AbilashTirupathi, V. G. P. B., & Arunachalam, S. (2018). Molecular mechanism of doxorubicin-induced cardiomyopathy: An update. European Journal of Pharmacology,818, 241–253. https://doi.org/10.1016/j.ejphar.2017.10.043
Govender, J., Loos, B., Marais, E., & Engelbrecht, A. M. (2014). Mitochondrial catastrophe during doxorubicin-induced cardiotoxicity: A review of the protective role of melatonin. Journal of Pineal Research,57(4), 367–380. https://doi.org/10.1111/jpi.12176
Carvalho, R. A., Sousa, R. P., Cadete, V. J., Lopaschuk, G. D., Palmeira, C. M., Bjork, J. A., & Wallace, K. B. (2010). Metabolic remodeling associated with subchronic doxorubicin cardiomyopathy. Toxicology,270(2–3), 92–98. https://doi.org/10.1016/j.tox.2010.01.019
Rochette, L., Ghibu, S., Muresan, A., & Vergely, C. (2015). Alpha-lipoic acid: Molecular mechanisms and therapeutic potential in diabetes. Canadian Journal of Physiology and Pharmacology,93(12), 1021–1027. https://doi.org/10.1139/cjpp-2014-0353
Kalivendi, S. V., Kotamraju, S., Zhao, H., Joseph, J., & Kalyanaraman, B. (2001). Doxorubicin-induced apoptosis is associated with increased transcription of endothelial nitric-oxide synthase. Effect of antiapoptotic antioxidants and calcium. Journal of Biological Chemistry,276(50), 47266–47276. https://doi.org/10.1074/jbc.M106829200
Wallace, K. B., Sardao, V. A., & Oliveira, P. J. (2020). Mitochondrial determinants of doxorubicin-induced cardiomyopathy. Circulation Research,126(7), 926–941. https://doi.org/10.1161/CIRCRESAHA.119.314681
Koleini, N., Nickel, B. E., Edel, A. L., Fandrich, R. R., Ravandi, A., & Kardami, E. (2019). Oxidized phospholipids in doxorubicin-induced cardiotoxicity. Chemico-Biological Interactions,303, 35–39. https://doi.org/10.1016/j.cbi.2019.01.032
Ziegler, D. (2004). Thioctic acid for patients with symptomatic diabetic polyneuropathy: A critical review. Treatments in Endocrinology,3(3), 173–189. https://doi.org/10.2165/00024677-200403030-00005
Henriksen, E. J. (2006). Exercise training and the antioxidant alpha-lipoic acid in the treatment of insulin resistance and type 2 diabetes. Free Radical Biology and Medicine,40(1), 3–12. https://doi.org/10.1016/j.freeradbiomed.2005.04.002
Salehi, B., Berkay, Y. Y., Antika, G., Boyunegmez, T. T., Fawzi, M. M., Lobine, D., Akram, M., Riaz, M., Capanoglu, E., & Sharopov, F. (2019). Insights on the use of alpha-lipoic acid for therapeutic purposes. Biomolecules. https://doi.org/10.3390/biom9080356
Li, W., Yin, L., Sun, X., Wu, J., Dong, Z., Hu, K., Sun, A., & Ge, J. (2020). Alpha-lipoic acid protects against pressure overload-induced heart failure via ALDH2-dependent Nrf1-FUNDC1 signaling. Cell Death and Diseases,11(7), 599. https://doi.org/10.1038/s41419-020-02805-2
Packer, L., & Cadenas, E. (2011). Lipoic acid: Energy metabolism and redox regulation of transcription and cell signaling. Journal of Clinical Biochemistry and Nutrition,48(1), 26–32. https://doi.org/10.3164/jcbn.11-005FR
Ng, F., & Tang, B. L. (2014). Pyruvate dehydrogenase complex (PDC) export from the mitochondrial matrix. Molecular Membrane Biology,31(7–8), 207–210. https://doi.org/10.3109/09687688.2014.987183
Gudiksen, A., & Pilegaard, H. (2017). PGC-1alpha and fasting-induced PDH regulation in mouse skeletal muscle. Physiological Reports. https://doi.org/10.14814/phy2.13222
Tian, L., Wu, D., Dasgupta, A., Chen, K. H., Mewburn, J., Potus, F., Lima, P. D. A., Hong, Z., Zhao, Y. Y., & Hindmarch, C. C. T. (2020). Epigenetic metabolic reprogramming of right ventricular fibroblasts in pulmonary arterial hypertension: A pyruvate dehydrogenase kinase-dependent shift in mitochondrial metabolism promotes right ventricular fibrosis. Circulation Research,126(12), 1723–1745. https://doi.org/10.1161/CIRCRESAHA.120.316443
Tareen, S. H., Kutmon, M., Arts, I. C., de Kok, T. M., Evelo, C. T., & Adriaens, M. E. (2019). Logical modelling reveals the PDC-PDK interaction as the regulatory switch driving metabolic flexibility at the cellular level. Genes and Nutrition,14, 27. https://doi.org/10.1186/s12263-019-0647-5
Sugden, M. C., & Holness, M. J. (2003). Recent advances in mechanisms regulating glucose oxidation at the level of the pyruvate dehydrogenase complex by PDKs. American Journal of Physiology and Endocrinology Metabolism,284(5), E855-862. https://doi.org/10.1152/ajpendo.00526.2002
Ackers-Johnson, M., Li, P. Y., Holmes, A. P., O’Brien, S. M., Pavlovic, D., & Foo, R. S. (2016). A simplified, langendorff-free method for concomitant isolation of viable cardiac myocytes and nonmyocytes from the adult mouse heart. Circulation Research,119(8), 909–920. https://doi.org/10.1161/CIRCRESAHA.116.309202
Dudek, M., Bednarski, M., Bilska, A., Iciek, M., Sokolowska-Jezewicz, M., Filipek, B., & Wlodek, L. (2008). The role of lipoic acid in prevention of nitroglycerin tolerance. European Journal Pharmacology,591(1–3), 203–210. https://doi.org/10.1016/j.ejphar.2008.06.073
Packer, L., Witt, E. H., & Tritschler, H. J. (1995). Alpha-lipoic acid as a biological antioxidant. Free Radical Biology and Medicine,19(2), 227–250. https://doi.org/10.1016/0891-5849(95)00017-r
He, L., Liu, B., Dai, Z., Zhang, H. F., Zhang, Y. S., Luo, X. J., Ma, Q. L., & Peng, J. (2012). Alpha lipoic acid protects heart against myocardial ischemia-reperfusion injury through a mechanism involving aldehyde dehydrogenase 2 activation. European Journal Pharmacology,678(1–3), 32–38. https://doi.org/10.1016/j.ejphar.2011.12.042
Wang, J., Wang, H., Hao, P., Xue, L., Wei, S., Zhang, Y., & Chen, Y. (2011). Inhibition of aldehyde dehydrogenase 2 by oxidative stress is associated with cardiac dysfunction in diabetic rats. Molecular Medicine,17(3–4), 172–179. https://doi.org/10.2119/molmed.2010.00114
Zhang, L., Zou, J., Chai, E., Qi, Y., & Zhang, Y. (2014). Alpha-lipoic acid attenuates cardiac hypertrophy via downregulation of PARP-2 and subsequent activation of SIRT-1. European Journal of Pharmacology,744, 203–210. https://doi.org/10.1016/j.ejphar.2014.09.037
Mohajeri, M., & Sahebkar, A. (2018). Protective effects of curcumin against doxorubicin-induced toxicity and resistance: A review. Critical Reviews in Oncology/Hematology,122, 30–51. https://doi.org/10.1016/j.critrevonc.2017.12.005
Songbo, M., Lang, H., Xinyong, C., Bin, X., Ping, Z., & Liang, S. (2019). Oxidative stress injury in doxorubicin-induced cardiotoxicity. Toxicology Letter,307, 41–48. https://doi.org/10.1016/j.toxlet.2019.02.013
Nebigil, C. G., & Desaubry, L. (2018). Updates in anthracycline-mediated cardiotoxicity. Frontiers in Pharmacology,9, 1262. https://doi.org/10.3389/fphar.2018.01262
Solmonson, A., & DeBerardinis, R. J. (2018). Lipoic acid metabolism and mitochondrial redox regulation. Journal of Biological Chemistry,293(20), 7522–7530. https://doi.org/10.1074/jbc.TM117.000259
Toker, A., & Newton, A. C. (2000). Cellular signaling: Pivoting around PDK-1. Cell,103(2), 185–188. https://doi.org/10.1016/s0092-8674(00)00110-0
Wang, X., Shen, X., Yan, Y., & Li, H. (2021). Pyruvate dehydrogenase kinases (PDKs): an overview toward clinical applications. Bioscience Reports. https://doi.org/10.1042/BSR20204402
Sugden, M. C., & Holness, M. J. (2006). Mechanisms underlying regulation of the expression and activities of the mammalian pyruvate dehydrogenase kinases. Archives Physiology and Biochemistry,112(3), 139–149. https://doi.org/10.1080/13813450600935263
Harris, R. A., Huang, B., & Wu, P. (2001). Control of pyruvate dehydrogenase kinase gene expression. Advances in Enzyme Regulation,41, 269–288. https://doi.org/10.1016/s0065-2571(00)00020-0
Woolbright, B. L., Rajendran, G., Harris, R. A., & Taylor, J. A., 3rd. (2019). Metabolic flexibility in cancer: Targeting the pyruvate dehydrogenase kinase: pyruvate dehydrogenase axis. Molecular Cancer Therapeutics,18(10), 1673–1681. https://doi.org/10.1158/1535-7163.MCT-19-0079
Harris, R. A., Bowker-Kinley, M. M., Huang, B., & Wu, P. (2002). Regulation of the activity of the pyruvate dehydrogenase complex. Advances in Enzyme Regulation,42, 249–259. https://doi.org/10.1016/s0065-2571(01)00061-9
Smolle, M., Prior, A. E., Brown, A. E., Cooper, A., Byron, O., & Lindsay, J. G. (2006). A new level of architectural complexity in the human pyruvate dehydrogenase complex. Journal of Biological Chemistry,281(28), 19772–19780. https://doi.org/10.1074/jbc.M601140200
Kolobova, E., Tuganova, A., Boulatnikov, I., & Popov, K. M. (2001). Regulation of pyruvate dehydrogenase activity through phosphorylation at multiple sites. Biochemical Journal,358(Pt 1), 69–77. https://doi.org/10.1042/0264-6021:3580069
Saunier, E., Benelli, C., & Bortoli, S. (2016). The pyruvate dehydrogenase complex in cancer: An old metabolic gatekeeper regulated by new pathways and pharmacological agents. International Journal of Cancer,138(4), 809–817. https://doi.org/10.1002/ijc.29564
Ferriero, R., Iannuzzi, C., Manco, G., & Brunetti-Pierri, N. (2015). Differential inhibition of PDKs by phenylbutyrate and enhancement of pyruvate dehydrogenase complex activity by combination with dichloroacetate. Journal of Inherited Metabolic Disease,38(5), 895–904. https://doi.org/10.1007/s10545-014-9808-2
Acknowledgements
The authors thank the support of Zhejiang Provincial Research Projects of Medical and Healthy Industries (grant nos. 2020KY424 and 2021KY485).
Funding
This work was supported by Zhejiang Provincial Research Projects of Medical and Healthy Industries (grant nos. 2020KY424 and 2021KY485).
Author information
Authors and Affiliations
Contributions
FG and JJ performed the formal analysis and wrote the original draft. HL supervised the study and validated the data. HM reviewed and edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical Approval
The study was approved by the Institutional Animal Care and Use Committee (IACUC) of Hangzhou Medical College (Reference no: HMC.NO.2020-98).
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Handling Editor: Y. James Kang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article has been retracted. Please see the retraction notice for more detail: https://doi.org/10.1007/s12012-023-09791-9"
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gong, F., Jin, J., Li, H. et al. RETRACTED ARTICLE: Alpha-Lipoic Acid Protects Against Doxorubicin-Induced Cardiotoxicity by Regulating Pyruvate Dehydrogenase Kinase 4. Cardiovasc Toxicol 22, 879–891 (2022). https://doi.org/10.1007/s12012-022-09766-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-022-09766-2