Abstract
Background
The multiple sclerosis (MS) landscape has changed over the past two decades across the world and in the Middle East. The Middle East is an ethnically diverse region located between 12° and 42° of latitude and 35° and 54° of longitude and varying altitudes. The magnitude of the shifts observed in the epidemiology and management of MS differ in each region and from country to country.
Objectives
The aim of this study was to provide a clinicodemographic overview of the cohorts of patients contributed to MSBase, a large international MS registry, in the Middle East and describe disease-modifying treatment (DMT) utilization in the different countries within the region. Understanding the differences between these cohorts is integral to interpretation of the studies conducted using registry data and provides insight into clinical practice in these cohorts.
Methods
The MSBase registry was searched for patients with MS or clinically isolated syndrome from the Middle Eastern countries with data captured between 2009 and 2018. In 2-year epochs, and with special focus on the most recent epoch (2017–2018), we explored the demographic, clinical characteristics and treatment exposures of the studied cohorts and reported the results using standard descriptive statistics.
Results
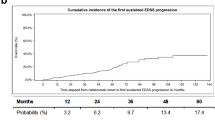
Over the 10-year study period, 13,356 patients from 17 centers in 8 Middle Eastern countries fulfilled the inclusion criteria. The represented countries were Egypt, Iran, Kuwait, Lebanon, Oman, Saudi Arabia, Turkey and the United Arab Emirates. Overall, the represented cohort was young (median 36 years, quartiles 29–45) and captured relatively early after the onset of MS (median disease duration < 10 years, quartiles 3–12). The relapsing-remitting phenotype was the most prevalent phenotype in all countries (73–97%) and the highest proportion of progressive MS was reported in Saudi Arabia (12%). Median Expanded Disability Status Scale (EDSS) ranged from 0 to 3, depicting a mildly disabled cohort, with the exception of Saudi Arabia where the median EDSS was 4 (quartiles 1.5–6.5). The median relapse frequency was highest in Lebanon (median 1.03, 95% CI 0.94–1.16) followed by Egypt (median 1.02, 95% CI 0.89–1.24) and lowest in Saudi Arabia (median 0.70, 95% CI 0.58–0.95) and Kuwait (median 0.75, 95% CI 0.71–0.80). The treatment landscape greatly varied between different countries. Platform injectable therapies were mostly utilized in Egypt, Iran and Turkey (86%, 79% and 53%, respectively), while oral therapies and monoclonal antibodies were more commonly used in Kuwait, Lebanon and the United Arab Emirates (87.2%, 67.3% and 58.7%, respectively).
Conclusion
Patients in the Middle East enrolled in a large multinational registry are representative of the general MS population. The spectrum of therapies used in the individual countries, however, is highly variable. Further studies that include rural and non-academic practices are needed to enhance our understanding of the MS cohorts in the Middle East.

Similar content being viewed by others
References
Esman MJ, Rabinovich I. Ethnicity, Pluralism, and the State in the Middle East [Internet]. Cornell University Press; 1988. https://doi.org/10.7591/j.ctvr7f5dw.
Online Latitude and Longitude Finder [Internet]. https://latitudelongitude.org/. Accessed 30 Apr 2021.
Wallin MT, Culpepper WJ, Nichols E, Bhutta ZA, Gebrehiwot TT, Hay SI, et al. Global, regional, and national burden of multiple sclerosis 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019;18:269–85.
Alroughani R, Ahmed S, Behbahani R, Khan R, Thussu A, Alexander K, et al. Increasing prevalence and incidence rates of multiple sclerosis in Kuwait. Mult Scler J. 2014;20:543–7.
Etemadifar M, Sajjadi S, Nasr Z, Firoozeei TS, Abtahi SH, Akbari M, et al. Epidemiology of multiple sclerosis in iran: a systematic review. Eur Neurol. 2013;70:356–63.
Heydarpour P, Mohammad K, Yekaninejad MS, Elhami SR, Khoshkish S, Sahraian MA. Multiple sclerosis in Tehran, Iran: A joinpoint trend analysis. Mult Scler J. 2014;20:512.
Türk Börü Ü, Duman A, Kulualp AŞ, Güler N, Taşdemir M, Yılmaz Ü, et al. Multiple sclerosis prevalence study The comparison of 3 coastal cities, located in the black sea and mediterranean regions of Turkey. Medicine (Baltimore). 2018;97:e12856.
Çelik Y, Birgili Ö, Kiyat A, Güldiken B, Özkan H, Yilmaz H, et al. Prevalence of multiple sclerosis in the metropolitan area of Edirne city, Turkey. Balkan Med J. 2011;28:193–6.
Heydarpour P, Khoshkish S, Abtahi S, Moradi-Lakeh M, Sahraian MA. Multiple sclerosis epidemiology in Middle East and North Africa: a systematic review and meta-analysis. Neuroepidemiology. 2015;44:232–44.
Al Tahan AM, Alsharoqi I, Bohlega SA, Dahdaleh M, Daif A, Deleu D, et al. Characteristics of multiple sclerosis in the Middle East with special reference to the applicability of international guidelines to the region. Int J Neurosci. 2014;124:635–41.
Abdollahpour I, Nedjat S, Mansournia MA, Eckert S, Weinstock-Guttman B. Infectious exposure, antibiotic use, and multiple sclerosis: A population-based incident case-control study. Acta Neurol Scand. 2018;138:308–14.
Abbasi M, Nabavi SM, Fereshtehnejad SM, Jou NZ, Ansari I, Shayegannejad V, et al. Multiple sclerosis and environmental risk factors: a case-control study in Iran. Neurol Sci. 2017;38:1941–51.
Tharakan J, Chand P, Jacob PC. 3–31-26 Multiple sclerosis in Oman: Clinical and epidemiological study. J Neurol Sci. 1997;150:S188.
Akhtar N, Elsetouhy A, Deleu D, Kamran S, Alhail H, Elalamy O, et al. Newly diagnosed multiple sclerosis in state of Qatar. Clin Neurol Neurosurg. 2013;115:1333–7.
Deleu D, Mir D, Al Tabouki A, Mesraoua R, Mesraoua B, Akhtar N, et al. Prevalence, demographics and clinical characteristics of multiple sclerosis in Qatar. Mult Scler J. 2013;19:816–9.
Yamout B, Itani S, Arabi A, Hamzeh D, Yaghi S. Prognostic factors of multiple sclerosis in Lebanon. Int J Neurosci. 2010;120:206–10.
Yamout B, Barada W, Tohme RA, Mehio-Sibai A, Khalifeh R, El-Hajj T. Clinical characteristics of multiple sclerosis in Lebanon. J Neurol Sci. 2008;270:88–93.
Butzkueven H, Chapman J, Cristiano E, Grand’Maison F, Hoffmann M, Izquierdo G, et al. MSBase: An international, online registry and platform for collaborative outcomes research in multiple sclerosis. Mult Scler. 2006;12:769–74.
Kalincik T, Butzkueven H. The MSBase registry: Informing clinical practice. Mult Scler J. 2019;25(14):1828–34.
Polman CH, Reingold SC, Edan G, Filippi M, Hartung H-P, Kappos L, et al. Diagnostic criteria for multiple sclerosis: 2005 revisions to the “McDonald Criteria.” Ann Neurol. 2005;58:840–6.
Polman CH, Reingold SC, Banwell B, Clanet M, Cohen JA, Filippi M, et al. Diagnostic criteria for multiple sclerosis: 2010 Revisions to the McDonald criteria. Ann Neurol. 2011;69:292–302.
Thompson AJ, Banwell BL, Barkhof F, Carroll WM, Coetzee T, Comi G, et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018;17:162–73.
Lublin FD, Reingold SC, Cohen JA, Cutter GR, Thompson AJ, Wolinsky JS, et al. Defining the clinical course of multiple sclerosis. Neurology. 2014;83(3):278–86.
D’Souza M, Yaldizli Ö, John R, Vogt DR, Papadopoulou A, Lucassen E, et al. Neurostatus e-scoring improves consistency of expanded disability status scale assessments: a proof of concept study. Mult Scler J. 2017;23:597–603.
Schumacher GA, Beebe G, Kibler RF, Kurland LT, Kurtzke JF, McDowell F, et al. Problems of experimental trials of therapy in multiple sclerosis: report by the panel on the evaluation of experimental trials of therapy in multiple sclerosis. Ann N Y Acad Sci. 1965;122:552–68.
Roxburgh RHSR, Seaman SR, Masterman T, Hensiek AE, Sawcer SJ, Vukusic S, et al. Multiple sclerosis severity score: Using disability and disease duration to rate disease severity. Neurology. 2005;64:1144–51.
Zhou Y, Claflin SB, Stankovich J, van der Mei I, Simpson S, Roxburgh RH, et al. Redefining the multiple sclerosis severity score (MSSS): the effect of sex and onset phenotype. Mult Scler J. 2019;00:1–10.
Martinez WL, Martinez AR. Computational Statistics Handbook with MATLAB, Second Edition (Chapman & Hall/Crc Computer Science & Data Analysis). Chapman & Hall/CRC; 2007.
El Tallawy HNA, Farghaly WMA, Rageh TA, Shehata GA, Badry R, Metwally NA, et al. Door-to-door survey of major neurological disorders (project) in Al Quseir City, Red Sea Governorate. Egypt Neuropsychiatr Dis Treat. 2013;9:767–71.
Ghandehari K, Riasi HR, Nourian A, Boroumand AR. Prevalence of multiple sclerosis in north east of Iran. Mult Scler. 2010;16:1525–6.
Mousavizadeh A, Dastoorpoor M, Naimi E, Dohrabpour K. Time-trend analysis and developing a forecasting model for the prevalence of multiple sclerosis in Kohgiluyeh and Boyer-Ahmad Province, southwest of Iran. Public Health. 2018;154:14–23.
Kazemi V, Sarmadi M, Tatari M, Naja F, Esmaeili A, Hadei M. Epidemiology of Multiple Sclerosis in Torbat-e Heydarieh (Northeast of Iran) during 1982–2016. Multiple Scler Related Disord. 2018;24:184–9.
Etemadifar M, Abtahi SH, Akbari M, Murray RT, Ramagopalan SV, Fereidan-Esfahani M. Multiple sclerosis in Isfahan, Iran: an update. Mult Scler J. 2014;20:1145–7.
Eskandarieh S, Nedjat S, Abdollahpour I, Moghadasi AN, Azimi AR, Sahraian MA. Comparing epidemiology and baseline characteristic of multiple sclerosis and neuromyelitis optica: a case-control study. Mult Scler Relat Disord. 2017;12:39–43.
Al-Din ASN, Khogali M, Poser CM, Al-Nassar KE, Shakir R, Hussain J, et al. Epidemiology of multiple sclerosis in Arabs in Kuwait: A comparative study between Kuwaitis and Palestinians. J Neurol Sci. 1990;100:137–41.
Atlas of MS 2013 [Internet]. Mult. Scler. Int. Fed. 2013. http://www.msif.org/wp-content/uploads/2014/09/Atlas-of-MS.pdf. Accessed 30 Apr 2021.
Tharakan JJ, Chand RP, Poovathoor CJ. Multiple sclerosis in Oman. Neuroscience. 2005;10:225.
Daif AK, Al-Rajeh S, Awada A, Al Bunyan M, Ogunniyi A, AbdulJabar M, et al. Pattern of presentation of multiple sclerosis in Saudi Arabia: analysis based on clinical and paraclinical features. Eur Neurol [Internet]. 1998;39:182–6. https://doi.org/10.1159/000007931.
Börü ÜT, Alp R, Sur H, Gül L. Prevalence of multiple sclerosis door-to-door survey in Maltepe, Istanbul. Turkey Neuroepidemiology. 2006;27:17–21.
Türk Börü Ü, Taşdemir M, Güler N, Dilaver Ayık E, Kumaş A, Yıldırım S, et al. Prevalence of multiple sclerosis: door-to-door survey in three rural areas of coastal black sea regions of Turkey. Neuroepidemiology. 2011;37:231–5.
Alp R, Ilhan Alp S, Planci Y, Yapici Z, Türk BÜ. The prevalence of multiple sclerosis in the north Caucasus region of turkey: door-to-door epidemiological field study. Noropsikiyatri Ars. 2012;49:272–5.
Inshasi J, Thakre M. Prevalence of multiple sclerosis in Dubai. United Arab Emirates Int J Neurosci. 2011;121:393–8.
Schiess N, Huether K, Fatafta T, Fitzgerald KC, Calabresi PA, Blair I, et al. How global MS prevalence is changing: a retrospective chart review in the United Arab Emirates. Mult Scler Relat Disord. 2016;9:73–9.
Yamout BI, Assaad W, Tamim H, Mrabet S, Goueider R. Epidemiology and phenotypes of multiple sclerosis in the Middle East North Africa (MENA) region. Mult Scler J - Exp Transl Clin. 2020;6(1):2055217319841881–2055217319841881.
The World Bank. Fairness and accountability: engaging in health systems in the Middle East and North Africa. 2015;1–67. http://www-wds.worldbank.org/external/default/WDSContentServer/WDSP/IB/2013/10/09/000356161_20131009152447/Rendered/PDF/817230WP0P12940Box0379842B00PUBLIC0.pdf. Accessed 30 Apr 2021.
Zeineddine MM, Yamout BI. Treatment of multiple sclerosis in special populations: the case of refugees. Mult Scler J Exp Transl Clin [Internet]. 2020;6:2055217319848466. https://doi.org/10.1177/2055217319848466.
El-Salem K, Al-Shimmery E, Horany K, Al-Refai A, Al-Hayk K, Khader Y. Multiple sclerosis in Jordan: A clinical and epidemiological study. J Neurol. 2006;253:1210–6.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Funding
No financial support was utilized in the study design, data gathering, analysis and writing of this manuscript.
Conflicts of interest
The MSBase Foundation acknowledges financial contributions to support the MSBase Registry from Biogen, Novartis, Merck, Roche and Sanofi Genzyme. NM has received compensation for consulting services from Actoverco Pharmaceuticals. RA received honoraria as a speaker and for serving on scientific advisory boards from Bayer, Biogen, GSK, Merck, Novartis, Roche and Sanofi-Genzyme. MT received travel grants from Novartis, Bayer-Schering, Merck and Teva; has participated in clinical trials by Sanofi Aventis, Roche and Novartis. CB received conference travel support from Biogen, Novartis, Bayer-Schering, Merck and Teva; has participated in clinical trials by Sanofi Aventis, Roche and Novartis. AA received personal fees and speaker honoraria from Teva, Merck, Biogen - Gen Pharma, Roche, Novartis, Bayer, Sanofi-Genzyme; received travel and registration grants from Merck, Biogen - Gen Pharma, Roche, Sanofi-Genzyme and Bayer. TK served on scientific advisory boards for BMS, Roche, Sanofi Genzyme, Novartis, Merck and Biogen, a steering committee for Brain Atrophy Initiative by Sanofi Genzyme, received conference travel support and/or speaker honoraria from WebMD Global, Novartis, Biogen, Sanofi-Genzyme, Teva, BioCSL and Merck and received research or educational event support from Biogen, Novartis, Genzyme, Roche, Celgene and Merck. SSH, CM, SO, VSH, BY, RT, RK, SH, AS, JI and TAH declare no conflicts of interest.
Ethics approval
This research was conducted in accordance with the World Medical Association Declaration of Helsinki and was approved by Melbourne Health Human Research Ethics Committee (2006.044) and local ethics committees in participating centers (unless exemption granted by the local regulations). MSBase is registered with WHO International Clinical Trials Registry Platform (ID ACTRN12605000455662). All participants have provided written or verbal consent to be registered in MSBase. Only pseudonymized non-identifiable data was utilized for the purpose of this study.
Availability of data and material
The data analyzed in this study are the property of the individual contributing centers. They can be made available upon reasonable request for the purpose of replication of the analyses included in this study and at the discretion of the principal investigators.
Code availability
Available upon request.
Consent for publication
Not applicable.
Author contributions
NM, SS, CM and TK designed, analyzed and drafted the manuscript. RA, SO, VS, MT, CB, BY, RT, RK, SH, AS, AA, JI, TAH revised the manuscript.All authors have read and approved the submitted manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Moradi, N., Sharmin, S., Malpas, C. et al. Utilization of Multiple Sclerosis Therapies in the Middle East Over a Decade: 2009–2018. CNS Drugs 35, 1097–1106 (2021). https://doi.org/10.1007/s40263-021-00833-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40263-021-00833-w