Abstract
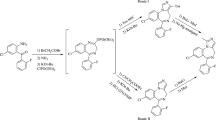
One-pot, three-component condensation reaction between (phenylsulfonyl)acetonitrile, aromatic aldehydes, and 6-aminouracil for preparation of 6,8a-dihydropyrido[2,3-d]pyrimidine derivatives has been reported. The method involves domino Knoevenagel condensation/Michael addition, and cyclization cascade. The reaction was performed in glycerol which is commercially available, inexpensive and non-toxic compound. High purity of the products, very high yields and wide scope of substrates are advantages of this protocol.



Similar content being viewed by others
References
M.C. Bagley, D.D. Hughs, R. Lioyd, V.E.C. Powers, Tetrahedron Lett. 42, 6585–6588 (2001)
F. Buron, J.Y. Merour, M. Akssira, G. Guillaumet, S. Routier, Eur. J. Med. Chem. 95, 46–95 (2015)
M. Mamaghani, R. Hossein Nia, J. Heterocyclic. Chem. 54, 1700–1722 (2017)
M. Shiri, Chem. Rev. 112, 3508–3549 (2012)
S. Asghari, S. Ramezania, M. Mohsenib, Chin. Chem. Lett. 25, 431–434 (2014)
D.J. Berry, Ch.V. Digiovanna, S.S. Metrick, R. Murugan, Arkivoc., 201–226 (2001)
M.F. Brana, M. Cacho, M.L. Garcia, E.P. Mayoral, B. Lopez, B. Teresa, J. Med. Chem. 48, 6843–6854 (2005)
M.L. Deb, P.J. Bhuyan, Synth. Commun. 36, 3085–3090 (2006)
A.B. Deyanov, R.K. Niyazov, F.Y. Nazmetdivov, B.Y. Syropyatov, V.E. Kolla, M.E. Konshin, J. Pharm. Chem. 25, 248–250 (1991)
I.O. Donkor, C.L. Klein, L. Liang, N. Zhu, E. Bradley, A.M. Clark, J. Pharm. Sci. 84, 661–664 (1995)
E.K. Goharshadi, Y. Ding, N.M. Jorabachi, P. Nancarrow, Ultrason. Sonochem. 16, 120–123 (2009)
S. Heck, A. Domling, Synlett. 3, 424–426 (2000)
P.T. Anastas, J.C. Warner, Green chemistry: theory and practice (Oxford University Press, Oxford, 1998)
M. Lancaster, Green Chemistry: an introductory text, 2nd edn. (RSC Publishing, Cambridge, 2010)
W.M. Nelson, Green solvents for chemistry: perspectives and practice (Oxford University Press, New York, 2003)
J.H. Clark, S.J. Taverner, Org. Process Res. Dev. 11, 149–155 (2007)
F.M. Kerton, Alternative solvents for green chemistry (RSC Publishing, Cambridge, 2009)
P.G. Jessop, Green Chem. 13, 1391–1398 (2011)
G. De Martino, M.C. Edler, G. La Regina, A. Coluccia, M.C. Barbera, D. Barrow, R.I. Nicholson, G. Chiosis, A. Brancale, E. Hamel, M. Artico, R. Silvestri, J. Med. Chem. 49, 947–954 (2006)
D.T. Johnson, K.A. Taconi, Environ. Prog. 26, 338–348 (2007)
N. Bakhrou, F. Lamaty, J. Martinez, E. Colacino, Tetrahedron Lett. 51, 3935–3937 (2010)
M. Li, C. Chen, F. He, Y. Gu, Adv. Synth. Catal. 352, 519–530 (2010)
J. Francos, V. Cadierno, Green Chem. 12, 1552–1555 (2010)
V.G. Ricordi, C.S. Freitas, G. Perin, E.J. Lenardão, R.G. Jacob, L. Savegnago, D. Alves, Green Chem. 14, 1030–1034 (2012)
G. Perin, L.G. Mello, C.S. Radatz, L. Savegnago, D. Alves, R.G. Jacob, E.J. Lenardão, Tetrahedron Lett. 51, 4354–4356 (2010)
C.S. Radatz, R.B. Silva, G. Perin, E.J. Lenardão, R.J. Jacob, D. Alves, Tetrahedron Lett. 52, 4132–4136 (2011)
Y. Dommaraju, Sh Bora, D. Prajapati, Org. Biomol. Chem. 13, 9181–9185 (2015)
S. Singh, M. Saquib, M. Singh, J. Tiwari, F. Tufail, J. Singh, J. Singh, New. J. Chem. 40, 63–67 (2016)
Acknowledgements
We gratefully appreciate the University of Sistan and Baluchestan Research Councils for the financial support of this work.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Morshedi, A., Shaterian, H.R. Green approach to synthesis of new series of 6,8a-dihydropyrido[2,3-d]pyrimidine derivatives. J IRAN CHEM SOC 16, 493–500 (2019). https://doi.org/10.1007/s13738-018-1528-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-018-1528-3