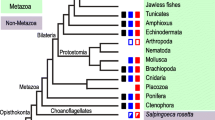
Abstract
Major sperm proteins (MSPs) are a nematode-specific system of cytoskeletal proteins required for amoeboid sperm movement. A number of MSP genes vary in different nematode species, but encoded protein sequences reveal high homology between these proteins. However, all studies of MSP localization and functions to date are based exclusively on the representatives of the order Rhabditida belonging to the nematode class Chromadorea, while MSP-driven sperm movement in Enoplea, another major clade of the phylum Nematoda is still unconfirmed. In this study, we documented the presence of MSPs in the enoplean nematode Enoplus brevis (Bastian, 1865) (Enoplida) and compared MSP localization in sperm of this species with that of the chromadorean nematode Panagrellus redivivus (Linnaeus, 1767) (Rhabditida). Then, we analyzed the putative MSP sequences of both species. Our results indicate that MSPs are presented in E. brevis spermatozoa and form filamentous structures after sperm activation, which may be considered as the evidence of their motor functions similar to those in the spermatozoa of chromadorean nematodes. We found that E. brevis MSPs show lower homology to known proteins of rhabditids whose species exhibit hyper-conservatism in MSP protein sequences. These results reflect the more distant evolutionary relationships between Enoplea and Chromadorea than exist within Rhabditida order. Our data reveal a need to reevaluate current views of MSP evolution within Nematoda.









Similar content being viewed by others
Data availability
Nucleotide sequences coding E. brevis MSP124-1, MSP124-2, and MSP124-3 were deposited to the Third Party Annotation Section of the DDBJ/ENA/GenBank databases under the accession numbers TPA: BK014294-BK014296.
References
Afgan, E., Baker, D., Batut, B., van den Beek, M., Bouvier, D., Cech, M. et al. (2018). The Galaxy platform for accessible, reproducible and collaborative biomedical analyses: 2018 update. Nucleic Acids Research, 46(W1), W537–W544. https://doi.org/10.1093/nar/gky379
Bastian, H. C. (1865). II. Monograph on the anguillulidae or free nematoids marine land and freshwater; with descriptions of 100 new species. Transactions of the Linnean Society of London 25(2), 73-184. https://doi.org/10.1111/j.1096-3642.1865.tb00179.x
Bik, H. M., Lambshead, P. J. D., Thomas, W. K., & Lunt, D. H. (2010). Moving towards a complete molecular framework of the Nematoda: A focus on the Enoplida and early-branching clades. BMC Evolutionary Biology, 10, 353. https://doi.org/10.1186/1471-2148-10-353
Blaxter, M. (2009). Nematodes (Nematoda). In S. B. Hedges & S. Kumar (Eds.), The timetree of life (pp. 247–250). Oxford University Press.
Blaxter, M., & Koutsovoulos, G. (2015). The evolution of parasitism in Nematoda. Parasitology, 142, S26–S39. https://doi.org/10.1017/s0031182014000791
Bolger, A. M., Lohse, M., & Usadel, B. (2014). Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics, 30(15), 2114–2120. https://doi.org/10.1093/bioinformatics/btu170
Chu, D. S., & Shakes, D. C. (2013). Spermatogenesis. Advances in Experimental Medicine and Biology, 757, 171–203. https://doi.org/10.1007/978-1-4614-4015-4_7
De Ley, P., & Blaxter, M. (2002). Systematic position and phylogeny. In D. L. Lee (Ed.), The biology of nematodes (pp. 1–30). Taylor & Francis.
del Castillo-Olivares, A., & Smith, H. E. (2008). Critical contact residues that mediate polymerization of nematode major sperm protein. Journal of Cellular Biochemistry, 104(2), 477–487. https://doi.org/10.1002/jcb.21636
Dereeper, A., Guignon, V., Blanc, G., Audic, S., Buffet, S., Chevenet, F. et al. (2008). Phylogeny.fr: Robust phylogenetic analysis for the non-specialist. Nucleic Acids Research, 36, W465–W469. https://doi.org/10.1093/nar/gkn180
Ding, L., & Candido, E. P. M. (2000). HSP25, a small heat shock protein associated with dense bodies and M-lines of body wall muscle in Caenorhabditis elegans. Journal of Biological Chemistry, 275(13), 9510–9517. https://doi.org/10.1074/jbc.275.13.9510
Do, C. B., Mahabhashyam, M. S., Brudno, M., & Batzoglou, S. (2005). ProbCons: Probabilistic consistency-based multiple sequence alignment. Genome Research, 15(2), 330–340. https://doi.org/10.1101/gr.2821705
Duggal, C. L. (1978). In vitro studies on the functional maturation and locomotion of sperm in a free-living nematode, Panagrellus redivivus. Journal of Zoology, 186, 39–46. https://doi.org/10.1111/j.1469-7998.1978.tb03355.x
Edgar, R. C. (2004). MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Research, 32(5), 1792–1797. https://doi.org/10.1093/nar/gkh340
El-Gebali, S., Mistry, J., Bateman, A., Eddy, S. R., Luciani, A., Potter, S. C. et al. (2019). The Pfam protein families database in 2019. Nucleic Acids Research, 47(D1), D427–D432. https://doi.org/10.1093/nar/gky995
Ellis, R. E., & Stanfield, G. M. (2014). The regulation of spermatogenesis and sperm function in nematodes. Seminars in Cell & Developmental Biology, 29, 17–30. https://doi.org/10.1016/j.semcdb.2014.04.005
Fraire-Zamora, J. J., Broitman-Maduro, G., Maduro, M., & Cardullo, R. A. (2011). Evidence for phosphorylation in the MSP cytoskeletal filaments of amoeboid spermatozoa. International Journal of Biochemistry and Molecular Biology, 2(3), 263–273.
Felix, M. A. (2004). Developmental biology of nematodes–What we learn from Caenorhabditis elegans. In Z. X. Chen, S. Y. Chen, & D. W. Dickson (Eds.), Nematology-Advances and perspectives (Vol. 1, pp. 71–174). CABI Publishing.
Gasteiger, E., Hoogland, C., Gattiker, A., Wilkins, M. R., Appel, R. D., & Bairoch, A. (2005). Protein identification and analysis tools on the ExPASy server. In J. M. Walker (Ed.), The Proteomics Protocols Handbook (pp. 571–607): Humana Press.
Heger, P., Kroiher, M., Ndifon, N., & Schierenberg, E. (2010). Conservation of MAP kinase activity and MSP genes in parthenogenetic nematodes. BMC Developmental Biology, 10, 51. https://doi.org/10.1186/1471-213X-10-51
Höglund, J., Engstrom, A., Morrison, D. A., Mineur, A., & Mattsson, J. G. (2008). Limited sequence variation in the major sperm protein 1 (MSP) gene within populations and species of the genus Dictyocaulus (Nematoda). Parasitology Research, 103(1), 11–20. https://doi.org/10.1007/s00436-008-0877-8
Hojas, R. M., & Post, R. J. (2000). Regional genetic variation in the major sperm protein genes of Onchocerca volvulus and Mansonella ozzardi (Nematoda: Filarioidea). International Journal of Parasitology, 30(14), 1459–1465.
Holterman, M., van der Wurff, A., van den Elsen, S., van Megen, H., Bongers, T., Holovachov, O. et al. (2006). Phylum-wide analysis of SSU rDNA reveals deep phylogenetic relationships among nematodes and accelerated evolution toward crown clades. Molecular Biology and Evolution, 23(9), 1792–1800. https://doi.org/10.1093/molbev/msl044
Joshi, P. M., & Rothman, J. H. (2005). Nematode gastrulation: Having a BLASTocoel! Current Biology, 15(13), R495–R498. https://doi.org/10.1016/j.cub.2005.06.030
Justine, J. L. (2002). Male and female gametes and fertilisation. In D. L. Lee (Ed.), The Biology of Nematodes (pp. 73–120). Taylor & Francis.
Justine, J. L., & Jamieson, B. G. M. (1999). Nematoda. In B. G. M. Jamieson (Ed.), Reproductive biology of invertebrates (Vol. 9, Part B, pp. 183–266). New Delhi & Calcutta: Oxford & IBH.
Kasimatis, K. R., & Phillips, P. C. (2018). Rapid gene family evolution of a nematode sperm protein despite sequence hyper-conservation. G3 (Bethesda), 8(1), 353–362. https://doi.org/10.1534/g3.117.300281
King, K. L., Stewart, M., & Roberts, T. M. (1994a). Supramolecular assemblies of the Ascaris suum major sperm protein (MSP) associated with amoeboid cell motility. Journal of Cell Science, 107(Pt 10), 2941–2949.
King, K. L., Essig, J., Roberts, T. M., & Moerland, T. S. (1994b). Regulation of the Ascaris major sperm protein (MSP) cytoskeleton by intracellular pH. Cell Motility and the Cytoskeleton, 27(3), 193–205. https://doi.org/10.1002/cm.970270302
King, K. L., Stewart, M., Roberts, T. M., & Seavy, M. (1992). Structure and macromolecular assembly of two isoforms of the major sperm protein (MSP) from the amoeboid sperm of the nematode, Ascaris suum. Journal of Cell Science, 101(Pt 4), 847–857.
Klass, M. R., & Hirsh, D. (1981). Sperm isolation and biochemical analysis of the major sperm protein from Caenorhabditis elegans. Developmental Biology, 84(2), 299–312. https://doi.org/10.1016/0012-1606(81)90398-5
Kosinski, M., McDonald, K., Schwartz, J., Yamamoto, I., Greenstein, D. (2005). C. elegans sperm bud vesicles to deliver a meiotic maturation signal to distant oocytes. Development, 132(15), 3357–3369, doi: https://doi.org/10.1242/dev.01916
Lak, B., Yushin, V. V., Slos, D., Claeys, M., Decraemer, W., & Bert, W. (2015). High-pressure freezing and freeze-substitution fixation reveal the ultrastructure of immature and mature spermatozoa of the plant-parasitic nematode Trichodorus similis (Nematoda; Triplonchida; Trichodoridae). Micron, 77, 25–31. https://doi.org/10.1016/j.micron.2015.05.012
LeClaire, L. L., Stewart, M., & Roberts, T. M. (2003). A 48 kDa integral membrane phosphoprotein orchestrates the cytoskeletal dynamics that generate amoeboid cell motility in Ascaris sperm. Journal of Cell Science, 116(13), 2655–2663. https://doi.org/10.1242/jcs.00469
Lev, S., Ben Halevy, D., Peretti, D., & Dahan, N. (2008). The Vap protein family: From cellular functions to motor neuron disease. Trends in Cell Biology, 18(6), 282–290. https://doi.org/10.1016/j.tcb.2008.03.006
Linnaeus, C. (1767). Systema naturae per regna tria naturae: secundum classes, ordines, genera, species, cum characteribus, differentiis, synonymis, locis. Ed. 12. 1., Regnum Animale. 1 & 2. Holmiae [Stockholm], Laurentii Salvii. pp. 1-532 [1766] pp. 533-1327 [1767].
Malakhov, V. V. (1994). Nematodes. Structure, development, classification and phylogeny. Washington, USA: Smithsonian Institution Press.
Malakhov, V. V. (1998). Embryological and histological peculiarities of the order Enoplida, a primitive group of nematodes. Russian Journal of Nematology, 6(1), 41–46.
Marcello, M. R., Singaravelu, G., & Singson, A. (2012). Fertilization. Advances in Experimental Medicine and Biology, 757, 321–350. https://doi.org/10.1007/978-1-4614-4015-4_11
Miller, M. A., Nguyen, V. Q., Lee, M. H., Kosinski, M., Schedl, T., Caprioli, R. M., & Greenstein, D. (2001). A sperm cytoskeletal protein that signals oocyte meiotic maturation and ovulation. Science, 291(5511), 2144–2147. https://doi.org/10.1126/science.1057586
Miller, M. A., Ruest, P. J., Kosinski, M., Hanks, S. K., & Greenstein, D. (2003). An Eph receptor sperm-sensing control mechanism for oocyte meiotic maturation in Caenorhabditis elegans. Genes & Development, 17(2), 187–200. https://doi.org/10.1101/gad.1028303
Miyata, M., Robinson, R. C., Uyeda, T. Q. P., Fukumori, Y., Fukushima, S. I., Haruta, S. et al. (2020). Tree of motility - A proposed history of motility systems in the tree of life. Genes to Cells, 25(1), 6–21. https://doi.org/10.1111/gtc.12737
Morrow, E. H. (2004). How the sperm lost its tail: The evolution of aflagellate sperm. Biological Reviews of the Cambridge Philosophical Society, 79(4), 795–814. https://doi.org/10.1017/s1464793104006451
Poinar, G. O., & Hess-Poinar, R. T. (1993). The fine-structure of Gastromermis sp (Nematoda, Mermithidae) sperm. Journal of Submicroscopic Cytology and Pathology, 25(3), 417–431.
Roberts, T. M., & Stewart, M. (2000). Acting like actin. The dynamics of the nematode major sperm protein (MSP) cytoskeleton indicate a push-pull mechanism for amoeboid cell motility. Journal of Cell Biology, 149(1), 7–12. https://doi.org/10.1083/jcb.149.1.7
Roberts, T. M., & Stewart, M. (2012). Role of major sperm protein (MSP) in the protrusion and retraction of Ascaris sperm. International Review of Cell and Molecular Biology, 297, 265–293. https://doi.org/10.1016/b978-0-12-394308-8.00007-8
Rusin, L. Y., & Malakhov, V. V. (1998). Free-living marine nematodes possess no eutely. Doklady Biological Sciences, 361, 331–333.
Ryan, G. L., Petroccia, H. M., Watanabe, N., & Vavylonis, D. (2012). Excitable actin dynamics in lamellipodial protrusion and retraction. Biophysical Journal, 102(7), 1493–1502. https://doi.org/10.1016/j.bpj.2012.03.005
Schmidt-Rhaesa, A. (1997/98). Phylogenetic relationships of the Nematomorpha - A discussion of current hypotheses. Zoologischer Anzeiger, 236, 203–216.
Schulze, J., & Schierenberg, E. (2011). Evolution of embryonic development in nematodes. EvoDevo, 2(1), 18. https://doi.org/10.1186/2041-9139-2-18
Scott, A. L., Dinman, J., Sussman, D. J., & Ward, S. (1989). Major sperm protein and actin genes in free-living and parasitic nematodes. Parasitology, 98(Pt 3), 471–478. https://doi.org/10.1017/s0031182000061564
Sen, A., & Caiazza, F. (2013). Oocyte maturation: A story of arrest and release. Frontiers in Bioscience, 5, 451–477. https://doi.org/10.2741/s383
Sepsenwol, S., Ris, H., & Roberts, T. M. (1989). A unique cytoskeleton associated with crawling in the amoeboid sperm of the nematode, Ascaris suum. Journal of Cell Biology, 108(1), 55–66. https://doi.org/10.1083/jcb.108.1.55
Shepherd, A. M. (1981). Interpretation of sperm development in nematodes. Nematologica, 27(1), 122. https://doi.org/10.1163/187529281X00151
Singaravelu, G., & Singson, A. (2011). New insights into the mechanism of fertilization in nematodes. International Review of Cell and Molecular Biology, 289, 211–238. https://doi.org/10.1016/b978-0-12-386039-2.00006-7
Slos, D., Yushin, V. V., Claeys, M., Ivanova, E. S., Kosaka, H., & Bert, W. (2020). Structure, development, and evolutive patterns of spermatozoa in rhabditid nematodes (Nematoda: Rhabditida). Journal of Morphology, 281(11), 1411–1435. https://doi.org/10.1002/jmor.21255
Smith, H. E. (2014). Nematode sperm motility WormBook, 1–15. https://doi.org/10.1895/wormbook.1.68.2
Smythe, A. B., Holovachov, O., & Kocot, K. M. (2019). Improved phylogenomic sampling of free-living nematodes enhances resolution of higher-level nematode phylogeny. BMC Evolutionary Biology, 19(1), 121. https://doi.org/10.1186/s12862-019-1444-x
Srinivasan, J., Dillman, A. R., Macchietto, M. G., Heikkinen, L., Lakso, M., Fracchia, K. M., Antoshechkin, I., Mortazavi, A., Wong, G., & Sternberg, P. W. (2013). The draft genome and transcriptome of Panagrellus redivivus are shaped by the harsh demands of a free-living lifestyle. Genetics, 193(4), 1279-1295. https://doi.org/10.1534/genetics.112.148809
Strube, C., Buschbaum, S., & Schnieder, T. (2009). Molecular characterization and real-time PCR transcriptional analysis of Dictyocaulus viviparus major sperm proteins. Parasitology Research, 104(3), 543–551. https://doi.org/10.1007/s00436-008-1228-5
Tarr, D. E. K., & Scott, A. L. (2005). MSP domain proteins. Trends in Parasitology, 21(5), 224–231. https://doi.org/10.1016/j.pt.2005.03.009
van Megen, H., van den Elsen, S., Holterman, M., Karssen, G., Mooyman, P., Bongers, T. et al. (2009). A phylogenetic tree of nematodes based on about 1200 full-length small subunit ribosomal DNA sequences. Nematology, 11, 927-S927. https://doi.org/10.1163/156854109x456862
Voronov, D. A. (1999). The embryonic development of Pontonema vulgare (Enoplida: Oncholaimidae) with a discussion of nematode phylogeny. Russian Journal of Nematology, 7(2), 105–114.
Voronov, D. A., & Panchin, Y. V. (1998). Cell lineage in marine nematode Enoplus brevis. Development, 125(1), 143–150.
Ward, S., & Klass, M. (1982). The location of the major protein in Caenorhabditis elegans sperm and spermatocytes. Developmental Biology, 92(1), 203–208. https://doi.org/10.1016/0012-1606(82)90164-6
Waterhouse, A. M., Procter, J. B., Martin, D. M., Clamp, M., & Barton, G. J. (2009). Jalview version 2—A multiple sequence alignment editor and analysis workbench. Bioinformatics, 25(9), 1189–1191. https://doi.org/10.1093/bioinformatics/btp033
Wolf, N., Hirsh, D., & McIntosh, J. R. (1978). Spermatogenesis in males of the free-living nematode, Caenorhabditis elegans. Journal of Ultrastructure Research, 63, 155–169. https://doi.org/10.1016/S0022-5320(78)80071-9
Yushin, V. V., Afanasiev-Grigoriev, A. G., & Malakhov, V. V. (2014). The male gonad of the marine nematode Enoplus: No single distal tip cell but multiple uniform epithelial cells. Invertebrate Zoology, 11(2), 361–372. https://doi.org/10.15298/invertzool.11.2.07
Yushin, V. V., Claeys, M., & Bert, W. (2016). Ultrastructural immunogold localization of major sperm protein (MSP) in spermatogenic cells of the nematode Acrobeles complexus (Nematoda, Rhabditida). Micron, 89, 43–55. https://doi.org/10.1016/j.micron.2016.07.004
Yushin, V. V., & Malakhov, V. V. (1994). Ultrastructure of sperm cells in the female gonoduct of free-living marine nematodes from genus Enoplus (Nematoda: Enoplida). Fundamental and Applied Nematology, 17(6), 513–520.
Yushin, V. V., & Malakhov, V. V. (2004). Spermatogenesis and nematode phylogeny. In R. C. Cook, & D. J. Hunt (Eds.), Proceeding of the fourth international congress of nematology (Vol. 2, pp. 655–665, Nematology Monographs and Perspectives).
Yushin, V. V., & Malakhov, V. V. (2014). The origin of nematode sperm: Progenesis at the cellular level. Russian Journal of Marine Biology, 40(2), 71–81. https://doi.org/10.1134/S1063074014020114
Zograf, J. K. (2014). Ultrastructure of spermatogenesis and sperm of the free-living soil nematode Panagrellus redivivus (Rhabditida: Panagrolaimidae). Russian Journal of Nematology, 22(1), 39–48.
Acknowledgements
The authors are grateful to staff of the Far Eastern Center of Electron Microscopy (NSCMB FEB RAS) for their assistance in imaging and to staff of the White Sea Biological Station (Lomonosov Moscow State University) for the help in E. brevis collection. The authors are also thankful for the copyright permissions to use previously published data from the Russian Journal of Nematology (Fig. 3a and Fig. S1a) and the Fundamental and Applied Nematology (Brill) (Fig. 6b and Fig. S2).
Funding
This study was supported by Russian Foundation for Basic Research (Grant Nos. 17–04-00719-a and 14–04-00334-a).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zograf, J.K., Trebukhova, Y.A., Yushin, V.V. et al. Analysis of major sperm proteins in two nematode species from two classes, Enoplus brevis (Enoplea, Enoplida) and Panagrellus redivivus (Chromadorea, Rhabditida), reveals similar localization, but less homology of protein sequences than expected for Nematoda phylum. Org Divers Evol 22, 117–130 (2022). https://doi.org/10.1007/s13127-021-00522-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13127-021-00522-y