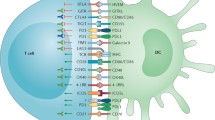
Abstract
Cancer immunotherapy with antibodies against immune checkpoints has made impressive advances in the last several years. The most relevant drugs target programmed cell death 1 (PD-1) expressed on T cells or its ligand, the programmed cell death ligand 1 (PD-L1), expressed on cancer cells, and cytotoxic T lymphocyte-associated protein 4 (CTLA-4). Unfortunately, cancer patients with HIV infection are usually excluded from cancer clinical trials, because there are concerns about the safety and the anti-tumoral activity of these novel therapies in patients with HIV infection. Several retrospective studies and some case reports now support the notion that antibodies against immune checkpoints are safe and active in cancer patients with HIV infection, but prospective data in these patients are lacking. In addition, signs of antiviral activity with increase in CD4 T cell counts, plasma viremia reduction or decrease in the viral reservoir have been reported in some of the patients treated, although no patient achieved a complete clearance of the viral reservoir. Here we briefly summarize all clinical cases reported in the literature, as well as ongoing clinical trials testing novel immunotherapy drugs in cancer patients with HIV infection.
Similar content being viewed by others
References
Barre-Sinoussi F, Chermann JC, Rey F, Nugeyre MT, Chamaret S, Gruest J, et al. Isolation of a T-lymphotropic retrovirus from a patient at risk for acquired immune deficiency syndrome (AIDS). Science. 1983;220(4599):868–71.
Klatzmann D, Barre-Sinoussi F, Nugeyre MT, Danquet C, Vilmer E, Griscelli C, et al. Selective tropism of lymphadenopathy associated virus (LAV) for helper-inducer T lymphocytes. Science. 1984;225(4657):59–63.
Arts EJ, Hazuda DJ. HIV-1 antiretroviral drug therapy. Cold Spring Harb Perspect Med. 2012;2(4):a007161.
Chereshnev VA, Bocharov G, Bazhan S, Bachmetyev B, Gainova I, Likhoshvai V, et al. Pathogenesis and treatment of HIV infection: the cellular, the immune system and the neuroendocrine systems perspective. Int Rev Immunol. 2013;32(3):282–306.
Deeks SG, Autran B, Berkhout B, Benkirane M, Cairns S, Chomont N, et al. Towards an HIV cure: a global scientific strategy. Nat Rev Immunol. 2012;12(8):607–14.
Katlama C, Deeks SG, Autran B, Martinez-Picado J, van Lunzen J, Rouzioux C, et al. Barriers to a cure for HIV: new ways to target and eradicate HIV-1 reservoirs. Lancet. 2013;381(9883):2109–17.
Finzi D, Blankson J, Siliciano JD, Margolick JB, Chadwick K, Pierson T, et al. Latent infection of CD4 + T cells provides a mechanism for lifelong persistence of HIV-1, even in patients on effective combination therapy. Nat Med. 1999;5(5):512–7.
Siliciano JD, Kajdas J, Finzi D, Quinn TC, Chadwick K, Margolick JB, et al. Long-term follow-up studies confirm the stability of the latent reservoir for HIV-1 in resting CD4+ T cells. Nat Med. 2003;9(6):727–8.
Martinez-Picado J, Deeks SG. Persistent HIV-1 replication during antiretroviral therapy. Curr Opin HIV AIDS. 2016;11(4):417–23.
Bonnet F, Burty C, Lewden C, Costagliola D, May T, Bouteloup V, et al. Changes in cancer mortality among HIV-infected patients: the Mortalite 2005 Survey. Clin Infect Dis. 2009;48(5):633–9.
Vandenhende MA, Roussillon C, Henard S, Morlat P, Oksenhendler E, Aumaitre H, et al. Cancer-related causes of death among HIV-infected patients in France in 2010: evolution since 2000. PLoS One. 2015;10(6):e0129550.
Suneja G, Boyer M, Yehia BR, Shiels MS, Engels EA, Bekelman JE, et al. Cancer treatment in patients with HIV infection and non-AIDS-defining cancers: a survey of US oncologists. J Oncol Pract Am Soc Clin Oncol. 2015;11(3):e380–7.
Suneja G, Lin CC, Simard EP, Han X, Engels EA, Jemal A. Disparities in cancer treatment among patients infected with the human immunodeficiency virus. Cancer. 2016;122(15):2399–407.
Suneja G, Shiels MS, Angulo R, Copeland GE, Gonsalves L, Hakenewerth AM, et al. Cancer treatment disparities in HIV-infected individuals in the United States. J Clin Oncol. 2014;32(22):2344–50.
Le Garff G, Samri A, Lambert-Niclot S, Even S, Lavole A, Cadranel J, et al. Transient HIV-specific T cells increase and inflammation in an HIV-infected patient treated with nivolumab. AIDS. 2017;31(7):1048–51.
Ribas A, Kefford R, Marshall MA, Punt CJ, Haanen JB, Marmol M, et al. Phase III randomized clinical trial comparing tremelimumab with standard-of-care chemotherapy in patients with advanced melanoma. J Clin Oncol. 2013;31(5):616–22.
Postow MA, Chesney J, Pavlick AC, Robert C, Grossmann K, McDermott D, et al. Nivolumab and ipilimumab versus ipilimumab in untreated melanoma. N Engl J Med. 2015;372(21):2006–17.
Inman BA, Longo TA, Ramalingam S, Harrison MR. Atezolizumab: a PD-L1-blocking antibody for bladder cancer. Clin Cancer Res. 2017;23(8):1886–90.
Schadendorf D, Hodi FS, Robert C, Weber JS, Margolin K, Hamid O, et al. Pooled analysis of long-term survival data from phase II and phase III trials of ipilimumab in unresectable or metastatic melanoma. J Clin Oncol. 2015;33(17):1889–94.
Ben-Aharon O, Magnezi R, Leshno M, Goldstein DA. Association of Immunotherapy with durable survival as defined by value frameworks for cancer care. JAMA Oncol. 2018;4(3):326–32.
Robert C, Long GV, Brady B, Dutriaux C, Maio M, Mortier L, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med. 2015;372(4):320–30.
Wherry EJ, Kurachi M. Molecular and cellular insights into T cell exhaustion. Nat Rev Immunol. 2015;15(8):486–99.
Moskophidis D, Lechner F, Pircher H, Zinkernagel RM. Virus persistence in acutely infected immunocompetent mice by exhaustion of antiviral cytotoxic effector T cells. Nature. 1993;362(6422):758–61.
Barber DL, Wherry EJ, Masopust D, Zhu B, Allison JP, Sharpe AH, et al. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature. 2006;439(7077):682–7.
Shiels MS, Islam JY, Rosenberg PS, Hall HI, Jacobson E, Engels EA. Projected cancer incidence rates and burden of incident cancer cases in HIV-infected adults in the United States through 2030. Ann Intern Med. 2018;168(12):866–73.
Robbins HA, Pfeiffer RM, Shiels MS, Li J, Hall HI, Engels EA. Excess cancers among HIV-infected people in the United States. J Natl Cancer Inst. 2015. https://doi.org/10.1093/jnci/dju503.
Robert C, Schachter J, Long GV, Arance A, Grob JJ, Mortier L, et al. Pembrolizumab versus ipilimumab in advanced melanoma. N Engl J Med. 2015;372(26):2521–32.
Reck M, Rodriguez-Abreu D, Robinson AG, Hui R, Csoszi T, Fulop A, et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med. 2016;375(19):1823–33.
Herbst RS, Baas P, Kim DW, Felip E, Perez-Gracia JL, Han JY, et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet. 2016;387(10027):1540–50.
Nghiem PT, Bhatia S, Lipson EJ, Kudchadkar RR, Miller NJ, Annamalai L, et al. PD-1 blockade with pembrolizumab in advanced merkel-cell carcinoma. N Engl J Med. 2016;374(26):2542–52.
D’Angelo SP, Russell J, Lebbé C, Chmielowski B, Gambichler T, Grob JJ, et al. Efficacy and safety of first-line avelumab treatment in patients with stage IV metastatic merkel cell carcinoma: a preplanned interim analysis of a clinical trial. JAMA Oncol. 2018;4(9):e180077.
Killock D. Haematological cancer: anti-PD-1 therapy with nivolumab after allo-HSCT for Hodgkin lymphoma. Nat Rev Clin Oncol. 2017;14(5):264.
Brower V. Pembrolizumab in advanced head and neck cancer. Lancet Oncol. 2017;18(5):e248.
Motzer RJ, Tannir NM, McDermott DF, Aren Frontera O, Melichar B, Choueiri TK, et al. Nivolumab plus ipilimumab versus sunitinib in advanced renal-cell carcinoma. N Engl J Med. 2018;378(14):1277–90.
Sidaway P. Bladder cancer: pembrolizumab is superior to chemotherapy. Nat Rev Urol. 2017;14(5):261.
Dudley JC, Lin MT, Le DT, Eshleman JR. Microsatellite instability as a biomarker for PD-1 blockade. Clin Cancer Res. 2016;22(4):813–20.
Gay CL, Bosch RJ, Ritz J, Hataye JM, Aga E, Tressler RL, et al. Clinical trial of the anti-PD-L1 antibody BMS-936559 in HIV-1 infected participants on suppressive antiretroviral therapy. J Infect Dis. 2017;215(11):1725–33.
Day CL, Kaufmann DE, Kiepiela P, Brown JA, Moodley ES, Reddy S, et al. PD-1 expression on HIV-specific T cells is associated with T-cell exhaustion and disease progression. Nature. 2006;443(7109):350–4.
Velu V, Titanji K, Zhu B, Husain S, Pladevega A, Lai L, et al. Enhancing SIV-specific immunity in vivo by PD-1 blockade. Nature. 2009;458(7235):206–10.
Strain MC, Lada SM, Luong T, Rought SE, Gianella S, Terry VH, et al. Highly precise measurement of HIV DNA by droplet digital PCR. PLoS One. 2013;8(4):e55943.
Guihot A, Marcelin AG, Massiani MA, Samri A, Soulie C, Autran B, et al. Drastic decrease of the HIV reservoir in a patient treated with nivolumab for lung cancer. Ann Oncol. 2018;29(2):517–8.
Scully EP, Rutishauser RL, Simoneau CR, Delagreverie H, Euler Z, Thanh C, et al. Inconsistent HIV reservoir dynamics and immune responses following anti-PD-1 therapy in cancer patients with HIV infection. Ann Oncol. 2018;29(10):2141–2.
Uldrick TS, Gonçalves PH, Fling S, Emu B, Ernstoff MS, Kaiser J, et al. Interim safety analysis of CITN- 12: pembrolizumab in patients with HIV and cancer. CROI Meeting. 2018;March:4–7.
Wightman F, Solomon A, Kumar SS, Urriola N, Gallagher K, Hiener B, et al. Effect of ipilimumab on the HIV reservoir in an HIV-infected individual with metastatic melanoma. AIDS. 2015;29(4):504–6.
Heppt MV, Schlaak M, Eigentler TK, Kahler KC, Kiecker F, Loquai C, et al. Checkpoint blockade for metastatic melanoma and Merkel cell carcinoma in HIV-positive patients. Ann Oncol. 2017;28(12):3104–6.
Lavole A, Guihot A, Veyri M, Lambotte O, Autran B, Cloarec N, et al. PD-1 blockade in HIV-infected patients with lung cancer: a new challenge or already a strategy? Ann Oncol. 2018;29(4):1065–6.
Davar D, Wilson M, Pruckner C, Kirkwood JM. PD-1 blockade in advanced melanoma in patients with hepatitis C and/or HIV. Case Rep Oncol Med. 2015;2015:737389.
Pinato DJ, Kythreotou A, Mauri FA, Suardi E, Allara E, Shiner RJ, et al. Functional immune characterization of HIV-associated non-small-cell lung cancer. Ann Oncol. 2018;29(6):1486–8.
Gonzalez-Cao M, Martinez-Picado J, Pulla MP, Clotet B, Juan O, Dalmau J, et al. A phase II exploratory study of durvalumab (MEDI4736) in HIV-1 patients with advanced solid tumors. Ann Oncol. 2017;28(suppl_5):v403–27.
Peligero C, Argilaguet J, Guerri-Fernandez R, Torres B, Ligero C, Colomer P, et al. PD-L1 blockade differentially impacts regulatory T cells from HIV-infected individuals depending on plasma viremia. PLoS Pathog. 2015;11(12):e1005270.
Acknowledgements
A.M. is supported by a grant from the Spanish Ministry of Economy, Industry and Competitiveness and FEDER Grant no. SAF2016-75505-R (AEI/MINEICO/FEDER, UE), and the “María de Maeztu” Programme for Units of Excellence in R&D (MDM-2014-0370). J.M-P’ team is supported by the Spanish Secretariat for Research through Grants SAF2016-80033-R and RTC-2016-5324-1, by the Foundation for AIDS Research amfAR (109858-64-RSRL), as well as by non-restricted grants from Merck, AstraZeneca, Gilead, and ViiV Healthcare. Work in the Dr. Rosell laboratory is partially supported by a Grant from La Caixa Foundation, and by a European Grant (ELBA no. 765492).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Research grants from funding agencies are commented in the Acknowledgement section. DURVAST trial (Sponsor: Spanish Lung Cancer Group; Coordinator: Dra Gonzalez Cao) has the financial support from Astra Zeneca (Spain).
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
For this type of study informed consent is not required.
Rights and permissions
About this article
Cite this article
Gonzalez-Cao, M., Martinez-Picado, J., Karachaliou, N. et al. Cancer immunotherapy of patients with HIV infection. Clin Transl Oncol 21, 713–720 (2019). https://doi.org/10.1007/s12094-018-1981-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-018-1981-6