Abstract
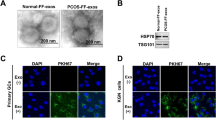
Small RNA sequences in follicular fluid (FF)-derived exosomes (extracellular vesicles contain proteins, DNA, and RNA) vitally function in the development of polycystic ovary syndrome (PCOS). It has been identified that microRNA (miR)-18b-5p is one of miRs that differ between control and PCOS women that passed the false discovery rate, and phosphatase and tensin homolog deleted on chromosome 10 (PTEN) is an important modifier of biological functions of ovarian granulosa cells (GCs) in PCOS. However, whether miR-18b-5p could functionally mediate the progression of PCOS via PTEN was not clarified completely, which was the issue we wanted to solve in our research. FF-derived exosomes were isolated using an extraction kit. KGN cells were co-cultured with miR-18b-5p-modified exosomes or transfected with a PTEN-related vector. After treatment, cell proliferation and apoptosis were observed. A rat model of PCOS was established by letrozole and then injected with miR-18b-5p-modified exosomes. Then, serum follicle-stimulating hormone (FSH), luteinizing hormone (LH), testosterone, and estradiol (E2) levels in PCOS rats were measured. miR-18b-5p, PTEN, and phosphatidylinositol 3 kinases/protein kinase B/mammalian target of rapamycin (PI3K/Akt/mTOR) pathway-related genes were tested. In PCOS patients, miR-18b-5p was downregulated, and PTEN was highly expressed in FF and GCs. PTEN knockdown increased KGN cell proliferation and limited apoptosis. FF-derived exosomes stimulated proliferation and suppressed apoptosis of KGN cells; decreased FSH, LH, and testosterone; and increased E2 in PCOS rats. Upregulating miR-18b-5p further enhanced the inhibitory effects of exosomes on suppressing the progression of PCOS. miR-18b-5p targeted PTEN and could activate PI3K/Akt/mTOR pathway. miR-18b-5p produced by FF-derived exosomes reduces PTEN expression and promotes the activation of the PI3K/Akt/mTOR signaling pathway to improve PCOS. Based on that, circulating miR-18b-5p levels can contribute to the progression of PCOS complications.







Similar content being viewed by others
Availability of Data and Material
Not applicable.
References
Mu Y et al (2020) Vitamin D and polycystic ovary syndrome: a narrative review. Reprod Sci
He FF, Li YM (2020) Role of gut microbiota in the development of insulin resistance and the mechanism underlying polycystic ovary syndrome: a review. J Ovarian Res 13(1):73
Radwan RA et al (2019) Decreased serum level of gamma-amino butyric acid in Egyptian infertile females with polycystic ovary syndrome is correlated with dyslipidemia, total testosterone and 25(OH) vitamin D levels. J Med Biochem 38(4):512–518
Sanchez-Garrido MA, Tena-Sempere M (2020) Metabolic dysfunction in polycystic ovary syndrome: pathogenic role of androgen excess and potential therapeutic strategies. Mol Metab 35:100937
Heshmati J et al (2020) The effect of cinnamon supplementation on glycemic control in women with polycystic ovary syndrome: a systematic review and meta-analysis. J Food Biochem e13543
Louwers YV, Laven JSE (2020) Characteristics of polycystic ovary syndrome throughout life. Ther Adv Reprod Health 14:2633494120911038
Liu M, Murthi S, Poretsky L (2020) Polycystic ovary syndrome and gender identity. Yale J Biol Med 93(4):529–537
Liu Y et al (2020) Extracellular vesicles: recent developments in aging and reproductive diseases. Front Cell Dev Biol 8:577084
Hu J et al (2020) The expression of small RNAs in exosomes of follicular fluid altered in human polycystic ovarian syndrome. PeerJ 8:e8640
Li H et al (2020) S100–A9 protein in exosomes derived from follicular fluid promotes inflammation via activation of NF-kappaB pathway in polycystic ovary syndrome. J Cell Mol Med 24(1):114–125
Xue Y et al (2018) Identification of microRNAs and genes associated with hyperandrogenism in the follicular fluid of women with polycystic ovary syndrome. J Cell Biochem 119(5):3913–3921
Chen B et al (2019) The role of MiRNA in polycystic ovary syndrome (PCOS). Gene 706:91–96
Butler AE et al (2020) Increased microRNA levels in women with polycystic ovarian syndrome but without insulin resistance: a pilot prospective study. Front Endocrinol (Lausanne) 11:571357
Han X et al (2017) Upregulation of microRNA-18b induces phosphatase and tensin homolog to accelerate the migration and invasion abilities of ovarian cancer. Oncol Lett 14(5):5631–5637
Shafiee MN et al (2016) Up-regulation of genes involved in the insulin signalling pathway (IGF1, PTEN and IGFBP1) in the endometrium may link polycystic ovarian syndrome and endometrial cancer. Mol Cell Endocrinol 424:94–101
Hopkins BD et al (2014) PTEN function: the long and the short of it. Trends Biochem Sci 39(4):183–190
Matrine derivative WM130 inhibits hepatocellular carcinoma by suppressing EGFR/ERK/MMP-2 and PTEN/AKT signaling pathways[J]. Cancer Letters, 2015, 368(1):126–134.
Yang M et al (2020) Bone marrow mesenchymal stem cell-derived exosomal miR-144-5p improves rat ovarian function after chemotherapy-induced ovarian failure by targeting PTEN. Lab Invest 100(3):342–352
Andreas E et al (2016) MicroRNA 17–92 cluster regulates proliferation and differentiation of bovine granulosa cells by targeting PTEN and BMPR2 genes. Cell Tissue Res 366(1):219–230
Abuelezz NZ et al (2020) Nanocurcumin alleviates insulin resistance and pancreatic deficits in polycystic ovary syndrome rats: insights on PI3K/AkT/mTOR and TNF-alpha modulations. Life Sci 256:118003
Liu J et al (2018) The role of mTOR in ovarian neoplasms, polycystic ovary syndrome, and ovarian aging. Clin Anat 31(6):891–898
Cai Z et al (2020) Plumbagin inhibits proliferation and promotes apoptosis of ovarian granulosa cells in polycystic ovary syndrome by inactivating PI3K/Akt/mTOR pathway. Anim Cells Syst (Seoul) 24(4):197–204
Song WJ et al (2018) Akt-mTOR signaling mediates abnormalities in the proliferation and apoptosis of ovarian granulosa cells in patients with polycystic ovary syndrome. Gynecol Obstet Invest 83(2):124–132
Huang X et al (2020) Depletion of exosomal circLDLR in follicle fluid derepresses miR-1294 function and inhibits estradiol production via CYP19A1 in polycystic ovary syndrome. Aging (Albany NY) 12(15):15414–15435
Ayuk SM, Abrahamse H, Houreld NN (2016) The role of photobiomodulation on gene expression of cell adhesion molecules in diabetic wounded fibroblasts in vitro. J Photochem Photobiol B 161:368–374
He T et al (2019) MicroRNA-200b and microRNA-200c are up-regulated in PCOS granulosa cell and inhibit KGN cell proliferation via targeting PTEN. Reprod Biol Endocrinol 17(1):68
Zhao Y et al (2019) Mesenchymal stem cells derived exosomal miR-323-3p promotes proliferation and inhibits apoptosis of cumulus cells in polycystic ovary syndrome (PCOS). Artif Cells Nanomed Biotechnol 47(1):3804–3813
Yang S et al (2020) miRNA-181a-5p enhances the sensitivity of cells to cisplatin in esophageal adenocarcinoma by targeting CBLB. Cancer Manag Res 12:4981–4990
Lee R et al (2020) A review of the impact of bariatric surgery in women with polycystic ovary syndrome. Cureus 12(10):e10811
Inoue Y et al (2020) Prediction of major microRNAs in follicular fluid regulating porcine oocyte development. J Assist Reprod Genet 37(10):2569–2579
Rodrigues TA et al (2019) Follicular fluid exosomes act on the bovine oocyte to improve oocyte competence to support development and survival to heat shock. Reprod Fertil Dev 31(5):888–897
Hung WT et al (2017) Stage-specific follicular extracellular vesicle uptake and regulation of bovine granulosa cell proliferation. Biol Reprod 97(4):644–655
Machtinger R et al (2017) Extracellular microRNAs in follicular fluid and their potential association with oocyte fertilization and embryo quality: an exploratory study. J Assist Reprod Genet 34(4):525–533
Wang S et al (2017) Expression and role of microRNA 18b and hypoxia inducible factor-1alpha in placental tissues of preeclampsia patients. Exp Ther Med 14(5):4554–4560
Sorensen SS et al (2014) miRNA expression profiles in cerebrospinal fluid and blood of patients with acute ischemic stroke. Transl Stroke Res 5(6):711–718
Xue F et al (2020) Non-coding RNA LOXL1-AS1 exhibits oncogenic activity in ovarian cancer via regulation of miR-18b-5p/VMA21 axis. Biomed Pharmacother 125:109568
Li H et al (2018) The oncoprotein HBXIP promotes human breast cancer growth through down-regulating p53 via miR-18b/MDM2 and pAKT/MDM2 pathways. Acta Pharmacol Sin 39(11):1787–1796
Xue M et al (2020) lncRNA ZFPM2-AS1 promotes proliferation via miR-18b-5p/VMA21 axis in lung adenocarcinoma. J Cell Biochem 121(1):313–321
Ouyang JX et al (2013) RNA interference mediated pten knock-down inhibit the formation of polycystic ovary. Mol Cell Biochem 380(1–2):195–202
Chen MJ et al (2015) The effect of androgens on ovarian follicle maturation: dihydrotestosterone suppress FSH-stimulated granulosa cell proliferation by upregulating PPARgamma-dependent PTEN expression. Sci Rep 5:18319
Ernst EH, Lykke-Hartmann K (2018) Transcripts encoding free radical scavengers in human granulosa cells from primordial and primary ovarian follicles. J Assist Reprod Genet 35(10):1787–1798
Thompson PA et al (2020) Targeting oncogene mRNA translation in B cell malignancies with eFT226, a potent and selective inhibitor of eIF4A1. Mol Cancer Ther
Xing D, Fadare O (2020) Molecular events in the pathogenesis of vulvar squamous cell carcinoma. Semin Diagn Pathol
Zhang N et al (2020) Berberine decreases insulin resistance in a PCOS rats by improving GLUT4: dual regulation of the PI3K/AKT and MAPK pathways. Regul Toxicol Pharmacol 110:104544
Mao Z et al (2018) C1QTNF3 in the murine ovary and its function in folliculogenesis. Reproduction 155(4):333–346
Acknowledgements
We would like to acknowledge the reviewers for their helpful comments on this paper.
Funding
This work was supported by the Science and Technology Department of Hainan Province (2019RC389, ZDYF2017086, ZDYF2019157).
Author information
Authors and Affiliations
Contributions
Weiying Lu contributed to study design; Zhi Zhou and Zhihua Tu contributed to manuscript editing; Juan Zhang, Can tan, Xiaoyong Shen, Bangbei Wan, and Yejuan Li contributed to experimental studies; Anguo Wang, Liqiang Zhao, Jiajia Hu, Ning Ma, Jing Zhou, Lin Chen, and Yanqin Song contributed to data analysis.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
Experimental approval was acquired from ethics committee of Hainan Women and Children’s Medical Center. Written informed consent was collected from all participants. The animal experiment was approved by the animal ethics committee of Hainan Women and Children’s Medical Center.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Zhi Zhou and Zhihua Tu are co-first authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhou, Z., Tu, Z., Zhang, J. et al. Follicular Fluid-Derived Exosomal MicroRNA-18b-5p Regulates PTEN-Mediated PI3K/Akt/mTOR Signaling Pathway to Inhibit Polycystic Ovary Syndrome Development. Mol Neurobiol 59, 2520–2531 (2022). https://doi.org/10.1007/s12035-021-02714-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-021-02714-1