Abstract
Introduction
There has been increased attention on the potential impact of the preservative benzalkonium chloride (BAK) on the ocular surface. This study compared the ocular surface tolerability of once-daily bimatoprost 0.01% and latanoprost 0.005% (both preserved with 0.02% BAK), and travoprost 0.004% preserved with sofZia™.
Methods
A randomized, multicenter (15 sites), investigator-masked study enrolled patients with open-angle glaucoma or ocular hypertension who had received latanoprost monotherapy for at least 1 month. Patients were randomized to oncedaily bimatoprost (n = 56), travoprost (n = 53), or latanoprost (n = 55) monotherapy for 3 months. Follow-up visits were at weeks 1, 4, and 12. The primary outcome measure was physician-graded conjunctival hyperemia (scale 0 to 3) at week 12. Secondary outcomes included corneal staining (scale 0 to 3) and tear break-up time (TBUT).
Results
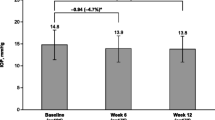
There were no significant differences in mean (standard deviation [SD]) outcome measures including conjunctival hyperemia (bimatoprost: 0.48 [0.52], travoprost: 0.49 [0.52], latanoprost: 0.51 [0.54]), corneal staining (bimatoprost: 0.31 [0.49], travoprost: 0.25 [0.46], latanoprost: 0.24 [0.45]), or TBUT (bimatoprost: 9.7 s [6.1], travoprost: 9.5 s [5.8], latanoprost: 9.8 s [5.0]) among subjects at latanoprost-treated baseline (P ≥ 0.664). At week 12, there were no significant differences in conjunctival hyperemia (bimatoprost: 0.42 [0.48], travoprost: 0.46 [0.44], latanoprost: 0.44 [0.57]), corneal staining (bimatoprost: 0.31 [0.45], travoprost: 0.32 [0.48], latanoprost: 0.22 [0.30]), or TBUT (bimatoprost: 9.7 s [5.7], travoprost 9.7 s [5.0], latanoprost: 9.3 s [4.0]) among the treatment groups (P ≥ 0.379). At week 1, there was a statistically significant among-group difference in mean change from baseline in hyperemia (+0.04, bimatoprost; +0.20, travoprost; 0.00, latanoprost; P = 0.018). There were no statistically significant among-group differences in mean corneal staining, mean TBUT, or change from baseline at any visit.
Conclusions
Despite preservative differences, there were no significant differences in objective clinical measures of ocular surface tolerability after 3 months of treatment with bimatoprost (with 0.02% BAK), travoprost (with sofZia), and latanoprost (with 0.02% BAK).
Article PDF
Similar content being viewed by others
References
Woodward DF, Krauss AH, Chen J, et al. The pharmacology of bimatoprost (Lumigan). Surv Ophthalmol. 2001;45:S337–S345.
Woodward DF, Krauss AH, Chen J, et al. Pharmacological characterization of a novel antiglaucoma agent, Bimatoprost (AGN 192024). J Pharmacol Exp Ther. 2003;305:772–785.
McKee HD, Gupta MS, Ahad MA, Saldaña M, Innes JR. First-choice treatment preferences for primary open-angle glaucoma in the United Kingdom. Eye (Lond). 2005;19:923–924.
Holló G. The side effects of the prostaglandin analogues. Expert Opin Drug Saf. 2007;6:45–52.
Cracknell KP, Grierson I. Prostaglandin analogues in the anterior eye: their pressure lowering action and side effects. Exp Eye Res. 2009;88:786–791.
Abelson MB, Mroz M, Rosner SA, Dirks MS, Hirabayashi D. Multicenter, open-label evaluation of hyperemia associated with use of bimatoprost in adults with open-angle glaucoma or ocular hypertension. Adv Ther. 2003;20:1–13.
Leal BC, Medeiros FA, Medeiros FW, Santo RM, Susanna R, Jr. Conjunctival hyperemia associated with bimatoprost use: a histopathologic study. Am J Ophthalmol. 2004;138:310–313.
Novack GD, Evans R. Commercially available ocular hypotensive products: preservative concentration, stability, storage, and in-life utilization. J Glaucoma. 2001;10:483–486.
Furrer P, Mayer JM, Gurny R. Ocular tolerance of preservatives and alternatives. Eur J Pharm Biopharm. 2002;53:263–280.
Caraccio TR, McGuigan MA. Benzalkonium chloride. In: Dart RC, ed. Medical Toxicology, 3rd ed. New York, NY: Lippincott Williams & Williams; 2004:1255–1257.
Charnock C. Are multidose over-the-counter artificial tears adequately preserved? Cornea. 2006;25:432–437.
Whitson JT, Trattler WB, Matossian C, Williams J, Hollander DA. Ocular surface tolerability of prostaglandin analogs in patients with glaucoma or ocular hypertension. J Ocul Pharmacol Ther. 2010;26:287–292.
Baudouin C, Labbé A, Liang H, Pauly A, Brignole-Baudouin F. Preservatives in eyedrops: the good, the bad and the ugly. Prog Retin Eye Res. 2010;29:312–334.
Schwartz GF, Kotak S, Mardekian J, Fain JM. Incidence of new coding for dry eye and ocular infection in open-angle glaucoma and ocular hypertension patients treated with prostaglandin analogs: retrospective analysis of three medical/pharmacy claims databases. BMC Ophthalmol. 2011;11:14.
Champeau EJ, Edelhauser HF. Effect of ophthalmic preservatives on the ocular surface: conjunctival and corneal uptake and distribution of benzalkonium chloride and chlorhexidine digluconate. In: Holly FJ, ed. The Preocular Tear Film in Health, Disease and Contact Lens Wear. Lubbock, TX: Dry Eye Institute; 1986:292–302.
Mishima S, Maurice DM. The oily layer of the tear film and evaporation from the corneal surface. Exp Eye Res. 1961;1:39–45.
Himebaugh NL, Begley CG, Bradley A, Wilkinson JA. Blinking and tear break-up during four visual tasks. Optom Vis Sci. 2009;86:E106–E114.
Author information
Authors and Affiliations
Corresponding author
Additional information
To view enhanced content go to www.advancesintherapy.com
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Crichton, A.C.S., Vold, S., Williams, J.M. et al. Ocular Surface Tolerability of Prostaglandin Analogs and Prostamides in Patients with Glaucoma or Ocular Hypertension. Adv Therapy 30, 260–270 (2013). https://doi.org/10.1007/s12325-013-0014-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-013-0014-7