Abstract
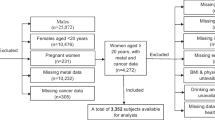
A growing body of evidence has pointed to a role of environmental chemical exposures in breast cancer etiology. This study was to examine the association between exposure to the endocrine-disrupting metals, including cadmium (Cd), lead (Pb), and mercury (Hg), and breast cancer in US women. A nationally representative subsample of 9260 women aged ≥ 20 years in the 2003–2012 National Health and Nutrition Examination Survey was analyzed for the association of blood levels of these metals with prevalent breast cancer using multivariate logistic regression models. Of the study participants, 284 women (weighted prevalence, 2.8%) were self-reported being diagnosed with breast cancer during 2003–2012. Breast cancer women showed significantly elevated blood levels of Cd and Pb, but not Hg. After adjusting for potential confounders, we found that women in all of the higher quartiles of blood lead levels (BLLs) had significantly increased odds ratio of prevalent breast cancer compared with those in the lowest quartile. However, a significant association with prevalent breast cancer was not seen with blood levels of either Cd or Hg. Our study demonstrates a potential relationship between lead exposure, measured as BLLs, and female breast cancer. Additional epidemiologic and mechanistic studies would further explore these interactions and elucidate the potential role of lead exposure in breast cancer etiology.
Similar content being viewed by others
References
Parkin DM, Muir CS. Cancer incidence in five continents. Comparability and quality of data. IARC Sci Publ. 1992;120:45–173.
Boulos DN, Ghali RR, Ibrahim EM, Boulos MN, AbdelMalik P. An eight-year snapshot of geospatial cancer research (2002-2009): clinico-epidemiological and methodological findings and trends. Med Oncol. 2011;28:1145–62.
Johnson-Thompson MC, Guthrie J. Ongoing research to identify environmental risk factors in breast carcinoma. Cancer. 2000;88:1224–9.
Brody JG, Rudel RA. Environmental pollutants and breast cancer. Environ Health Perspect. 2003;111:1007–19.
Brody JG, Moysich KB, Humblet O, Attfield KR, Beehler GP, Rudel RA. Environmental pollutants and breast cancer: epidemiologic studies. Cancer. 2007;109:2667–711.
Ekenga CC, Parks CG, Sandler DP. Chemical exposures in the workplace and breast cancer risk: a prospective cohort study. Int J Cancer. 2015;137:1765–74.
Rodgers KM, Udesky JO, Rudel RA, Brody JG. Environmental chemicals and breast cancer: an updated review of epidemiological literature informed by biological mechanisms. Environ Res. 2018;160:152–82.
Davis DL, Bradlow HL, Wolff M, Woodruff T, Hoel DG, Anton-Cuver H. Medical hypothesis: xenoestrogens as preventable causes of breast cancer. Environ Health Perspect. 1993;101:372–7.
Knower KC, To SQ, Leung YK, Ho SM, Clyne CD. Endocrine disruption of the epigenome: a breast cancer link. Endocr Relat Cancer. 2014;21:T33–55.
Lecomte S, Habauzit D, Charlier TD, Pakdel F. Emerging estrogenic pollutants in the aquatic environment and breast cancer. Genes (Basel). 2017;8:229.
Garcia-Esquinas E, Perez-Gomez B, Fernandez MA, Perez-Meixeira AM, Gil E, de Paz C, et al. Mercury, lead and cadmium in human milk in relation to diet, lifestyle habits and sociodemographic variables in Madrid (Spain). Chemosphere. 2011;85:268–76.
Darbre PD. Metalloestrogens: an emerging class of inorganic xenoestrogens with potential to add to the oestrogenic burden of the human breast. J Appl Toxicol. 2006;26:191–7.
Choe SY, Kim SJ, Kim HG, Lee JH, Choi Y, Lee H, et al. Evaluation of estrogenicity of major heavy metals. Sci Total Environ. 2003;312:15–21.
Martin MB, Reiter R, Pham T, Avellanet YR, Camara J, Lahm M, et al. Estrogen-like activity of metals in MCF-7 breast cancer cells. Endocrinology. 2003;144:2425–36.
Byrne C, Divekar SD, Storchan GB, Parodi DA, Martin MB. Metals and breast cancer. J Mammary Gland Biol Neoplasia. 2013;18:63–73.
Gallagher CM, Chen JJ, Kovach JS. Environmental cadmium and breast cancer risk. Aging. 2010;2:804–14.
Adams SV, Newcomb PA, White E. Dietary cadmium and risk of invasive postmenopausal breast cancer in the VITAL cohort. Cancer Causes Control. 2012;23:845–54.
Itoh H, Iwasaki M, Sawada N, Takachi R, Kasuga Y, Yokoyama S, et al. Dietary cadmium intake and breast cancer risk in Japanese women: a case-control study. Int J Hyg Environ Health. 2014;217:70–7.
Strumylaite L, Kregzdyte R, Bogusevicius A, Poskiene L, Baranauskiene D, Pranys D. Association between cadmium and breast cancer risk according to estrogen receptor and human epidermal growth factor receptor 2: epidemiological evidence. Breast Cancer Res Treat. 2014;145:225–32.
Adams SV, Shafer MM, Bonner MR, LaCroix AZ, Manson JE, Meliker JR, et al. Urinary cadmium and risk of invasive breast cancer in the women’s health initiative. Am J Epidemiol. 2016;183:815–23.
Lin J, Zhang F, Lei Y. Dietary intake and urinary levels of cadmium and breast cancer risk: a meta-analysis. Cancer Epidemiol. 2016;42:101–7.
Eriksen KT, McElroy JA, Harrington JM, Levine KE, Pedersen C, Sorensen M, et al. Urinary cadmium and breast cancer: a prospective Danish cohort study. J Natl Cancer Inst. 2017;109:djw204.
Gaudet MM, Deubler EL, Kelly RS, Ryan Diver W, Teras LR, Hodge JM, et al. Blood levels of cadmium and lead in relation to breast cancer risk in three prospective cohorts. Int J Cancer. 2019;144:1010–6.
White AJ, O’Brien KM, Niehoff NM, Carroll R, Sandler DP. Metallic air pollutants and breast cancer risk in a nationwide cohort study. Epidemiology. 2019;30:20–8.
McElroy JA, Shafer MM, Gangnon RE, Crouch LA, Newcomb PA. Urinary lead exposure and breast cancer risk in a population-based case-control study. Cancer Epidemiol Biomark Prev. 2008;17:2311–7.
Centers for Disease Control and Prevention (CDC), National Center for Health Statistics (NCHS). National Health and Nutrition Examination Survey Data. Hyattsville, MD: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention. https://www.cdc.gov/nchs/nhanes/about_nhanes.htm. Accessed 19 October 2017.
Sugita M. The biological half-time of heavy metals. Int Arch Occup Environ Health. 1978;41:25–40.
Gulson BL, Mizon KJ, Korsch MJ, Howarth D, Phillips A, Hall J. Impact on blood lead in children and adults following relocation from their source of exposure and contribution of skeletal tissue to blood lead. Bull Environ Contam Toxicol. 1996;56:543–50.
Centers for Disease Control and Prevention (CDC). Laboratory procedure manual (Method No: ITB001A). https://wwwn.cdc.gov/nchs/data/nhanes/2003-2004/labmethods/l06_c_met_pb_cd_hg.pdf. Accessed 19 October 2017.
Centers for Disease Control and Prevention (CDC). National Health and Nutrition Examination Survey: specifying weighting parameters. http://www.cdc.gov/nchs/tutorials/nhanes/surveydesign/Weighting/intro.htm. Accessed 7 November 2017.
White AJ, Weinberg CR, O’Meara ES, Sandler DP, Sprague BL. Airborne metals and polycyclic aromatic hydrocarbons in relation to mammographic breast density. Breast Cancer Res. 2019;21:24.
Ali I, Engstrom A, Vahter M, Skerfving S, Lundh T, Lidfeldt J, et al. Associations between cadmium exposure and circulating levels of sex hormones in postmenopausal women. Environ Res. 2014;134:265–9.
Nagata C, Konishi K, Goto Y, Tamura T, Wada K, Hayashi M, et al. Associations of urinary cadmium with circulating sex hormone levels in pre- and postmenopausal Japanese women. Environ Res. 2016;150:82–7.
Swarup D, Naresh R, Varshney VP, Balagangatharathilagar M, Kumar P, Nandi D, et al. Changes in plasma hormones profile and liver function in cows naturally exposed to lead and cadmium around different industrial areas. Res Vet Sci. 2007;82:16–21.
Chang SH, Cheng BH, Lee SL, Chuang HY, Yang CY, Sung FC, et al. Low blood lead concentration in association with infertility in women. Environ Res. 2006;101:380–6.
Pollack AZ, Schisterman EF, Goldman LR, Mumford SL, Albert PS, Jones RL, et al. Cadmium, lead, and mercury in relation to reproductive hormones and anovulation in premenopausal women. Environ Health Perspect. 2011;119:1156–61.
Krieg EF, Feng HA. The relationship between blood lead levels and serum follicle stimulating hormone and luteinizing hormone in the National Health and Nutrition Examination Survey 1999-2002. Reprod Toxicol. 2011;32:277–85.
Alatise OI, Schrauzer GN. Lead exposure: a contributing cause of the current breast cancer epidemic in Nigerian women. Biol Trace Elem Res. 2010;136:127–39.
Mohammadi M, Riyahi Bakhtiari A, Khodabandeh S. Concentration of Cd, Pb, Hg, and Se in different parts of human breast cancer tissues. J Toxicol. 2014;2014:413870.
Rzymski P, Rzymski P, Tomczyk K, Niedzielski P, Jakubowski K, Poniedzialek B, et al. Metal status in human endometrium: relation to cigarette smoking and histological lesions. Environ Res. 2014;132:328–33.
Garcia-Leston J, Mendez J, Pasaro E, Laffon B. Genotoxic effects of lead: an updated review. Environ Int. 2010;36:623–36.
Schrauzer GN. Effects of selenium and low levels of lead on mammary tumor development and growth in MMTV-infected female mice. Biol Trace Elem Res. 2008;125:268–75.
Hayes RB. The carcinogenicity of metals in humans. Cancer Causes Control. 1997;8:371–85.
Bushnik T, Haines D, Levallois P, Levesque J, Van Oostdam J, Viau C. Lead and bisphenol A concentrations in the Canadian population. Health Rep. 2010;21:7–18.
Vahter M, Akesson A, Liden C, Ceccatelli S, Berglund M. Gender differences in the disposition and toxicity of metals. Environ Res. 2007;104:85–95.
Barbosa F, Tanus-Santos JE, Gerlach RF, Parsons PJ. A critical review of biomarkers used for monitoring human exposure to lead: advantages, limitations, and future needs. Environ Health Perspect. 2005;113:1669–74.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wei, Y., Zhu, J. Blood levels of endocrine-disrupting metals and prevalent breast cancer among US women. Med Oncol 37, 1 (2020). https://doi.org/10.1007/s12032-019-1328-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-019-1328-3