Abstract
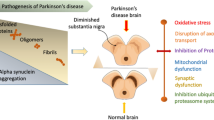
Globally the leading source of disability is neurological disorder. The chronic, complex and rapidly progressive neurological disorder in the world is Parkinson's disease (PD). PD is an age based neurodegenerative disorder. According to previous report the number of people with Parkinson’s disease has doubled from 1995 to 2015, to above 6 million. The protein α-synuclein has been strongly associated with the pathogenesis of Parkinson’s disease. α-synuclein plays a neuropathological role in the dysfunction of dopamine neurons in Parkinson’s disease. Here, we designed a structure-based drug galangin as an effective α-synuclein regulator, naturally occurring flavonoid, plants and traditional Chinese medicines. The present study was aimed to analyze the quantum chemical with the basis set of B3YLP/6–311 + + G (d,p) and molecular docking studies to find the binding efficiency of target protein and ligand molecule and determine the physiochemical and ADMET properties of galangin. Density function theory revealed the structural geometry, vibrational spectrum and the stability and reactivity of the lead molecule galangin. Molecular Docking score reveals that galangin showed a good binding efficiency of -9.4 kcal/mol at the active site of α-synuclein and RC plot analysis showed that the interacted protein and ligand had a stable-linkage, which suggests galangin a potential candidate for further development of drug in-vitro and in-vivo against Parkinson’s disease.









Similar content being viewed by others
References
Cookson MR (2009) α-Synuclein and neuronal cell death. Mol Neurodegener 4(1):1–14. https://doi.org/10.1186/1750-1326-4-9
Dauer W, Przedborski S (2003) Parkinson’s disease: mechanisms and models. Neuron 39(6):889–909. https://doi.org/10.1016/S0896-6273(03)00568-3
Delamarre A, Meissner WG (2017) Epidemiology, environmental risk factors and genetics of Parkinson’s disease. La Presse Médicale 46(2):175–181. https://doi.org/10.1016/j.lpm.2017.01.001
Dutheil F, Beaune P, Tzourio C, Loriot MA, Elbaz A (2010) Interaction between ABCB1 and professional exposure to organochlorine insecticides in Parkinson disease. Arch Neurol 67(6):739–745. https://doi.org/10.1001/archneurol.2010.101
Hu CY, Fang Y, Li FL, Dong B, Hua XG, Jiang W, Zhang XJ (2019) Association between ambient air pollution and Parkinson’s disease: systematic review and meta-analysis. Environ Res 168:448–459. https://doi.org/10.1016/j.envres.2018.10.008
Costa J, Lunet N, Santos C, Santos J, Vaz-Carneiro A (2010) Caffeine exposure and the risk of Parkinson’s disease: a systematic review and meta-analysis of observational studiess. J Alzheimers Dis 20(s1):S221–S238. https://doi.org/10.3233/JAD-2010−091525
Tan LC, Koh WP, Yuan JM, Wang R, Au WL, Tan JH, Yu MC (2008) Differential effects of black versus green tea on risk of Parkinson’s disease in the Singapore Chinese Health Study. Am J Epidemiol 167(5):553–560. https://doi.org/10.1093/aje/kwm338
Ritz B, Ascherio A, Checkoway H, Marder KS, Nelson LM, Rocca WA, Gorell J (2007) Pooled analysis of tobacco use and risk of Parkinson disease. Arch Neurol 64(7):990–997. https://doi.org/10.1001/archneur.64.7.990
Wolozin B, Wang SW, Li NC, Lee A, Lee TA, Kazis LE (2007) Simvastatin is associated with a reduced incidence of dementia and Parkinson’s disease. BMC Med 5(1):1–11. https://doi.org/10.1186/1741-7015-5-20
Yan J, Qiao L, Tian J, Liu A, Wu J, Huang J, Lai X (2019) Effect of statins on Parkinson’s disease: a systematic review and meta-analysis. Medicine 98:12
Pasternak B, Svanström H, Nielsen NM, Fugger L, Melbye M, Hviid A (2012) Use of calcium channel blockers and Parkinson’s disease. Am J Epidemiol 175(7):627–635. https://doi.org/10.1093/aje/kwr362
Goedert M, Jakes R, Spillantini MG (2017) The synucleinopathies: twenty years on. J Parkinsons Dis 7(s1):S51–S69. https://doi.org/10.3233/JPD-179005
Polymeropoulos MH, Lavedan C, Leroy E, Ide SE, Dehejia A, Dutra A, Nussbaum RL (1997) Mutation in the α-synuclein gene identified in families with Parkinson’s disease. Science 276(5321):2045–2047. https://doi.org/10.1126/science.276.5321.2045
Golbe LI, Di Iorio G, Bonavita V, Miller DC, Duvoisin RC (1990) A large kindred with autosomal dominant Parkinson’s disease. Ann Neurol 27(3):276–282. https://doi.org/10.1002/ana.410270309
Polymeropoulos MH, Higgins JJ, Golbe LI, Johnson WG, Ide SE, Di Iorio G, Duvoisin RC (1996) Mapping of a gene for Parkinson’s disease to chromosome 4q21-q23. Science 274(5290):1197–1199. https://doi.org/10.1126/science.274.5290.1197
Spillantini MG, Schmidt ML, Lee VMY, Trojanowski JQ, Jakes R, Goedert M (1997) α-Synuclein in Lewy bodies. Nature 388(6645):839–840. https://doi.org/10.1038/42166
Morgan J (2015) A seat at the table for people with Parkinson’s disease. Lancet Neurol 14(11):1077–1078. https://doi.org/10.1016/S1474-4422(15)00246-X
Viswanathan TM, Chitradevi K, Zochedh A, Vijayabhaskar R, Sukumaran S, Kunjiappan S, Kathiresan T (2022) Guanidine–curcumin complex-loaded amine-functionalised hollow mesoporous silica nanoparticles for breast cancer therapy. Cancers 14(14):3490. https://doi.org/10.3390/cancers14143490
Naylor J, Minard A, Gaunt HJ, Amer MS, Wilson LA, Migliore M, Bon RS (2016) Natural and synthetic flavonoid modulation of TRPC5 channels. Br J Pharmacol 173(3):562–574. https://doi.org/10.1111/bph.13387
Haid S, Novodomská A, Gentzsch J, Grethe C, Geuenich S, Bankwitz D, Pietschmann T (2012) A plant-derived flavonoid inhibits entry of all HCV genotypes into human hepatocytes. Gastroenterology 143(1):213–222. https://doi.org/10.1053/j.gastro.2012.03.036
Chen GL, Fan MX, Wu JL, Li N, Guo MQ (2019) Antioxidant and anti-inflammatory properties of flavonoids from lotus plumule. Food Chem 277:706–712. https://doi.org/10.1016/j.foodchem.2018.11.040
Pharmacopoeia, C. (2010). Commission. Pharmacopoeia of the People’s Republic of China.
Zhang W, Lan Y, Huang Q, Hua Z (2013) Galangin induces B16F10 melanoma cell apoptosis via mitochondrial pathway and sustained activation of p38 MAPK. Cytotechnology 65(3):447–455. https://doi.org/10.1007/s10616−012-9499-1
Zha WJ, Qian Y, Shen Y, Du Q, Chen FF, Wu ZZ, Huang M (2013) Galangin abrogates ovalbumin-induced airway inflammation via negative regulation of NF-B. Evid-Based Complement Alternat Med. https://doi.org/10.1155/2013/767689
Huang H, Chen AY, Rojanasakul Y, Ye X, Rankin GO, Chen YC (2015) Dietary compounds galangin and myricetin suppress ovarian cancer cell angiogenesis. J Funct Foods 15:464–475. https://doi.org/10.1016/j.jff.2015.03.051
Ciolino HP, Yeh GC (1999) The flavonoid galangin is an inhibitor of CYP1A1 activity and an agonist/antagonist of the aryl hydrocarbon receptor. Br J Cancer 79(9):1340–1346. https://doi.org/10.1038/sj.bjc.6690216
Janeoo S, Saroa A, Kumar R, Kaur H (2022) Computational investigation of bioactive 2, 3-diaryl quinolines using DFT method: FT-IR, NMR spectra, NBO, NLO, HOMO-LUMO transitions, and quantum–chemical properties. J Mol Struct 1253:132285. https://doi.org/10.1016/j.molstruc.2021.132285
Thangarasu S, Chitradevi A, Siva V, Shameem A, Murugan A, Viswanathan TM, Bahadur SA (2022) Structural, spectroscopic, cytotoxicity and molecular docking studies of charge transfer salt: 4-aminiumantipyrine salicylate. Polycyclic Aromat Compd. https://doi.org/10.1080/10406638.2022.2064883
Prathap KC, Lokanath NK (2018) Three novel coumarin-benzenesulfonylhydrazide hybrids: synthesis, characterization, crystal structure, Hirshfeld surface, DFT and NBO studies. J Mol Struct 1171:564–577. https://doi.org/10.1016/j.molstruc.2018.06.022
Naray-Szabo G, Peterson MR (1981) d-orbital participation in bonding in thiophene: comparison of CNDO/2 and ab initio results. J Mol Struct (Thoechem) 85(3–4):249–255. https://doi.org/10.1016/0166-1280(81)85023-3
Murugan M, Rajamohan R, Anitha A, Fatiha M (2022) Non-covalent bonding interaction between primaquine as guest and 2-(hydroxypropyl)-β-cyclodextrin as host. Polycyclic Aromat Compd 42(4):1861–1878. https://doi.org/10.1080/10406638.2020.1813181
Praveena A, Prabu S, Madi F, Rajamohan R (2021) Theoretical investigation of inclusion complexes of 3-hydroxyflavone and quercetin as guests with native and modified β-cyclodextrins as hosts. Polycycl Aromat Compd. https://doi.org/10.1080/10406638.2021.2009526
Chiş V (2004) Molecular and vibrational structure of 2, 4-dinitrophenol: FT-IR, FT-Raman and quantum chemical calculations. Chem Phys 300(1–3):1–11. https://doi.org/10.1016/j.chemphys.2004.01.003
Asensio A, Kobko N, Dannenberg JJ (2003) Cooperative hydrogen-bonding in adenine—thymine and guanine—cytosine base pairs. Density functional theory and Møller—Plesset molecular orbital study. J Phys Chem 107(33):6441–6443. https://doi.org/10.1021/jp0344646
Frisch, M. J. E. A., Trucks, G. W., Schlegel, H. B., Scuseria, G. E., Robb, M. A., Cheeseman, J. R., & Fox, A. D. (2009). Gaussian 09, revision D. 01.
Glendening, E. D., Badenhoop, J. K., Reed, A. E., Carpenter, J. E., Bohmann, J. A., Morales, C. M., Weinhold, F. (2004). NBO 6.0, Theoretical Chemistry Institute, University of Wisconsin, Madison, WI, 2013.
Etter MC, Urbańczyk-Lipkowska Z, Baer S, Barbara PF (1986) The crystal structures and hydrogen-bond properties of three 3-hydroxy-flavone derivatives. J Mol Struct 144(1–2):155–167. https://doi.org/10.1016/0022-2860(86)80175-2
Novena LM, Athimoolam S, Anitha R, Bahadur SA (2022) Synthesis, crystal structure, hirshfeld surface analysis, spectral and quantum chemical studies of pharmaceutical cocrystals of a bronchodilator drug (Theophylline). J Mol Struct 1249:131585. https://doi.org/10.1016/j.molstruc.2021.131585
Elanthiraiyan M, Jayasudha B, Arivazhagan M (2015) Molecular structure, vibrational spectroscopy, NBO and HOMO, LUMO studies of o-methoxybenzonitrile. Spectrochim Acta Part A Mol Biomol Spectrosc 134:543–552. https://doi.org/10.1016/j.saa.2014.04.103
Siva V, Devi AC, Thangarasu S, Viswanathan TM, Athimoolam S, Bahadur SA (2022) Design, structural, DFT, molecular docking studies and biological evaluation of 4-aminiumantipyrine dihydrogenphosphate monohydrate. J Mol Struct 1250:131866. https://doi.org/10.1016/j.molstruc.2021.131866
Silverstein RM, Bassler GC (1962) Spectrometric identification of organic compounds. J Chem Educ 39(11):546. https://doi.org/10.1021/ed039p546
Balachandran V, Nataraj A, Karthick T (2013) Molecular structure, spectroscopic (FT-IR, FT-Raman) studies and first-order molecular hyperpolarizabilities, HOMO–LUMO, NBO analysis of 2-hydroxy-p-toluic acid. Spectrochim Acta Part A Mol Biomol Spectrosc 104:114–129. https://doi.org/10.1016/j.saa.2012.11.052
Premkumar S, Jawahar A, Mathavan T, Dhas MK, Sathe VG, Benial AMF (2014) DFT calculation and vibrational spectroscopic studies of 2-(tert-butoxycarbonyl (Boc)-amino)-5-bromopyridine. Spectrochim Acta Part A Mol Biomol Spectrosc 129:74–83. https://doi.org/10.1016/j.saa.2014.02.147
Tsimogiannis D, Samiotaki M, Panayotou G, Oreopoulou V (2007) Characterization of flavonoid subgroups and hydroxy substitution by HPLC-MS/MS. Molecules 12(3):593–606. https://doi.org/10.3390/12030593
Dávila YA, Sancho MI, Almandoz MC, Gasull E (2018) Spectroscopic and electronic analysis of chelation reactions of galangin and related flavonoids with nickel (II). J Chem Eng Data 63(5):1488–1497. https://doi.org/10.1021/acs.jced.7b01058
Siva V, Murugan A, Shameem AS, Priya MU, Thangarasu S, Athimoolam S, Bahadur SA (2022) Design and supramolecular architecture of stepped molecular aggregation in monochloroacetate salt of 2-aminopyridine: Its bacterial and cancer inhibitory properties. J Mol Struct 1250:131888. https://doi.org/10.1016/j.molstruc.2021.131888
Thamarai A, Vadamalar R, Raja M, Muthu S, Narayana B, Ramesh P, Aayisha S (2020) Molecular structure conformational analyses, solvent-electronic studies through theoretical studies and biological profiling of (2E)-1-(3-bromo-2-thienyl)-3-(4–Chlorophenyl)-prop-2-en-1-one. J Mol Struct 1202:127349. https://doi.org/10.1016/j.molstruc.2019.127349
Sıdır İ, Sıdır YG, Kumalar M, Taşal E (2010) Ab initio Hartree-Fock and density functional theory investigations on the conformational stability, molecular structure and vibrational spectra of 7-acetoxy-6-(2, 3-dibromopropyl)-4, 8-dimethylcoumarin molecule. J Mol Struct 964(1–3):134–151. https://doi.org/10.1016/j.molstruc.2009.11.023
Zochedh AA, Bahadur SA, Kathiresan T (2021) Quantum chemical and molecular docking studies of naringin: a potent anti-cancer drug. J Cardiovasc Dis Res 12:1140–1148
Jeyaseelan SC, Premkumar R, Kaviyarasu K, Benial AMF (2019) Spectroscopic, quantum chemical, molecular docking and in vitro anticancer activity studies on 5-Methoxyindole-3–Carboxaldehyde. J Mol Struct 1197:134–146. https://doi.org/10.1016/j.molstruc.2019.07.042
Devi AC, Siva V, Thangarasu S, Athimoolam S, Bahadur SA (2021) Supramolecular architecture, thermal, quantum chemical analysis and in vitro biological properties on sulfate salt of 4-aminoantipyrine. J Mol Struct 1245:131033. https://doi.org/10.1016/j.molstruc.2021.131033
Scrocco E, Tomasi J (1978) Electronic molecular structure, reactivity and intermolecular forces: an euristic interpretation by means of electrostatic molecular potentials. Advances in quantum chemistry, vol 11. Academic Press, New York, pp 115–193
Munoz-Caro C, Nino A, Senent ML, Leal JM, Ibeas S (2000) Modeling of protonation processes in acetohydroxamic acid. J Org Chem 65(2):405–410. https://doi.org/10.1021/jo991251x
Deb BM (1981) Force concept in chemistry. Van Nostrand Reinhold
Politzer P, Laurence PR, Jayasuriya K (1985) Molecular electrostatic potentials: an effective tool for the elucidation of biochemical phenomena. Environ Health Perspect 61:191–202. https://doi.org/10.1289/ehp.8561191
Politzer, P., & Murray, J. S. (1991). Theoretical biochemistry and molecular biophysics: a comprehensive survey, vol. 2. Protein. In Electrostatic Potential Analysis of Dibenzo-p-dioxins and Structurally Similar Systems in Relation to their Biological Activities. Adenine Press Schenectady, NY.
Thangarasu S, Siva V, Asath Bahadur S, Athimoolam S (2021) Structural, vibrational, quantum chemical calculations, thermal and antimicrobial studies on nitrate salt of 3-nitroaniline. Opt Quant Electron 53(10):1–16. https://doi.org/10.1007/s11082−021−03146-w
Politzer P, Truhlar DG (eds) (2013) Chemical applications of atomic and molecular electrostatic potentials: reactivity, structure, scattering, and energetics of organic, inorganic, and biological systems. Springer Science & Business Media, Berlin
Sheela NR, Muthu S, Sampathkrishnan S (2014) Molecular orbital studies (hardness, chemical potential and electrophilicity), vibrational investigation and theoretical NBO analysis of 4–4′-(1H–1, 2, 4-triazol-1-yl methylene) dibenzonitrile based on abinitio and DFT methods. Spectrochim Acta Part A Mol Biomol Spectrosc 120:237–251. https://doi.org/10.1016/j.saa.2013.10.007
AlRabiah H, Muthu S, Al-Omary F, Al-Tamimi AM, Raja M, Muhamed RR, El-Emam AAR (2017) Molecular structure, vibrational spectra, NBO, Fukui function, HOMO-LUMO analysis and molecular docking study of 6-[(2-methylphenyl) sulfanyl]-5-propylpyrimidine-2, 4 (1H, 3H)-dione. Maced J Chem Chem Eng 36(1):59–80
Szafran M, Komasa A, Bartoszak-Adamska E (2007) Crystal and molecular structure of 4–Carboxypiperidinium chloride (4-piperidinecarboxylic acid hydrochloride). J Mol Struct 827(1–3):101–107. https://doi.org/10.1016/j.molstruc.2006.05.012
Sebastian S, Sundaraganesan N (2010) The spectroscopic (FT-IR, FT-IR gas phase, FT-Raman and UV) and NBO analysis of 4-hydroxypiperidine by density functional method. Spectrochim Acta Part A Mol Biomol Spectrosc 75(3):941–952. https://doi.org/10.1016/j.saa.2009.11.030
Zochedh A, Priya M, Chakaravarthy C, Sultan AB, Kathiresan T (2022) Experimental and computational evaluation of syringic acid-structural, spectroscopic, biological activity and docking simulation. Polycyclic Aromat Compd. https://doi.org/10.1080/10406638.2022.2118332
Clark DE (1999) Rapid calculation of polar molecular surface area and its application to the prediction of transport phenomena. 1. Prediction of intestinal absorption. J Pharmac Sci 88(8):807–814. https://doi.org/10.1021/js9804011
Zochedh A, Priya M, Shunmuganarayanan A, Thandavarayan K, Sultan AB (2022) Investigation on structural, spectroscopic, DFT, biological activity and molecular docking simulation of essential oil Gamma-Terpinene. J Mol Struct 1268:133651. https://doi.org/10.1016/j.molstruc.2022.133651
Sharmili Banu R, Mohana Priya I, Azar Zochedh A (2022) Identification of novel bioactive compounds from banana fruit (Musa sapientum) as antidepressant in pregnant women: molecular docking, physiochemical and ADMET evaluation. AJBGE 55(1):9–24
Chandran K, Shane DI, Zochedh A, Sultan AB, Kathiresan T (2022) Docking simulation and ADMET prediction based investigation on the phytochemical constituents of Noni (Morinda citrifolia) fruit as a potential anticancer drug. In Silico Pharmacol 10(1):1–14. https://doi.org/10.1007/s40203−022−00130-4
Funding
The authors declare that no funds, grants, or other support were received during this study.
Author information
Authors and Affiliations
Contributions
The work was carried out in collaboration among all authors. Quantum chemical investigation and analysis was performed by ABS and AZ. The Molecular docking and Drug-likeness investigation was performed by MP and analyzed by KA. RC plot analysis was performed by AZ. Validation of the manuscript was done by ABS and AZ. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors have no relevant financial or non-financial interests to disclose.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Priya, M., Zochedh, A., Arumugam, K. et al. Quantum Chemical Investigation, Drug-Likeness and Molecular Docking Studies on Galangin as Alpha-Synuclein Regulator for the Treatment of Parkinson’s Disease. Chemistry Africa 6, 287–309 (2023). https://doi.org/10.1007/s42250-022-00508-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42250-022-00508-z