Abstract
Encephalitis is a multifaceted syndrome with a myriad of clinical presentations. It is defined as inflammation of the brain with functional disturbance. Infectious diseases, particularly viruses, and autoimmune disorders are the commonest causes in children. Young infants have the highest burden of disease for most infectious causes. Recent advances include the publication of international consensus guidelines, emergence of new aetiologies including human parechoviruses and better characterisation of known infectious causes, including enterovirus 71, which continue to cause large epidemics, death and impairment in children although a number promising vaccines are on the horizon; and neurological syndromes associated with influenza which occur most commonly in children who are not immunised against the virus. Understanding of childhood autoimmune encephalopathies is also rapidly expanding including responses to immunomodulation and prognosis. Future research should see better understanding of the pathogenesis of all forms of encephalitis, with hope for advances in therapy and prevention.
Similar content being viewed by others
Introduction
Acute encephalitis is a syndrome characterised by severe neurological dysfunction due to inflammation of the brain parenchyma. The presence of central nervous system (CNS) inflammation enables differentiation from ‘encephalopathy’ where there is altered cerebral function alone. The highest attack rate for encephalitis is in children, particularly in those less than 1 year of age [1–3]. The commonest causes of acute encephalitis are infections and autoimmune disorders in children, with viruses being the most commonly identified pathogens [4, 5••]. The diagnosis of encephalitis is complex because it is now recognised that some infectious agents (e.g. influenza virus) can cause encephalopathic syndromes without directly infecting the brain [6••, 7], and that infection or immunisation can trigger autoimmune encephalopathies [8]. There are also toxic and metabolic syndromes that cause brain inflammation and can mimic infectious encephalitis [8], including Mild Encephalopathy with Reversible Splenial Lesions [9]. It is also well established that a high proportion of cases (between 30 and 70 % depending on the population studied) will not have an aetiology identified despite extensive investigation [1, 4, 5••], although this proportion can be reduced with rigorous application of consensus guidelines [10, 11].
The incidence and case fatality rate of acute childhood encephalitis rates varies with geographic location, aetiology, age and case definition. The incidence of childhood encephalitis from all causes in industrialised countries has been calculated in a systematic review to be 10.5 cases per 100,000 population <15 years per year [11]. The overall mortality of encephalitis is approximately 10 % across all age groups in recent population studies [11], although mortality in children alone is much lower in most paediatric series, except in some Asian countries where there are ongoing outbreaks of non-polio enteroviruses, particularly human enterovirus-type 71 (EV71) and endemic Japanese Encephalitis Virus (JEV) [12–14]. Mortality rates from encephalitis have reduced in recent decades, with the availability of immunisations against measles and JEV in particular, antiviral agents and better supportive care [15, 16]. Although death rates have reduced, disability in encephalitis survivors can be high, depending on the pathogen and age [5••, 17•].
This review will discuss recent advances in paediatric acute encephalitis over the last few years commencing with a review of recently published consensus definitions and clinical guidelines, and of infectious encephalitis in children including emerging/re-merging pathogens, therapy and prevention and outcomes, and of childhood autoimmune encephalitides, including characterisation of individual syndromes, advances in immunomodulation and long-term prognosis. Finally, it will then provide summary of remaining knowledge gaps and suggested directions for future research.
Encephalitis Case Definitions and Practice Guidelines
The diagnosis or classification of encephalitis poses challenges for both clinicians and epidemiologists. A number of groups have developed consensus definitions for both brain inflammation and dysfunction for different purposes. In 2007, the Brighton collaboration Encephalitis Working group’s developed definitions to provide clarify around reported of adverse neurological events post immunisation including acute disseminated encephalomyelitis (ADEM). These have been widely adapted by many groups who study the condition [8]. The World Health organisation (WHO) also developed a clinical case definition for acute encephalitis syndrome (AES) in resource poor settings, mainly for the purpose of JEV surveillance [18]. Although brain biopsy provides a gold standard definition for brain inflammation, most schemas substitute a combination of surrogate markers of CNS inflammation and clinical signs of brain dysfunction. Definitions for CSF pleocytosis need to be adjusted for age, as the upper limit of normal values of CSF white cell counts is higher in newborn infants, than in older infants, children and adults. Recent advances have incorporated the fact that encephalitis can occur in the absence of CSF pleocytosis, and without neuroimaging abnormalities. In 2012, the international encephalitis consortium (IEC) published standardised definitions (Table 1) and consensus practice guidelines for children and adults to capture both encephalitis and encephalopathies of presumed infectious aetiologies [19••].
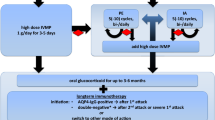
A number of clinical guidelines for the management of encephalitis in children have been recently been published [20, 21]. Common features of these have been the emphasis on early lumbar puncture where not contraindicated to exclude treatable causes of encephalitis (particularly HSV), the importance of early neuroimaging (preferably MRI) to confirm the diagnosis, the need to cease acyclovir where an alternative cause of encephalitis is found or HSV PCR is negative, early consultation with specialist neurologists and infectious diseases for advice re-extended investigations and diagnosis/management of autoimmune encephalitides. Key features of a pathognomonic infectious encephalitis, herpes simplex virus; an infection-associated encephalopathy, influenza virus; and an autoimmune encephalitis; N-methyl-d-aspartate receptor, or NMDAR, antibody mediated encephalitis, are compared in Table 2.
Infectious Encephalitis
Epidemiology, Clinical Characteristics
More than 100 pathogens have been reported to cause encephalitis, although for most infectious agents, this is usually a rare complication [16]. Granerod and colleagues devised a hierarchical relationship of diagnostics tests to define causality of an infectious agent [22]. In their classification, a confirmed case of infectious encephalitis is when an organism is identified in the brain, a highly probable case is when it is detected in a sterile site, a probable case is where there is organism carriage with a specific immune response, and a possible case is where there is the organism carriage (i.e. it is detected in a non-sterile site) without evidence of a specific immune response.
The epidemiology of childhood infectious encephalitis has been reported in a number of large series from around the world [3, 4, 12, 23–33]. Recent publications have provided new information. A hospital-based study of childhood encephalitis which arose out of the California Encephalitis project and published in 2013, mostly included children who were older than 12 months of age, were male and either Hispanic or White [4]. A key finding of this series was the identification of race/ethnicity as an independent predictor of outcomes. Clinical characteristics were similar to previous published series. Fever was the commonest presenting symptom (75 %) followed by seizures (50 %), ataxia (42 %), personality change (23 %), movement disorders (15 %) and hallucinations (6 %). Confirmed, probable or possible infectious causes were identified in just over 38 % of children, however, almost 50 % of children in this series did not have a defined aetiology. Enteroviruses were the commonest confirmed or probable infectious cause, and Mycoplasma pneumoniae was the commonest possible cause. A recent single-centre, retrospective case series of over 150 children with encephalitis from Australia identified infectious encephalitis in 30 % of cases, most commonly, enterovirus, M. pneumoniae and herpes simplex virus (HSV), and infection-associated encephalopathy in a further 8 %. Almost 30 % of children in this series did not have an identified aetiology [5••].
Prospective studies of encephalitis have been thought to provide more accurate estimates of incidence, aetiology and epidemiology of the syndrome. This notion has recently been evaluated in a recent French study which compared data from a prospective population-based study with national hospital discharge and mortality datasets [34]. The authors concluded that although misclassification can underestimate incidence and weaken conclusions from analysis of large datasets, the method does provide an inexpensive surveillance tool and accurate estimates of case fatality rates. A recent Canadian evaluation of national hospital morbidity datasets included admissions and deaths with attributable codes for encephalitis from all age groups [35]. Similar to other studies, it also identified that infants less than 12 months of age have highest risk of viral encephalitis (4.2/100,000 population) compared to other children (0.36–0.70/100,000 population), and identified clusters of unknown encephalitis, possibly vector-borne aetiology in older age groups.
Influenza-Associated Encephalopathies
Influenza RNA is very rarely identified directly in the CSF. However, influenza virus infection has been increasingly recognised to be complicated by a variety of encephalopathic syndromes, many but not all with evidence of CNS inflammation [6••, 7, 36, 37]. These include acute necrotizing encephalopathy (ANE), acute hemorrhagic shock and encephalopathy (HSE) and acute hemorrhagic leukoencephalopathy (AHL). Influenza-associated encephalopathies (IAE) have been reported after both pandemic and seasonal influenza [6••, 7, 36], affect children more than adults and can be associated with poor outcomes including death. Case series from around the world suggest somewhere between 6-20 % of children hospitalised with Influenza A H1N1 pandemic had neurological manifestations [7]. Surveillance studies from the United Kingdom (UK) and Japan have further characterised IAE in children [6••, 36, 38]. Recent results from UK surveillance for neurological complications of influenza (2011–2013) confirmed that children (84 %) were more commonly affected than adults, and that Influenza A (mostly H1N1) caused the majority (81 %) of cases [6••]. Of the 21 children in the series, slightly more had encephalopathy than encephalitis/encephalomyelitis; many had specific encephalopathy syndromes (ANE, HSE and AHL). Similar to previous series, many cases required intensive care (80 %) and had impairment at discharge (68 %), and death was not uncommon (16 %). Of note, and similar to previous reports from Australia [7], none of the children or adults had been previously vaccinated against influenza, including 32 % who had indications for this reinforcing that these syndromes are potentially preventable. Japanese surveillance for influenza-associated encephalopathy (IAE) (2009–2011) noted that attack rates were approximately 10-fold higher during the H1N1 Influenza A pandemic than from seasonal influenza, and again were highest in young children [36]. Case fatality rates of approximately 5–18 % were reported in this series.
Although clinical features of influenza encephalopathy syndromes can be similar, neuroimaging features are often distinct. A revised classification schema for IAE has recently been published [6••] which distinguishes syndromes associated with acute onset which are thought to arise from cytokine storm, from those with subacute onset which are hypothesised to arise from aberrant adaptive responses, or those with delayed/late onset where the pathogenesis is unknown. This classification is supported by neuroimaging findings from a small Chinese series of 17 children with severe IAE (H1N1) [38]. Treatment of influenza encephalopathy syndromes is supportive. Corticosteroids are often given early for some forms of IAE (e.g. ANE), but there are limited data of their efficacy [6••].
Herpes Simplex Virus (HSV)
In most series, HSV is one of the commonest confirmed infectious cause of encephalitis in children in the developed world together with enteroviruses [5••, 39] (Table 2). HSV encephalitis (HSE) in the newborn period typically arises after vertical transmission from active maternal genital herpes disease during labour and delivery. Sporadic HSE cases in later infancy and childhood are rare, and have been associated with specific signalling defects in innate immunity [40]. Results from 15 years of active surveillance for neonatal HSV disease in Australia (1997–2011) have recently been published [41•]. Approximately one-third of infants in this cohort of almost 250 neonates, had HSE alone or as part of a multiorgan disease. HSV-1 was the commonest serotype causing all forms of neonatal HSV disease, reflecting the recent increase in genital herpes due to HSV-1 disease in many countries including Australia. There were no deaths from HSE in the absence of disseminated infection, and only half the infants with encephalitis alone had cutaneous lesions at presentation.
Antiviral resistance is rarely encountered in neonatal HSV disease, but can occur, as shown by a recent case report from Japan of a neonate with HSE who developed a virologically confirmed acyclovir-resistant HSV 1 strain during therapy after initial treatment response [42]. The infant was successfully treated with vidarabine in the absence of foscarnet, but survived with significant neurological sequelae. Baja and colleagues recently described neuroimaging findings in a series of 29 infants who had neonatal HSE, the majority of whom had MRIs [43]. In contrast to older infants and children with HSE, temporal lobe predilection was uncommon, with most neonates having a diffuse restriction pattern on T2 images with involvement of thalamus and internal capsule. MRI abnormalities correlated with adverse neurological outcomes in this small series.
There have been a number of advances in antiviral therapy for neonatal or childhood HSE. Kimberlin et al. [44] have reported results from a randomised controlled trial of oral antiviral suppressive therapy (acyclovir at 30 mg/m2/dose three times a day) versus placebo at the cessation of intravenous treatment and found that acyclovir reduced neurological sequelae after neonatal HSV disease (predominantly HSV2 in this series) as shown by higher scores on developmental assessment at 12 months of age compared to controls. As HSV is a rare cause of encephalitis beyond the neonatal period, diagnosis and treatment can be problematic. Gaensbauer and colleagues [45] recently reported high rates of empiric acyclovir usage older infants and children for suspected HSE, and highlighted that in most cases the therapy was given in the absence of clinical characteristics of the condition. Expert opinion has seen a move towards the use of high dose acyclovir therapy (20 mg/kg/dose) for HSE not only in neonates, but in all children up to 12 years of age. The rarity of HSE makes it difficult to adequately power controlled trials to evaluate the efficacy of this recommendation [46]. Kendrick et al. has recently reported safety data from a retrospective cohort of 61 children aged from 1 month to 18 years of age who were given standard versus high dose acyclovir therapy for HSE. They did not identify a significant difference in the incidence of renal injury between the two groups.
Complications after HSE beyond the neonatal period can include choreiform movement disorders and epileptic encephalopathies. A number of authors have postulated an autoimmune basis for these phenomena [47, 48]. This notion is supported by a recent report of nine children from Australia where HSE relapse with chorea was associated with development to NMDAR or Dopamine-2 receptor antibodies [49•]. The initiation of immune suppression should be considered early in these conditions.
Non-polio Enterovirus Encephalitis Including HEV71
Non-polio enteroviruses (HEV) are one of the most common causes of infectious encephalitis in children [5••, 13, 39, 50]. In the California encephalitis project, HEV accounted for almost 5 % of all cause encephalitis, and occurred more commonly in the young and in males [4]. One of the most serious forms of EV encephalitis has been reported after enterovirus-type 71 infection (HEV71). HEV71 was first identified late last century during large epidemics of hand-foot-mouth disease (HFMD) across Europe and South East Asia [50]. More recently, epidemics of EV71 HFMD and febrile illnesses have emerged in eastern Asia, complicated by high rates of severe neurological disease in children including severe brainstem encephalitis, with high mortality and frequent permanent neurological sequelae [51]. HEV71 epidemics are seasonal, with the highest transmission rates occurring during warmer, wetter months [52]. Recent outbreak data from Malaysia and Australia suggest that epidemics occur in two to three year cycles corresponding with new, naive birth cohorts [53, 54]. Characteristic clinical features of HEV CNS disease include myoclonus, tremor, ataxia, nystagmus and cranial nerve palsies, which may develop with or without HFMD. Cohort studies have identified that serious HEV71 neurological disease (encephalomyelitis) is associated mortality and morbidly, particularly in the presence of neurogenic pulmonary oedema [55•, 56]. Characteristics of severe disease variably include young age (<2 years of age), male sex, prolonged high fever, high white cell count on admission, neurological signs at presentation, delayed medical evaluation and the existence of comorbidities such as pre-existing developmental retardation [56–58]. Predictors of neurological sequelae after EV71 encephalomyelitis included CSF pleocytosis and myoclonic jerks at disease onset [59].
There has also been considerable interest in identifying the pathogenesis of EV71 neurological disease. As encephalitis is a rare manifestation of EV71 disease, a number of studies have investigated genetic polymorphisms associated with EV71 encephalitis compared to uncomplicated HFMD. Single nucleotide polymorphisms within the genes encoding interleukin (IL)-17F, CCL2 gene, IL-6, IL-8 [60–62] appear to confer increased susceptibility to severe EV disease including encephalitis in Chinese populations. Elevation of human leukocyte antigen-G expression has recently been associated with severe encephalitis with pulmonary oedema [63].
Given the serious public health concerns from EV71 outbreaks, there have been concerted efforts to develop improved prevention strategies including infection control and vaccines. A comprehensive guide for clinical management of HFMD has recently been published by the World Health Organization [64]. Infection control practices consist mainly of hand washing, disinfection and isolation during epidemics. Over the last two years, results from three large phase three trials in China of vaccine candidates (all inactivated vaccines against EV71 C4Agenotype) have been published [65–67]. All candidates provided protection against HFMD (vaccine efficacy 80–97 %), but to date the trialled vaccines have been proven to work only for one sub-type of the HEV71. There is a need to determine EV subtypes causing disease around the globe, and to test the current HEV71 vaccines against other subtypes. There is also a need to develop potent antiviral treatments for children and adults who acquire this potentially devastating infection.
Parechovirus Encephalitis
Human parechoviruses (HPeV) have recently been recognised as causing similar clinical syndromes to EV, particularly in young infants [68–70]. HPeV like enteroviruses, are members of the picornavirus family of small, positive strand non enveloped RNA viruses, but are not detected by molecular testing for EV (e.g. polymerase chain reaction) and so must be specifically requested by clinicians. Only three HPeV serotypes (serotypes 1, 2 and 3) have been reported to cause disease in humans to date. HPeV-type 3 has been particularly associated with neurological disease including encephalitis, which is characterised by the absence of CSF pleocytosis and a significantly elevated CSF protein [69], and distinct subcortical white matter involvement on cerebral MRI [71]. Harvala and colleagues [68] have recently compared HPeV and EV RNA detections in the CSF and blood in infants presenting with sepsis or CNS disease and suggested that presence of either virus in the CSF alone may not be indicative of CNS disease in the absence of CSF pleocytosis. Infants with sepsis like syndromes due to HPeV3 had high viral load in the blood, but low to undetectable load in the CSF. Neurological sequelae after HPeV3 meningoencephalitis has been reported in some but not all infants [69, 71]. Further research is required to define the pathogenesis and prognostic risk factors after HPeV CNS infection.
Arbovirus Encephalitides
Arboviruses are an uncommon but emerging and possibly under recognised causes of encephalitis in children. Population studies using hospital datasets from around the world suggest that diagnostic testing for arboviruses in not always performed for suspected encephalitis, particularly in the young. Gaensbauer et al. have recently reported the epidemiology of paediatric neuroinvasive arboviral infections in the United States (2003–2013) [72•]. They reported over 1200 cases in their series, including over 20 deaths from a variety of viruses each with different seasonal and environmental triggers, most commonly La Crosse Virus (particularly in young children) and West Nile Virus (WNV). Eastern Equine encephalitis was an uncommon cause of neuroinvasive infection but had the highest case fatality rate. The study serves to reinforce the need to utilise personal protection against insect bites in children in areas of possible exposure.
The epidemiology of meningoencephalitis from tickborne encephalitis (TBE) virus in Switzerland from 2005 to 2011 has recently been reported [73]. TBE was uncommon in children under 6 years of age in this country, but thereafter occured at the same rate til late adulthood, where there was a further peak. Males were more commonly affected than females at all age groups and deaths were rare. The majority of those infected were not vaccinated. Large outbreaks of WNV are continuing to occur in Greece [74], particularly in rural areas, but neuroinvasive disease appears to be uncommon in the young.
Japanese Encephalitis Virus
Despite the availability of an effective vaccine against Japanese Encephalitis Virus (JEV), the virus remains the leading cause of AES in many parts of the Asia and the Western Pacific, particularly in rural areas [13, 75, 76]. The Centres for Disease Control in the United States recently provided an update on JEV surveillance and immunisation [77]. They estimated that there are almost 70,000 cases of JEV in Asia per year, and that while surveillance for JE occurred in 75 % of Asian countries with identified transmission risk, only 46 % of these countries had immunisation programs. Two recent reports have highlighted that burden of death and disability from this infection is in children. Wang et al. [78] have described characteristics of deaths from JEV in China, and found that children less than 15 years of age accounted for almost 80 % of over 1500 deaths from JEV from 2005 to 2010. Griffiths et al. [79•] reported a longitudinal follow-up study of JEV infection in Nepal, and highlighted that almost 70 % of child survivors of JEV had functional impairment at follow-up, frequently behavioural, and resulted in significant adverse economic impact to their families.
Considerable effort to develop new cell culture-derived JEV vaccines has been driven by theoretical safety concerns as the current inactivated JEV vaccines were developed in mouse brains. Results of phase III clinical trials of three cell culture JEV vaccines suggest that they are safe and with equivalent immunogenicity to inactivated vaccines [80].
Varicella Zoster Virus Encephalitis
Encephalitis is a known rare complication of varicella zoster virus (VZV) infection. Introduction of VZV immunisation into national schedules has seen the reduction of disease in many countries. However, a recent case series reported over 80 children with CNS complications of VZV from Canada, the majority of whom were not vaccinated against VZV, including 17 cases of encephalitis (three of whom died), and 10 who developed strokes, highlighted that vaccine preventable encephalitis can occur even in highly developed countries [81]. This series serves as a reminder that neurological symptoms associated with VZV infection can predate the rash. A recent case report of fatal wild-type VZV encephalitis which developed without a rash in a young child who had received one dose of VZV vaccine serves as a further reminder that VZV encephalitis should be considered even in there is a history of vaccination and the absence of rash at presentation [82].
Autoimmune Encephalitides
A major advance in encephalitis research in recent years has been in the recognition, diagnosis and treatment of immune mediated encephalitides in both children and adults. There is now clear evidence that these disorders can lead to significant long-term neurological sequelae. A recent review by Ramanathan et al. [83], provides a detailed update of autoimmune encephalitides, and highlights differences between neuronal surface antibody syndromes (NSAS) which are caused by antibodies against surface receptors or synaptic proteins and limbic encephalitis syndromes (LE), which are typically caused by antibodies directed against intracellular antigens. NSAS with onset in childhood include N-methyl-d-aspartate receptor (NMDAR), glycine receptor and recently described anti-dopamine-2 receptor (D2R) basal ganglia encephalitides [84•]. NMDAR are characterised by a number of features including behavioural disturbance, psychosis and movement disorders (Table 2). D2R manifest primarily as extrapyramidal movement disturbances with psychiatric features. LE are characterised by subacute onset of memory loss, confusion and agitation often with sleep disturbance hallucinations and hippocampal and medial temporal lobe changes on neuroimaging. Many LE are associated with underlying malignancies and often a relentlessly progressive clinical course.
The last few years have seen further characterisation of clinical phenotypes of these disorders in childhood including long-term outcome and further definition of treatment. The Spanish NMDAR encephalitis group [85] have recently highlighted age-associated differences in initial symptoms of the disorder, whereby children have more neurological and fewer psychiatric symptoms and signs at onset than adults. Consistent with previous reports, females were over-represented in this series, and most children did not have a tumour. All children in this series received immunomodulatory therapy (intravenous immunoglobulin, plasma exchange, steroids and or rituximab/cyclophosphamide) and most were reported to have responded to this therapy, but as this was an uncontrolled case series, efficacy data are hard to interpret. The benefits of rituximab, a monoclonal antibody against CD20 used to deplete B cells, have been shown in randomised controlled trials of adults with multiple sclerosis [86], but use in NMDAR has been limited to European observational cohort study of NMDAR in all age groups, including 211 children [87, 88•]. In this study, early treatment, and no admission to ICU were predictors of good outcome in children and adults. Dale et al. have recently reported higher quality evidence of the efficacy of rituximab in child and adolescent NMDAR [89•]. This multicentre case control study of 144 children with autoimmune neurological disorders, predominantly NMDAR, reported benefit in 87 % of patients with the greatest effect seen in early treatment. Adverse effects were observed in 7.6 %, most notably infections.
As mentioned previously, another new area of interest has been description of autoimmune syndromes, after infectious encephalitis particularly choreoathetosis post HSE from autoantibodies to NMDAR or D2R [85, 90]. Other associations include respiratory viruses with basal ganglia/thalamic encephalitis, influenza A and voltage-gated potassium channel complex encephalitis [91] and NMDAR and parvovirus B19 [92].
The field of autoimmune encephalitis is rapidly evolving with molecular techniques enabling new pathological antibodies being identified to a wide variety of CNS antigens, and better definition of known syndromes. However, as highlighted by Ramanathan and colleagues [83], there remain many gaps in our knowledge of the pathogenesis, diagnosis and management of these conditions in all age group including the overlap and role of antecedent infections, the use of CNS antibodies as biomarkers and the timing and duration of first line and second line immunotherapy.
Conclusions
Encephalitis remains a complex disorder to diagnose and treat, particularly in children. The condition is a well-recognised sentinel for emerging infectious agents and we remain without vaccines or therapies for known infectious causes which cause significant death and disability (e.g. HEV71). The child health clinician needs to be aware of the clinical phenotypes of autoimmune encephalitides and refer early for specialist advice and treatment as there is clear evidence of the benefits of early immunotherapy. However, even with advanced diagnostics and practice guidelines, many children with encephalitis do not have an aetiology identified. Many survivors of childhood encephalitis are left with lifelong consequences, the magnitude and character of which is poorly understood worldwide. Future collaborative international research is required to address these knowledge gaps, and identify new diagnosis, vaccines and therapies.
References
Recently published papers of particular interest have been highlighted as: • Of importance •• Of major importance
Granerod J, Ambrose HE, Davies NW, Clewley JP, Walsh AL, Morgan D, et al. Causes of encephalitis and differences in their clinical presentations in England: a multicentre, population-based prospective study. Lancet Infect Dis. 2010;10(12):835–44.
Mailles A, Stahl JP. Infectious encephalitis in France in 2007: a national prospective study. Clin Infect Dis. 2009;49(12):1838–47.
Glaser CA, Gilliam S, Schnurr D, Forghani B, Honarmand S, Khetsuriani N, et al. In search of encephalitis etiologies: diagnostic challenges in the California Encephalitis Project, 1998–2000. Clin Infect Dis. 2003;36(6):731–42.
DuBray K, Anglemyer A, LaBeaud AD, Flori H, Bloch K, Joaquin KS, et al. Epidemiology, outcomes and predictors of recovery in childhood encephalitis: a hospital-based study. Pediatr Infect Dis J. 2013;32(8):839–44.
•• Pillai SC, Hacohen Y, Tantsis E, Prelog K, Merheb V, Kesson A, Barnes E, Gill D, Webster R, Menezes M, Ardern-Holmes S, Gupta S, Procopis P, Troedson C, Antony J, Ouvrier RA, Polfrit Y, Davies NWS, Waters P, Lang B, Lim M, Brilot F, Vincent A, Dale RC. Infectious and autoantibody-associated encephalitis: clinical features and long-term outcome. Pediatric 2015;135(4):e974–e84. Retrospective cohort of 164 children with autoimmune and infectious encephalitis. Important as largest cohort to define long term outcomes in this age group.
•• Goenka A, Michael BD, Ledger E, Hart IJ, Absoud M, Chow G, et al. Neurological manifestations of influenza infection in children and adults: results of a National British Surveillance Study. Clin Infect Dis. 2014;58(6):775–84. Large cohort of influenza associated encephalopathies. Important because it provides a comprehensive classification for these disorders.
Khandaker G, Zurynski Y, Buttery J, Royle J, Richmond P, Marshall H, et al. Neurologic complications of pandemic influenza A (H1N1) 2009: surveillance results from 6 paediatric hospitals in Australia and critical analysis of the literature. Neurology. 2012;79(2):1474–81.
Sejvar JJKK, Bilynsky R, Blumberg D, Cvetkovich T, Galama J, Gidudu J, Katikaneni L, Khuri-Bulos N, Oleske J, Tapiainen T, Wiznitzer M, Brighton Collaboration Encephalitis Working Group. Encephalitis, myelitis, and acute disseminated encephalomyelitis (ADEM): case definitions and guidelines for collection, analysis, and presentation of immunization safety data. Vaccine. 2007;25:5771–92.
Ka ABP, Troedson C, Webster R, Procopis P, Ging J, Chua YW, Buckmaster A, Wood N, Jones C, Dale RC. Mild encephalopathy with reversible splenial lesion: an important differential of encephalitis. Europ J Paediatr Neurol. 2015;13:S1090-3798(15)00034-3.
Ambrose HEGJ, Clewley JP, Davies NW, Keir G, Cunningham R, Zuckerman M, Mutton KJ, Ward KN, Ijaz S, Crowcroft NS, Brown DW, UK Aetiology of Encephalitis Study Group. Diagnostic strategy used to establish etiologies of encephalitis in a prospective cohort of patients in England. J Clin Microbiol. 2011;49(10):3576–83.
Jmor F, Emsley HCA, Fischer M, Solomon T, Lewthwaite P. The incidence of acute encephalitis syndrome in Western industrialised and tropical countries. Virol J. 2008;5:134.
Ho Dang Trung N, Le Thi Phuong T, Wolbers M, Nguyen Van Minh H, Nguyen Thanh V, Van MP, et al. Aetiologies of central nervous system infection in Viet Nam: a prospective provincial hospital-based descriptive surveillance study. PLoS One. 2012;7(5):e37825.
Britton PNKG, Booy R, Jones CA. The causes and consequences of childhood encephalitis in Asia. Infect Disord Drug Targets. 2014;14(2):78–88.
Beig FK, Malik A, Rizvi M, Acharya D, Khare S. Etiology and clinico-epidemiological profile of acute viral encephalitis in children of western Uttar Pradesh, India. Int J Infect Dis. 2010;14(2):e141–6.
Huppatz C, Kelly PM, Levi C, Dalton C, Williams D, Durrheim DN. Encephalitis in Australia, 1979–2006: trends and aetiologies. Commun Dis Intell Q Rep. 2009;33(2):192–7.
Granerod J, Crowcroft NS. The epidemiology of acute encephalitis. Neuropsychol Rehabil. 2007;17(4–5):406–28.
• Ramanuj PP Granerød J, Davies NW, Conti S, Brown DW, Crowcroft NS. Quality of life and associated socio-clinical factors after encephalitis in children and adults in England: a population-based, prospective cohort study. PloS One. 2014;9(7):e103496. Most comprehresnsive study of QOL and social impact of childhood encephalitis to date.
Solomon TTT, Lewthwaite P, Ooi MH, Kneen R, Dung NM, White N. A cohort study to assess the new WHO Japanese encephalitis surveillance standards. Bull World Health Organ. 2008;86(3):178–86.
•• Venkatesan A, Tunkel AR, Bloch KC, Lauring AS, Sejvar J, Bitnun A, et al. Case definitions, diagnostic algorithms, and priorities in encephalitis: consensus statement of the international encephalitis consortium. Clin Infect Dis. 2013;57(8):1114–28. Most up to date nternational consensus definitions for encephalitis.
Britton PN, Eastwood K, Paterson B, Durrheim DN, Dale RC, Cheng AC, Kenedi C, Brew BJ, Burrow J, Nagree Y, Leman P, Smith DW, Booy R, Jones C. Consensus guidelines for the Investigation and Management of Encephalitis in adults and children in Australia and New Zealand: ASID, ACEM, ANZAN, PHAA. Intern Med J. 2015;45(5):563–76.
Kirkham FJ. Guidelines for the management of encephalitis in children. Dev Med Child Neurol. 2013;55(2):107–10.
Granerod J, Cunningham R, Zuckerman M, Mutton K, Davies NW, Walsh AL, et al. Causality in acute encephalitis: defining aetiologies. Epidemiol Infect. 2010;138(6):783–800.
Rantala H, Uhari M. Occurrence of childhood encephalitis: a population-based study. Pediatr Infect Dis J. 1989;8(7):426–30.
Koskiniemi M, Korppi M, Mustonen K, Rantala H, Muttilainen M, Herrgard E, et al. Epidemiology of encephalitis in children. A prospective multicentre study. Eur J Pediatr. 1997;156(7):541–5.
Kennedy CR, Chrzanowska K, Robinson RO, Tyrrell DA, Valman HB, Webster AD. A major role for viruses in acute childhood encephalopathy. Lancet. 1986;1(8488):989–91.
Fowler A, Stodberg T, Eriksson M, Wickstrom R. Childhood encephalitis in Sweden: etiology, clinical presentation and outcome. Eur J Paediatr Neurol. 2008;12(6):484–90.
Ilias A, Galanakis E, Raissaki M, Kalmanti M. Childhood encephalitis in Crete, Greece. J Child Neurol. 2006;21(10):910–2.
Kolski H, Ford-Jones EL, Richardson S, Petric M, Nelson S, Jamieson F, et al. Etiology of acute childhood encephalitis at The Hospital for Sick Children, Toronto, 1994–1995. Clin Infect Dis. 1998;26(2):398–409.
Ishikawa T, Asano Y, Morishima T, Nagashima M, Sobue G, Watanabe K, et al. Epidemiology of acute childhood encephalitis. Aichi Prefecture, Japan, 1984–1990. Brain Dev. 1993;15(3):192–7.
Chokephaibulkit K, Kankirawatana P, Apintanapong S, Pongthapisit V, Yoksan S, Kositanont U, et al. Viral etiologies of encephalitis in Thai children. Pediatr Infect Dis J. 2001;20(2):216–8.
Srey VH, Sadones H, Ong S, Mam M, Yim C, Sor S, et al. Etiology of encephalitis syndrome among hospitalized children and adults in Takeo, Cambodia, 1999–2000. Am J Trop Med Hyg. 2002;66(2):200–7.
Lee TC, Tsai CP, Yuan CL, Wei CY, Tsao WL, Lee RJ, et al. Encephalitis in Taiwan: a prospective hospital-based study. Jpn J Infect Dis. 2003;56(5–6):193–9.
Le VT, Phan TQ, Do QH, Nguyen BH, Lam QB, Bach V, et al. Viral etiology of encephalitis in children in southern Vietnam: results of a 1-year prospective descriptive study. PLoS Negl Trop Dis. 2010;4(10):e854.
Bernard SMA, Stahl JP, Steering Committee and Investigators Group. Epidemiology of infectious encephalitis, differences between a prospective study and hospital discharge data. Epidemiol Infect. 2013;141(11):2256–68.
LA Kulkarni MA, Artsob H, Drebot MA, Ogden NH. Epidemiology and aetiology of encephalitis in Canada, 1994–2008: a case for undiagnosed arboviral agents? Epidemiol Infect. 2013;141(11):2243–55.
Gu YST, Yasui Y, Tada Y, Kaku M, Okabe N. National surveillance of influenza-associated encephalopathy in Japan over 6 years, before and during the 2009–2010 influenza pandemic. PLoS One. 2013;8(1):e54786.
Mistry RD, Fischer JB, Prasad PA, Coffin SE, Alpern ER. Severe complications in influenza-like illnesses. Pediatrics. 2014;134(3):e684–90.
Zeng H, Quinet S, Huang W, Gan Y, Han C, He Y, Wang Y. Clinical and MRI features of neurological complications after influenza A (H1N1) infection in critically ill children. Pediatr Radiol. 2013;43(9):1182–9.
Britton PN, Dale RC, Booy R, Jones CA. Acute encephalitis in children: progress and priorities from an Australasian perspective. J Paediatr Child Health. 2015;51(2):147–58.
Casrouge A, Zhang S-Y, Eidenschenk C, Jouanguy E, Puel A, Yang K, et al. Herpes simplex virus encephalitis in human UNC-93B deficiency. Science. 2006;314(5797):308–12.
• Jones CA, Raynes Greenow C, Isaacs D; on behalf of APSU HSV study team and contributors. Population-based surveillance of neonatal HSV infection in Australia (1997–2011). Clin Infect Dis. 2014;59(4):525–31. Largest cohort of neonatal HSV including encephalitis from Southern Hemisphere. Documents the increased incidence of neonatal HSV1 disease.
Kakiuchi S, Nonoyama S, Wakamatsu H, Kogawa K, Wang L, Kinoshita-Yamaguchi H, Takayama-Ito M, Lim CK, Inoue N, Mizuguchi M, Igarashi T, Saijo M. Neonatal herpes encephalitis caused by a virologically confirmed acyclovir-resistant herpes simplex virus 1 strain. J Clin Microbiol. 2013;51(1):356–9.
Bajaj M, Mody S, Natarajan G. Clinical and neuroimaging findings in neonatal herpes simplex virus infection. J Pediatr. 2014;165(2):404–7.
Kimberlin DWWR, Wan W, Powell DA, Storch G, Ahmed A, Palmer A, Sánchez PJ, Jacobs RF, Bradley JS, Robinson JL, Shelton M, Dennehy PH, Leach C, Rathore M, Abughali N, Wright P, Frenkel LM, Brady RC, Van Dyke R, Weiner LB, Guzman-Cottrill J, McCarthy CA, Griffin J, Jester P, Parker M, Lakeman FD, Kuo H, Lee CH, Cloud GA, National Institute of Allergy and Infectious Diseases Collaborative Antiviral Study Group. Oral acyclovir suppression and neurodevelopment after neonatal herpes. N Engl J Med. 2011;365(14):1284–92.
Gaensbauer JT, Birkholz M, Pfannenstein K, Todd JK. Herpes PCR testing and empiric acyclovir use beyond the neonatal period. Pediatrics. 2014;134(3):e651–6.
Kendrick JG, Ensom MH, Steer A, White CT, Kwan E, Carr RR. Standard-dose versus high-dose acyclovir in children treated empirically for encephalitis: a retrospective cohort study of its use and safety. Paediatr Drugs. 2014;16(3):229–34.
Bektaş ÖTT, Kocabaş BA, Fitöz S, Ince E, Deda G. Anti-N-methyl-d-aspartate receptor encephalitis that developed after herpes encephalitis: a case report and literature review. Neuropediatrics. 2014;45(6):396–401.
Armangue TLF, Málaga I, Raspall-Chaure M, Marti I, Nichter C, Pugh J, Vicente-Rasoamalala M, Lafuente-Hidalgo M, Macaya A, Ke M, Titulaer MJ, Höftberger R, Sheriff H, Glaser C, Dalmau J. Herpes simplex virus encephalitis is a trigger of brain autoimmunity. Ann Neurol. 2014;75(2):317–23.
• Mohammad SS WG, Ramanathan S, Brilot F, Dale RC. Antipsychotic-induced akathisia and neuroleptic malignant syndrome in anti-NMDAR encephalitis. Ann Clin Psychiatry. 2014;26(4):297–8. Largest case series to date of childhood autoimmune encephalitis associated with HSV encephalitis.
Solomon T, Lewthwaite P, Perera D, Cardosa MJ, McMinn P, Ooi MH. Virology, epidemiology, pathogenesis, and control of enterovirus 71. Lancet Infect Dis. 2010;10(11):778–90.
McMinn PC. An overview of the evolution of enterovirus 71 and its clinical and public health significance. FEMS Microbiol Rev. 2002;26(1):91–9107.
Bek EJMP. The pathogenesis and prevention of encephalitis due to human enterovirus 71. Curr Infect Dis Rep. 2012;14(4):397–407.
Zander ABP, Navin T, Horsley E, Tobin S, McAnulty JM. An outbreak of enterovirus 71 in metropolitan Sydney: enhanced surveillance and lessons learnt. Med J Aust. 2014;201(11):63–6.
Tee KKTY, Kamarulzaman A. Emerging and re-emerging viruses in Malaysia, 1997–2007. Int J Infect Dis. 2009;13(3):307–18.
• Cheng HY, Huang YC, Yen TY, Hsia SH, Hsieh YC, Li CC, et al. The correlation between the presence of viremia and clinical severity in patients with enterovirus 71 infection: a multi-center cohort study. BMC Infect Dis. 2014;14:417. This study is important because it defines risk factors for severe EV71 disease in children.
Zhang Q, MacDonald NE, Smith JC, Cai K, Yu H, Li H, et al. Severe enterovirus type 71 nervous system infections in children in the Shanghai region of China: clinical manifestations and implications for prevention and care. Pediatr Infect Dis J. 2014;33(5):482–7.
Huang WC, Shih W, Yang SC, Yen TY, Lee JT, Huang YC, Li CC, Hsieh YC, Lin TY, Chang LY, Huang LMJ. Predicting severe enterovirus 71 infection: Age, comorbidity, and parental behavior matter. Microbiol Immunol Infect. 2014;S1684-182(14):00243-6.
Nguyen NT, Pham HV, Hoang CQ, Nguyen TM, Nguyen LT, Phan HC, et al. Epidemiological and clinical characteristics of children who died from hand, foot and mouth disease in Vietnam, 2011. BMC Infect Dis. 2014;14:341.
Tsai JD, Kuo HT, Chen SM, Lue KH, Sheu JN. Neurological images and the predictors for neurological sequelae of epidemic herpangina/hand-foot-mouth disease with encephalomyelitis. Neuropediatrics. 2014;45(2):102–8.
Li F, Liu X-P, Li J-A, Han Z-L, Liu P-P, Wang Y-Y, et al. Correlation of an interleukin-4 gene polymorphism with susceptibility to severe enterovirus 71 infection in Chinese children. Arch Virol. 2015;160(4):1035–42.
Li JA, Chen ZB, Lv TG, Han ZL. Genetic polymorphism of CCL2-2518, CXCL10-201, IL8 + 781 and susceptibility to severity of Enterovirus-71 infection in a Chinese population. Inflamm Res. 2014;63(7):549–56.
Yuan A, Li J, Liu P, Chen Z, Hou M, Wang J, et al. Association of interleukin-6-572C/G gene polymorphism and serum or cerebrospinal fluid interleukin-6 level with enterovirus 71 encephalitis in Chinese Han patients with hand, foot, and mouth disease. Inflammation. 2015;38(2):728–35.
Zheng XQ, Chen XQ, Gao Y, Fu M, Chen YP, Xu DP, et al. Elevation of human leukocyte antigen-G expression is associated with the severe encephalitis associated with neurogenic pulmonary edema caused by Enterovirus 71. Clin Exp Med. 2014;14(2):161–7.
World Heatlh Organisation. A Guide to clinical management and public health response for hand, foot and mouth disease (HFMD). 2011. http://www.wpro.who.int/publications/docs/GuidancefortheclinicalmanagementofHFMD.pdf. Accessed 30 March 2015.
Li R, Liu L, Mo Z, Wang X, Xia J, Liang Z, et al. An inactivated enterovirus 71 vaccine in healthy children. N Engl J Med. 2014;370(9):829–37.
Zhu F, Xu W, Xia J, Liang Z, Liu Y, Zhang X, et al. Efficacy, safety, and immunogenicity of an enterovirus 71 vaccine in China. N Engl J Med. 2014;370(9):818–28.
Zhu F-C, Meng F-Y, Li J-X, Li X-L, Mao Q-Y, Tao H, et al. Efficacy, safety, and immunology of an inactivated alum-adjuvant enterovirus 71 vaccine in children in China: a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2013;381(9882):2024–32.
Harvala H, Griffiths M, Solomon T, Simmonds P. Distinct systemic and central nervous system disease patterns in enterovirus and parechovirus infected children. J Infect. 2014;69(1):69–74.
Verboon-Maciolek MA, Groenendaal F, Hahn CD, Hellmann J, van Loon AM, Boivin G, et al. Human parechovirus causes encephalitis with white matter injury in neonates. Ann Neurol. 2008;64(3):266–73.
Esposito S, Rahamat-Langendoen J, Ascolese B, Senatore L, Castellazzi L, Niesters HG. Pediatric parechovirus infections. J Clin Virol. 2014;60(2):84–9.
Renna S, Bergamino L, Pirlo D, Rossi A, Furione M, Piralla A, et al. A case of neonatal human parechovirus encephalitis with a favourable outcome. Brain Dev. 2014;36(1):70–3.
• Gaensbauer JT, Lindsey NP, Messacar K, Staples JE, Fischer M. Neuroinvasive arboviral disease in the United States: 2003 to 2012. Pediatrics. 2014;134(3):e642–50. Largest cohort of arbovirus encephalits in all age groups to date.
Schuler M, Zimmermann H, Altpeter E, Heininger U. Epidemiology of tick-borne encephalitis in Switzerland, 2005 to 2011. Euro Surveill. 2014;19(13):1–7.
Pervanidou D, Detsis M, Danis K, Mellou K, Papanikolaou E, Terzaki I, et al. West Nile virus outbreak in humans, Greece, 2012: third consecutive year of local transmission. Euro Surveill. 2014;19:13.
Solomon T. Control of Japanese encephalitis—within our grasp? N Engl J Med. 2006;355(9):869–71.
Solomon T. Recent advances in Japanese encephalitis. J Neurovirol. 2003;9(2):274–83.
Campbell GL, Hills SL, Fischer M, Jacobson JA, Hoke CH, Hombach JM, et al. Estimated global incidence of Japanese encephalitis: a systematic review. Bull World Health Organ. 2011;89(10):766–74.
Wang LZW, Ding F, Sun H, Yu S, Li S, Chen Z, Huang L, Li C. Deaths associated with Japanese encephalitis, China, 2005–2010. Clin Infect Dis. 2013;56(5):752.
• Griffiths MJ LJ, Rayamajhi A, Poudel P, Shrestha P, Srivastav V, Kneen R, Medina-Lara A, Singh RR, Solomon T. The functional, social and economic impact of acute encephalitis syndrome in Nepal—a longitudinal follow-up study. PLoS Negl Trop Dis 2013;7(9):e2383. This study is important as it highlights the consequences of encephalitis in resource poor settings.
Yun S-I, Lee Y-M. Japanese encephalitis: the virus and vaccines. Hum Vaccin Immunother. 2014;10(2):263–79.
Science M, MacGregor D, Richardson SE, Mahant S, Tran D, Bitnun A. Central nervous system complications of varicella-zoster virus. J Pediatr. 2014;165(4):779–85.
Ibraheem MMM, Leung J, Bryce CH, Schmid DS, Zaki SR, Drew C, Liu L, Smelser C. Fatal wild-type varicella-zoster virus encephalitis without a rash in a vaccinated child. Pediatr Infect Dis J. 2013;32(2):183–5.
Ramanathan SMS, Brilot F, Dale RC. Autoimmune encephalitis: recent updates and emerging challenges. J Clin Neurosci. 2014;21(5):722–30.
• Dale RC MV, Pillai S, Wang D, Cantrill L, Murphy TK, Ben-Pazi H, Varadkar S, Aumann TD, Horne MK, Church AJ, Fath T, Brilot F. Antibodies to surface dopamine-2 receptor in autoimmune movement and psychiatric disorders. Brain Dev. 2012;135(11):3453–6. This study is the first description of this new autoimmune mediated encephalitis syndrome.
Armangue T, Titulaer M, Málaga I, Bataller L, Gabilondo I, Graus F, Dalmau J, Spanish Anti-N-methyl-D-Aspartate Receptor (NMDAR) Encephalitis Work Group. Pediatric anti-N-methyl-d-aspartate receptor encephalitis-clinical analysis and novel findings in a series of 20 patients. J Pediatr. 2013;162(4):850–6.
Hauser SLWE, Arnold DL, Vollmer T, Antel J, Fox RJ, Bar-Or A, Panzara M, Sarkar N, Agarwal S, Langer-Gould A, Smith CH, HERMES Trial Group. B-cell depletion with rituximab in relapsing-remitting multiple sclerosis. N Engl J Med. 2008;358(7):676–88.
Titulaer MJ, Hoftberger R, Iizuka T, Leypoldt F, McCracken L, Cellucci T, et al. Overlapping demyelinating syndromes and anti-N-methyl-d-aspartate receptor encephalitis. Ann Neurol. 2014;75(3):411–28.
• Titulaer MJ McCracken L, Gabilondo I, Armangué T, Glaser C, Iizuka T, Honig LS, Benseler SM, Kawachi I, Martinez-Hernandez E, Aguilar E, Gresa-Arribas N, Ryan-Florance N, Torrents A, Saiz A, Rosenfeld MR, Balice-Gordon R, Graus F, Dalmau J. Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: an observational cohort study. Lancet Neurol. 2013;12(2):157–65. This cohort study describes effects of immunomodulation for immune mediated encephalitis.
• Dale RC, Brilot F, Duffy LV, Twilt M, Waldman AT, Narula S, et al. Utility and safety of rituximab in pediatric autoimmune and inflammatory CNS disease. Neurology. 2014;83(2):142–50. This case control study reports the efficacy of B cell depleting agents in improving outcomes for autoimmune encephalitis in children.
Mohammad SSSK, Pillai S, Merheb V, Aumann TD, Gill D, Dale RC, Brilot F. Herpes simplex encephalitis relapse with chorea is associated with autoantibodies to N-Methyl-d-aspartate receptor or dopamine-2 receptor. Mov Disord. 2014;29(1):117–22.
Lin JJ, Lin KL, Hsia SH, Chou ML, Hung PC, Hsieh MY, et al. Anti-N-methyl-d-aspartate receptor encephalitis in Taiwan—a comparison between children and adults. Pediatr Neurol. 2014;50(6):574–80.
Grillo E, Silva RJM. Anti-N-methyl-d-aspartate receptor encephalitis and parvovirus B19: A possible link? J Pediatr. 2013;163(4):1223–4.
Britton PN, Jones CA. Central nervous system herpesvirus infections. Paediatr Child Health. 2014;24(6):248–54.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
Cheryl A Jones has received grants from NHMRC during the conduct of this study.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Infectious Diseases.
Rights and permissions
About this article
Cite this article
Jones, C.A. Paediatric Acute Encephalitis: Infection and Inflammation. Curr Pediatr Rep 3, 201–210 (2015). https://doi.org/10.1007/s40124-015-0089-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40124-015-0089-5