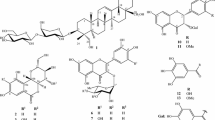
Abstract
A new terthiophene, 3′-hydroxy-2,2′:5′,2″-terthiophene-3′-O-β-d-glucopyranoside (1) and a new oleanane-type saponin, echinocystic acid-3-O-(6-O-acetyl)-β-d-glucopyranoside (7) were isolated from the aerial parts of Eclipta prostrata L. Moreover, five thiophenes (2–6), seven triterpenoids (8–14), two coumestans (15 and 16), and four flavonoids (17–20) having previously known chemical structures were isolated during the same course of this study. All the isolates 1–20 were evaluated for their cytotoxicity against human ovarian cancer cells (SKOV3) using MTT assays.


Similar content being viewed by others
References
Anhut, S., H.D. Zinsmeister, R. Mues, W. Barz, K. Mackenbrock, J. Koster, and K.R. Markham. 1984. The first identification of isoflavones from a bryophyte. Phytochemistry 23: 1073–1075.
Bittner, M., J. Jakupovic, F. Bohlmann, and M. Silva. 1988. 5-methylcoumarins from Nassauvia species. Phytochemistry 27: 3845–3847.
Bohlmann, F., and C. Zdero. 1976. Polyacetylenic compounds. 238: On the constituents of the genus Dyssodia. Chemische Berichte 109: 901–905.
Brinkmeier, E., H. Geiger, and H.D. Zinsmeister. 1999. Biflavonoids and 4,2′-epoxy-3-phenylcoumarins from the moss Mnium hornum. Phytochemistry 52: 297–302.
Chan, G.F.Q., G.H.N. Towers, and J.C. Mitchell. 1975. Ultraviolet-mediated antibiotic activity of thiophene compounds of Tagetes. Phytochemistry 14: 2295–2296.
Eachern, A.M., C. Soucy, L.C. Leitch, J.T. Arnason, and P. Morand. 1998. Synthesis and characterization of alkyl-, halo-, and hetero- substituted derivatives of the potent phototoxin α-terthienyl. Tetrahedron 44: 2403–2412.
Feng, X., Z. Zou, S. Fu, L. Sun, Z. Su, and D.A. Sun. 2010. Microbial oxidation and glucosidation of echinocystic acid by Nocardia coralline. Journal of Molecular Catalysis B 66: 219–223.
Han, X.H., S.S. Hong, J.S. Hwang, M.K. Lee, B.Y. Hwang, and J.S. Ro. 2007. Monoamine oxidase inhibitory components from Cayratia japonica. Archives of Pharmacal Research 30: 13–17.
Hudson, J.B., R.J. Marles, C. Soucy-Breau, L. Harris, and J.T. Arnason. 1994. Photoactive terthiophenes: The influence of serum on anti-HIV (human immunodeficiency virus) activites. Photochemistry and Photobiology 60: 591–593.
Kim, D.S., C.L. Ashendel, Q. Zhou, C.-T. Chang, E.-S. Lee, and C.-J. Chang. 1998. Novel protein kinase C inhibitors: Alpha-terthiophene derivatives. Bioorganic & Medicinal Chemistry Letters 8: 2695–2698.
Kobori, M., Z. Yang, D. Gong, V. Heissmeyer, H. Zhu, Y.K. Jung, M.A. Gakidis, A. Rao, T. Sekine, F. Ikegami, C. Yuan, and J. Yuan. 2004. Wedelolactone suppresses LPS-induced caspase-11 expression by directly inhibiting the IKK complex. Cell Death and Differentiation 11: 123–130.
Kumari, C.S., S. Govindasamy, and E. Sukumar. 2006. Lipid lowering activity of Eclipta prostrata in experimental hyperlipidemia. Journal of Ethnopharmacology 105: 332–335.
Lee, M.K., N.R. Ha, H. Yang, S.H. Sung, G.H. Kim, and Y.C. Kim. 2008. Antiproliferative activity of triterpenoids from Eclipta prostrata on hepatic stellate cells. Phytomedicine 15: 775–780.
Lirdprapamongkol, K., J.P. Kramb, D. Chokchaichamnankit, C. Srisomsap, R. Surarit, M. Sila-Asna, A. Bunyaratvej, G. Dannhardt, and J. Svasti. 2008. Juice of Eclipta prostrata inhibits cell migration in vitro and exhibits anti-angiogenic activity in vivo. In Vivo 22: 363–368.
Liu, Q.M., H.Y. Zhao, X.K. Zhong, and J.G. Jiang. 2012. Eclipta prostrata L. phytochemicals: Isolation, structure elucidation, and their antitumor activity. Food and Chemical Toxicology 50: 4016–4022.
Liu, X., Y. Jiang, Y. Zhao, and H. Tang. 2000. Effect of ethyl acetate extract of Eclipta prostrata on mice of normal and immunosuppression. Zhong Yao Cai 23: 407–409.
Liu, X., Y. Zhao, Y. Jiang, and H. Tang. 2001. Effect of ethylacetate extract of Eclipta prostrata on function of T-lymphocyte. Disi Junyi Daxue Xuebao 22: 754–756.
Luo, Z.G., Z.Y. Liu, and Z.H. Yang. 2014. The synthesis and photoactivated cytotoxicity of novel 5-phenyl-3-(2,2′:5′,2″-terthien-5-yl)-4,5-dihydro-1H-pyrazolines. Chinese Chemical Letters 25: 333–336.
Melo, P.A., M.C. do Nascimento, W.B. Mors, and G. Suarez-Kurtz. 1994. Inhibition of the myotoxic and hemorrhagic activities of crotalid venoms by Eclipta prostrata (Asteraceae) extracts and constituents. Toxicon 32: 595–603.
Roberts, D.W., D.R. Doerge, M.I. Churchwell, G.G. da Costa, M.M. Marques, and W.H. Tolleson. 2004. Inhibition of extrahepatic human cytochromes P450 1A1 and 1B1 by metabolism of isoflavones found in Trifolium pretense (red clover). Journal of Agricultural and Food Chemistry 52: 6623–6632.
Takahashi, H.T., C.R. Novello, T. Ueda-nakamura, B.P.D. Filho, J.C.P. de Mello, and C.V. Nakamura. 2011. Thiophene derivatives with antileishmanial activity isolated from aerial parts of Porophyllum ruderale (jacq.) Cass. Molecules 16: 3469–3478.
Tewtrakul, S., S. Subhadhirasakul, P. Tansakul, S. Cheenpracha, and C. Karalai. 2011. Antiinflammatory constituents from Eclipta prostrata using RAW264.7 macrophage cells. Phytotherapy Research 25: 1313–1316.
Tewtrakul, S., S. Subhadhirasakul, S. Cheenpracha, and C. Karalai. 2007. HIV-1 protease and HIV-1 integrase inhibitory substances from Eclipta prostrata. Phytotherapy Research 21: 1092–1095.
Wei, Y., K. Zhang, L. Yin, J. Du, and G. Zhang. 2012. Isolation of bioactive components from Flaveria bidentis (L.) Kuntze using high-speed counter-current chromatography and time-controlled collection method. Journal of Separation Science 35: 869–874.
Wiart, C., S. Mogana, S. Khalifah, M. Mahan, S. Ismail, M. Buckle, A.K. Narayana, and M. Sulaiman. 2004. Antimicrobial screening of plants used for traditional medicine in the state of Perak, Peninsular Malaysia. Fitoterapia 75: 68–73.
Yahara, S., N. Ding, and T. Nohara. 1994. Oleanane glycosides from Eclipta alba. Chemical & Pharmaceutical Bulletin 42: 1336–1338.
Yahara, S., N. Ding, T. Nohara, K. Masuda, and H. Ageta. 1997. Taraxastane glycosides from Eclipta alba. Phytochemistry 44: 131–135.
Acknowledgments
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2013R1A1A2004398) and by a grant from the Bio-Synergy Research Project (NRF-2014M3A9C4066594) of the Ministry of Science, ICT and Future Planning through the National Research Foundation. We thank Korea Basic Science Institute (KBSI) for running NMR and MS experiments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, HY., Kim, H.M., Ryu, B. et al. Constituents of the aerial parts of Eclipta prostrata and their cytotoxicity on human ovarian cancer cells in vitro. Arch. Pharm. Res. 38, 1963–1969 (2015). https://doi.org/10.1007/s12272-015-0599-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-015-0599-2