Abstract
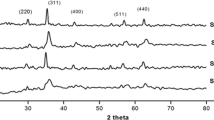
Nickel ferrite powders with particle sizes in the 3–5 μm range have been prepared from coprecipitated nickel–iron oxalate precursors. Firing the nickel–iron oxalate precursor in the range 300–1100°C produced samples of high chemical purity, while introducing significant variations in the distribution of crystallite sizes and surface morphologies. An increase in the powder density from 4.2 to 5.2 g cm-3 and a decrease in the surface area of the nickel ferrite from 120 to 0.2 m2g-1 were effected by increasing the firing temperature to 1100°C.
Similar content being viewed by others
References
T. E. Rummery, J. J. Hawton and D. Owen, Corrosion 33 (1977) 369.
J. Y. Chung and K. J. Lee, High Temp. Sci. 30 (1990) 51.
P. Lahiri and S. K. Sengupta, J. Chem. Soc., Faraday Trans. 91 (1995) 3489.
M. Tsuji, H. Kato, T. Kodama, S. Chang, N. Hesegawa and Y. Tamaura, J. Mater. Sci. 29 (1994) 6227.
M. Do Carmo Rangel, R. M. Sassaki and F. Galembeck, Catal. Lett. 33 (1995) 237.
Y. Torii, A. Tsuziki, K. Kato, Y. Uwamino, B. H. Choi and M. J. Lee, J. Mater. Sci. 31 (1996) 2603.
V. Moye, K. S. Rane and V. N. Kamat Dalal, J. Mater. Sci.: Mater. Electr. 1 (1990) 212.
E. Uzunova, D. Klissurski and S. Kassabov, J. Mater. Chem. 4 (1994) 153.
K. Sajata and T. O. Mason, J. Amer. Ceram. Soc. 75 (1992) 557.
F. Novelo and R. Valenzuela, Mater. Res. Bull. 30 (1995) 335.
L. Fa-Shen, Y. De-Sheng, Z. Hua-Xin, L. Xin-Fen and Y. Zheng, Hyperfine Interactions, 41 (1988) 525.
K. Suresh and K. C. Patil, J. Solid State Chem. 99 (1992) 12.
D. Gajapathy and K. C. Patil, Mater. Chem. Phys. 9 (1983) 423.
T. E. Rummery and D. D. Macdonald, J. Nucl. Mater. 55 (1975) 23.
F. Gonzalez, J. M. T. Raycheba and P. Spekkens, in 'Proceedings of the Second International Symposium on the Environmental Degradation of Materials in Nuclear Reactors' (American Nuclear Society, Lagrange Park, Ill 1986) p. 539.
R. L. Tapping, C. W. Turner, R. H. Thompson and R. H. Thompson, Corrosion 47 (1991) 489.
R. A. Speranzini, R. Voit and M. Helms, Nucl. Engng. Int. 53 (1990) 52.
P. V. Balakrishnan, P. Mcsweeny, C. R. Frost and P. Walmsley, Nucl. Technol. 55 (1981) 349.
E. W. Nuffield, “X-ray Diffraction Methods” (J. Wiley, New York, 1966) p. 157.
S. Brunauer, P. H. Emmett and E. Teller, J. Amer. Chem. Soc. 60 (1938) 309.
W. J. Scheule and V. D. Deetscreek, in “Ultrafine Particles”, edited by W. E. Kuhn (J Wiley, New York, 1963), p. 218.
C. R. Weast, (ed.), tin “Handbook of Chemistry and Physics”, 50th Edn (Chemical Rubber Company, Cleveland, OH, 1969) p. B232.
M. G. Segal and R. M. Sellers, J. Chem. Soc., Faraday Trans. I 78 (1982) 1149.
R. I. Haines and D. G. Owen, J. Phys. Chem. Solids 47 (1986) 647.
E. R. Vago, E. J. Calvo and M. Stratmann, Electrochimica Acta 39 (1994) 1655.
Y. Nishino, Y. Asakura, T. Sawa, S. Uchida, K. Ohsumi, S. Yosikawa, O. Amano and N. Suzuki, J. Nucl. Sci. Technol. 28 (1991) 848.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
McGARVEY, G.B., Owen, D.G. Control of the morphology and surface properties of nickel ferrite. Journal of Materials Science 33, 35–40 (1998). https://doi.org/10.1023/A:1004325108714
Issue Date:
DOI: https://doi.org/10.1023/A:1004325108714