Abstract
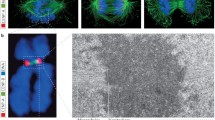
The chicken genome comprises 78 chromosomes which include several macrochromosomes and many microchromosomes. Very little information is currently available concerning chicken centromere structure and function and it is unclear if the two types of chromosomes share a common centromere mechanism or whether this mechanism resembles those in other species. Immunofluorescence studies using antibodies to mammalian constitutive centromere proteins CENP-A, CENP-B, and CENP-C and the passenger proteins CENP-E, and CENP-F revealed the presence of each of these proteins at the centromeres of both macro- and microchromsomes. CENP-A, CENP-B, and CENP-E levels showed variability between metaphase centromeres while CENP-C and CENP-F levels were relatively constant. These results suggest a common centromere mechanism for both types of chromosomes as well as indicating a high degree of conservation of individual proteins between widely divergent vertebrate classes and an overall conservation of centromere function throughout vertebrate evolution.
Similar content being viewed by others
References
Ainsztein AM, Kandels-Lewis SE, Mackay AM, Earnshaw WC (1998) INCENP centromere and spindle targeting: Identification of essential conserved motifs and involvement of heterochromatin protein HP1. J Cell Biol 143: 1763-1774.
Barbosa-Cisneros O, Fraire-Velazquez S, Moreno J, Herrera-Esparza R (1997) CENP-B autoantigen is a conserved protein from humans to higher plants: identification of the amino terminal domain in Phaseolus vulgaris. Rev Rhum Engl Ed 64: 368-374.
Choo KHA (1997a) The Centromere. Oxford, New York, Tokyo: Oxford University Press, pp. 304.
Choo KH (1997b) Centromere DNA dynamics: latent centromeres and neocentromere formation. Am J Hum Genet 61: 1225-1233.
Cooke CA, Heck MMS, Earnshaw WC (1987) The inner centromere protein (INCENP) antigens: Movement from inner centromere to midbody during mitosis. J cell Biol 105: 2053-2067.
du Sart D, Cancilla MR, Earle E et al. (1997) A functional neocentromere formed through activation of a latent human centromere and consisting of non-alpha satellite DNA. Nat Genet 16: 144-153.
Earnshaw WC, Bernat RL (1991) Chromosomal passengers: towards an integrated view of mitosis. Chromosoma 100: 139-146.
Faulkner NE, Vig B, Echeverri CJ, Wordeman L, Vallee RB (1998) Localization of motor-related proteins and associated complexes to active, but not inactive, centromeres. Hum Mol Genet 7: 671-677.
Fukagawa, T, Brown WRA (1997) Efficient conditional mutation of the vertebrate CENP-C gene. Hum Mol Genet 6: 2301-2308.
Hudson DF, Fowler KJ, Earle E et al. (1998) Centromere protein B null mice are mitotically and meiotically normal but have lower body weight and testis weights. J Cell Biol 141: 309-319.
Kalitsis P, Fowler KJ, Earle E, Hill J, Choo KH (1998) Targeted disruption of mouse centromere protein C gene leads to mitotic disarray and early embryo death. Proc Natl Acad Sci USA 95(3): 1136-1141.
Karpen GH, Allshire RC (1997) The case for epigenetic effects on centromere identity and function. Trends Genet 13: 489-496.
Kipling D, Warburton PE (1997) Centromeres, CENP-B and Tigger too. Trends Genet 13: 141-145.
Liao H, Winkfein RJ, Mack G, Rattner JB, Yen TJ (1995) CENP-F is a protein of the nuclear matrix that assembles onto kinetochores at late G2 and is rapidly degraded after mitosis. J Cell Biol 130: 507-518.
Mackay AM, Eckley DM, Chue C, Earnshaw WC (1993) Molecular analysis of the INCENPs (inner centromere proteins): separate domains are required for association with microtubules during interphase and with the central spindle during anaphase. J Cell Biol 123: 373-385.
Mackay AM, Ainsztein AM, Eckley DM, Earnshaw WC (1998) A dominant mutant of inner centromere protein (INCENP), a chromosomal protein, disrupts prometaphase congression and cytokinesis. J Cell Biol 140: 991-1002.
Masumoto H, Masukata H, Muro Y, Nozaki N, Okazaki T (1989) A human centromere antigen (CENP-B) interacts with a short specific sequence in alphoid DNA, a human centromeric satellite. J Cell Biol 109: 1963-1973.
McQueen HA, Fantes J, Cross SH, Clark VH, Archibald AL, Bird AP (1996) CpG islands of chicken are concentrated on microchromosomes. Nat Genet 12: 321.
McQueen HA, Siriaco G, Bird AP (1998) Chicken microchromosomes are hyperacetylated, early replicating, and gene rich. Genome Res 8: 621-630.
Meluh PB, Yang P, Glowczewski L, Koshland D, Smith MM (1998) Cse4p is a component of the core centromere of Saccharomyces cerevisiae. Cell 94: 607-613.
Page SL, Earnshaw WC, Choo KHA, Shaffer LG (1995) Further evidence that CENP-C is a necessary component of active centromeres: studies of a dic(X;15) with simultaneous immunofluorescence and FISH. Hum Mol Gen 4: 289.
Perez-Castro AV, Shamanski FL, Meneses JJ et al. (1998) Centromeric protein B null mice are viable with no apparent abnormalities. Dev Biol 201(2): 135-143.
Saffery R, Irvine DV, Kile BT, Hudson DF, Cutts S, Choo KHA (1999) Cloning, expression, and promoter structure of a mammalian Inner Centromere Protein (INCENP). Mamm Genome 4: 415-418.
Solovei IV, Joffe BI, Gaginskaya ER, Macgregor HC (1996) Transcription of lampbrush chromosomes of a centromerically localized highly repeated DNA in pigeon (Columba) relates to sequence arrangement. Chromosome Res 4: 588-603.
Stukenburg BT, Lustig KD, McGarry TJ, King RW, Kuang J, Kirschner MW (1997) Systematic identification of mitotic phosphoproteins. Curr Biol 7: 338-348.
Thrower DA, Jordan MA, Schaar BT, Yen TJ, Wilson L (1995) Mitotic HeLa cells contain a CENP-E-associated minus end-directed microtubule motor. EMBO J 14: 918-926.
Tomkiel J, Cooke CA, Saitoh H, Bernat RL, Earnshaw WC (1994) CENP-C is required for maintaining proper kinetochore size and for a timely transition to anaphase. J Cell Biol 125: 531-545.
Warburton PE, Cooke CA, Bourassa S et al. (1997) Immunolocalization of CENP-A suggests a distinct nucleosome structure at the inner kinetochore plate of active centromeres. Curr Biol 7: 901-904.
Wood KW, Sakowicz R, Goldstein LS, Cleveland DW (1997) CENP-E is a plus end-directed kinetochore motor required for metaphase chromosome alignment. Cell 91: 357-366.
Zhu X, Mancini MA, Chang KH et al. (1995) Characterization of a novel 350-kilodalton nuclear phosphoprotein that is specifically involved in mitotic-phase progression. Mol Cell Biol 15: 5017-5029.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Saffery, R., Earle, E., Irvine, D.V. et al. Conservation of Centromere Proteins in Vertebrates. Chromosome Res 7, 261–265 (1999). https://doi.org/10.1023/A:1009222729850
Issue Date:
DOI: https://doi.org/10.1023/A:1009222729850