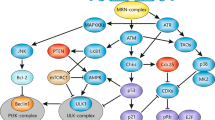
Abstract
Because of the singular importance of DNA for genetic inheritance, all organisms have evolved mechanisms to recognize and respond to DNA damage. In metazoans, cells can respond to DNA damage either by undergoing cell cycle arrest, to facilitate DNA repair, or by undergoing cell suicide. Cell death can either occur by activation of the apoptotic machinery or simply be a consequence of irreparable damage that prevents further cell division. In germ cells, mechanisms for limiting alterations to the genome are required for faithful propagation of the species whereas in somatic cells, responses to DNA damage prevent the accumulation of mutations that might lead to aberrant cell proliferation or behavior. Several of the genes that regulate cellular responses to DNA damage function as tumor suppressors. The clinical use of DNA damaging agents in the treatment of cancer can activate these tumor suppressors and exploits the cellular suicide and growth arrest mechanisms that they regulate. It appears that in some but not all types of tumors the propensity to undergo apoptosis is a critical determinant of their sensitivity to anti-cancer therapy. This review describes current understanding of the molecular control of DNA damage-induced apoptosis with particular attention to its role in tumor suppression and cancer therapy.
Similar content being viewed by others
References
Singer B, Kusmierek JT. Chemical mutagenesis. Annual Review of Biochemistry1982;51:655–693.
Friedberg EC. DNA Repair, New York: WH Freeman and Company, 1985.
Kornberg A, Baker TA. DNA Replication, New York: WH Freeman and Company, 1992.
Freeman BA, Crapo JD. Biology of disease: Free radicals and tissue injury. Laboratory Investigation1982;47:412–426.
Tonegawa S. Somatic generation of antibody diversity. Nature1983;302:575–581.
Davis MM. T cell receptor gene diversity and selection. Annu Rev Biochem1990;59:475–496.
Hartwell LH, Weinert TA. Checkpoints: Controls that ensure the order of cell cycle events. Science1989;246:629–634.
Weinert T, Lydall D. Cell cycle checkpoints, genetic instability and cancer. Cancer Biol1993;4:129–140.
Kastan MB, Zhan Q, El-Deiry WS, et alA mammalian cell cycle checkpoint pathway utilizing p53 and TIGADD45 is defective in ataxia-telangiectasia. Cell1992;71: 587–597.
Cohen JJ, Duke RC, Fadok VA, Sellins KS. Apoptosis and programmed cell death in immunity. Annual Review of Immunology1992;10:267–293.
Brown JM, Wouters BG. Apoptosis, p53, and tumor cell sensitivity to anticancer agents. Cancer Research1999;59: 1391–1399.
Kerr JFR, Wyllie AH, Currie AR. Apoptosis: A basic biological phenomenon with wide-ranging implications in tissue kinetics. British Journal of Cancer1972;26:239–257.
Wang JYJ. Cellular responses to DNA damage. Current Opinion in Cell Biology1998;10:240–247.
Xu Y, Ashley T, Brainerd EE, Bronson RT, Meyn MS, Baltimore D. Targeted disruption of TIATM leads to growth retardation, chromosomal fragmentation during meiosis, immune defects, and thymic lymphoma. Genes and Development1996;10:2411–2422.
Chen PC, Lavin MF, Kidson C, Moss D. Identification of ataxia telangiectasia heterozygotes, a cancer prone population. Nature1978;274:484–486.
Taylor AM, Harnden DG, Arlett CF, et alAtaxia telangiectasia: A human mutation with abnormal radiation sensitivity. Nature1975;258:427–429.
Lavin MF, Shiloh Y. The genetic defect in ataxiatelangiectasia. Annual Review of Immunology1997;15:177–202.
Houldsworth J, Lavin MF. Effect of ionizing radiation on DNA synthesis in ataxia telangiectasia cells. Nucleic Acids Research1980;8:3709–3720.
Morgan SE, Kastan MB. p53 and ATM: Cell cycle, cell death, and cancer. Advances in Cancer Research1997;71:1–25.
Oren M. Regulation of the p53 tumor suppressor protein. Journal of Biological Chemistry1999;274: 36031–36034.
Banin S, Moyal L, Shieh S-Y. Enhanced phosphorylation of p53 by ATM in response to DNA damage. Science1998;281: 1674–1677.
Matsuoka S, Huang M, Elledge SJ. Linkage of ATM to cell cycle regulation by the Chk2 protein kinase. Science1998;282:1893–1897.
Chehab NH, Malikzay A, Appel M, Halazonetis TD. Chk2/hCds1 functions as a DNA damage checkpoint in G1 by stabilizing p53. Genes and Development2000;14: 278–288.
Shieh SY, Ahn J, Tamai K, Taya Y, Prives C. The human homologs of checkpoint kinases chk1 and cds1 (Chk2) phosphorylate p53 at multipleDNAdamage-inducible sites. Genes and Development2000;14:289–300.
Hirao A, Kong YY, Matsuoka S, et alDNA damage-induced activation of p53 by the checkpoint kinase Chk2. Science2000; 287:1824–1827.
Unger T, Juven-Gershon T, Moallem E, et alCritical role for Ser20 of human p53 in the negative regulation of p53 by Mdm2. EMBO Journal1999;18:1805–1814.
Chehab NH, Malikzay A, Stavridi ES, Halazonetis TD. Phosphorylation of Ser-20 mediates stabilization of human p53 in response toDNAdamage. Proceedings of the National Academy of Sciences of the United States of America1999;96: 13777–13782.
Brown EJ, Baltimore D. TIATR disruption leads to chromosomal fragmentation and early embryonic lethality. Genes and Development2000;14:397–402.
Tibbetts RS, Brumbaugh KM, Williams JM, et alA role for ATR in the DNA damage-induced phosphorylation of p53. Genes and Development1999;13:152–157.
Liu Q, Guntuku S, Cui XS. Chk1 is an essential kinase that is regulated by Atr and required for the G2/M DNA damage checkpoint. Genes and Development2000;14:1448–1459.
Takai H, et alAberrant cell cycle checkpoint function and early embryonic death in Chk1 mice. Genes and Development2000;14:1439–1447.
Donehower LA, Harvey M, Slagle BL, et alMice deficient for p53 are developmentally normal but are susceptible tospontaneous tumours. Nature1992;356:215–221.
Bosma GC, Custer RP, Bosma MJ. A severe combined immunodeficiency mutation in the mouse. Nature1983;301: 527–530.
Peterson SR, Kurimasa A, Oshimura M, Dynan WS, Bradbury EM, Chen DJ. Loss of the catalytic subunit of the DNAdependent protein kinase inDNAdouble-strand-break-repair mutant mammalian cells. Proceedings of the National Academy of Sciences of the United States of America1995;92:3171–3174.
Blunt T, Gell D, Fox M, et alIdentification of a nonsense mutation in the carboxyl-terminal region of DNA-dependent protein kinase catalytic subunit in the scid mouse. Proceedings of the National Academy of Sciences of the United States of America1996;93: 10285–10290.
Danska JS, Holland DP, Mariathasan S, Williams KM, Guidos CJ. Biochemical and genetic defects in the DNA-dependent protein kinase in murine lymphocytes. Molecular and Cellular Biology1996;16:5507–5517.
Gao Y, Chaudhuri J, Zhu C, Davidson L, Weaver DT, Alt FW. A targeted DNA-PKcs-null mutation reveals DNA-PKindependent functions for KU in V(D)J recombination. Immunity1998;9:367–376.
Taccioli GE, Amatucci AG, Beamish HJ. Targeted disruption of the catalytic subunit of the DNA-PK gene in mice confers severe combined immunodeficiency and radiosensitivity. Immunity1998;9:355–366.
Schuler W, Weiler IJ, Schuler A, et alRearrangement of antigen receptor genes is defective in mice with severe combined immune deficiency. Cell1986;46:963–972.
Fulop GM, Phillips RA. The scid mutation in mice causes a general defect in DNA repair. Nature1990;347: 479–482.
Gu Y, Seidl KJ, Rathbun GA. Growth retardation and leaky SCID phenotype of Ku70-deficient mice. Immunity1997;7: 653–665.
Ouyang H, et alKu70 is required for DNA repair but not for T cell antigen receptor gene recombination in vivo. Journal of Experimental Medicine1997;186:921–929.
Nussenzweig A, Chen C, da Costa Soares V, et alRequirement for Ku80 in growth and immunoglobulin V(D)J recombination. Nature1996;382:551–555.
Biedermann KA, Sun JR, Giaccia AJ, Tosto LM, Brown JM. mutation in mice confers hypersensitivity to ionizing radiation and a deficiency in DNA double-strand break repair. Proceedings of the National Academy of Sciences of the United States of America1991;88:1394–1397.
Hendrickson EA, Qin XQ, Bump EA, Schatz DG, Oettinger M, Weaver DT. A link between double-strand break-related repair and V(D)J recombination: The scid mutation. Proceedings of the National Academy of Sciences of the United States ofAmerica1991;88:4061–4065.
Nussenzweig A, Sokol K, Burgman P, Li L, Li GC. Hypersensitivity of Ku80-deficient cell lines and mice toDNAdamage: The effects of ionizing radiation on growth, survival, and development. Proceedings of the National Academy of Sciences of the United States of America1997;94: 13588–13593.
Takata M, Sasaki MS, Sonoda E, et alHomologous recombination and non-homologous end-joining pathways of DNA double-strand break repair have overlapping roles in the maintenance of chromosomal integrity in vertebrate cells. Embo Journal1998;17:5497–5508.
Van Dyck E, Stasiak AZ, Stasiak A, West SC. Binding of double-strand breaks in DNA by human Rad52 protein. Nature1999;398:728–731.
Kolodner R. Biochemistry and genetics of eukaryotic mismatch repair. Genes and Development1996;10:1433–1442.
Kuerbitz SJ, Plunkett BS, Walsh WV, Kastan MB. Wildtype p53 is a cell cycle checkpoint determinant following irradiation. Proc Natl Acad Sci USA1992;89:7491–7495.
Livingstone LR, White A, Sprouse J, Livanos E, Jacks T, Tlsty TD. Altered cell cycle arrest and gene amplification potential accompany loss of wild-type p53. Cell1992;70:923–935.
Yin Y, Tainsky MA, Bischoff FZ, Strong LC, Wahl GM.Wildtype p53 restores cell cycle control and inhibits gene amplification in cells with mutant p53 alleles. Cell1992;70: 937–948.
El-Deiry WS, Tokino T, Velculescu VE. TIWAF1, a potential mediator of p53 tumor suppression. Cell1993;75:817–825.
Brugarolas J, Chandrasekaran C, Gordon JI, Beach D, Jacks T, Hannon GJ. Radiation-induced cell cycle arrest compromised by p21 deficiency. Nature1995;377:552–557.
Deng C, Zhang P, Harper JW, Elledge SJ, Leder P. Mice lacking p21CIP1/WAF1 undergo normal development, but are defective in G1 checkpoint control. Cell1995;82: 675–684.
Tanaka N, et alCooperation of the tumour suppressors IRF-1 and p53 in response to DNA damage. Nature1996;382: 816–818.
Peng C-Y, Graves PR, Thoma RS, Wu Z, Shaw AS, Piwnica-Worms H. Mitotic and G2 checkpoint control: Regulation of 14–3–3 protein binding by phosphorylation of Cdc25C on Serine-216. Science1997;277:1501–1505.
Sanchez Y, Wong C, Thoma RS, et alConservation of the Chk1 checkpoint pathway in mammals: Linkage of DNA damage to Cdk regulation through Cdc25. Science1997;277: 1497–1501.
Furnari B, Rhind N, Russell P. Cdc25 mitotic inducer targeted by Chk1 DNA damage checkpoint kinase. Science1997;277:1495–1497.
Draetta G, Eckstein J. Cdc25 protein phosphatases in cell proliferation. Biochimica et Biophysica Acta1997;1332: M53-M63.
Roberts JM. Evolving ideas about cyclins. Cell1999;98: 129–132.
Bunz F, Dutriaux A, Lengauer C, et alRequirement for p53 and p21 to sustain G2 arrest after DNA damage. Science1998; 282:1497–1501.
Waldman T, Lengauer C, Kinzler KW, Vogelstein B. Uncoupling of S phase and mitosis induced by anticancer agents in cells lacking p21. Nature1996;381:713–716.
Clarke AR, Purdie CA, Harrison DJ, et alThymocyte apoptosis induced by p53-dependent and independent pathways. Nature1993;362:849–852.
Lowe SW, Schmitt EM, Smith SW, Osborne BA, Jacks T. p53 is required for radiation-induced apoptosis in mouse thymocytes. Nature1993;362:847–849.
Strasser A, Harris AW, Jacks T, Cory S. DNA damage can induce apoptosis in proliferating lymphoid cells via p53-independent mechanisms inhibitable by Bcl-2. Cell1994;79: 329–339.
Clarke AR, Gledhill S, Hooper ML, Bird CC, Wyllie AH. p53 dependence of early apoptotic and proliferative responses within the mouse intestinal epithelium following-irradiation. Oncogene1994;9:1767–1773.
Merritt AJ, Potten CS, Kemp CJ, et alThe role of p53 in spontaneous and radiation-induced apoptosis in the gastrointestinal tract of normal and p53-deficient mice. Cancer Research1994;54:614–617.
Lotem J, Sachs L. Hematopoietic cells from mice deficient in wild-type p53 are more resistant to induction of apoptosis by some agents. Blood1993;82:1092–1096.
Hasegawa M, Zhang Y, Niibe H, Terry NH, Meistrich ML. Resistance of differentiating spermatogonia to radiationinduced apoptosis and loss in p53-deficient mice. Radiation Research1998;149:263–270.
Wood KA, Youle RJ. The role of free radicals and p53 in neuron apoptosis in vivo. Journal of Neuroscience1995;15: 5851–5857.
Herzog K-H, Chong MJ, Kapsetaki M, Morgan JI, McKinnon PJ. Requirement for Atm in ionizing radiation-induced cell death in the developing central nervous system. Science1998; 280:1089–1091.
Merritt AJ, Allen TD, Potten CS, Hickman JA. Apoptosis in small intestinal epithelia from p53-null mice: Evidence for a delayed, p53-independent G2/M-associated cell death after-irradiation. Oncogene1997;14:2759–2766.
Tamura T, Ishihara M, Amphier MS. An IRF-1-dependent pathway of DNA damage-induced apoptosis in mitogenactivated T lymphocytes. Nature1995;376:596–599.
Gottlieb TM, Oren M. p53 and apoptosis. Seminars in Cancer Biology1998;8:359–368.
Vaux DL, Haecker G, Strasser A. An evolutionary perspective on apoptosis. Cell1994;76:777–779.
Steller H. Mechanisms and genes of cellular suicide. Science1995;267:1445–1449.
Ellis RE, Yuan J, Horvitz HR. Mechanisms and functions of cell death. Annual Review of Cell Biology1991;7: 663–698.
Thornberry NA, Lazebnik Y. Caspases: Enemies within. Science1998;281:1312–1316.
Cryns V, Yuan J. Proteases to die for. Genes and Development1998;12:1551–1570.
Strasser A, O'Connor L, Dixit VM. Apoptosis signaling. Annual Review of Biochemistry2000;69:217–245.
Salvesen GS, Dixit VM. Caspase activation: The inducedproximity model. Proceedings of the National Academy of Sciences of the United States of America1999;96: 10964–10967.
Muzio M, et alFLICE, a novel FADD homologous ICE/CED-3-like protease, is recruited to the CD95 (Fas/Apo-1) deathinducing signaling complex. Cell1996;85:817–827.
Boldin MP, Goncharov TM, Goltsev YV, Wallach D. Involvement ofMACH,a novelMORT1/FADD-interacting protease, in Fas/APO-1-and TNF receptor-induced cell death. Cell1996;85:803–815.
Komoriya A, Packard BZ, Brown MJ, Wu ML, Henkart PA. Assessment of caspase activities in intact apoptotic thymocytes using cell-permeable fluorogenic caspase substrates. Journal of Experimental Medicine2000;191:1819–1828.
Tanaka M, Itai T, Adachi M, Nagata S. Downregulation of Fas ligand by shedding. Nature Medicine1998;4:31–36.
Schneider P, Holler N, Bodmer JL, et alConversion of membrane-bound Fas(CD95) ligand to its soluble form is associated with downregulation of its proapoptotic activity and loss of liver toxicity. Journal of Experimental Medicine1998; 187:1205–1213.
Huang DC, Hahne M, Schroeter M, et alActivation of Fas by FasL induces apoptosis by a mechanism that cannot be blocked by Bcl-2 or Bcl-xL. Proceedings of the National Academy of Sciences of the USA1999;96: 14871–14876.
Hsu H, Xiong J, Goeddel DV. The TNF receptor 1-associated proteinTRADDsignals cell death andNF-KBactivation. Cell1995;81:495–504.
Boldin MP, Varfolomeev EE, Pancer Z, Mett IL, Camonis JH, Wallach D. A novel protein that interacts with the death domain of Fas/APO1 contains a sequence motif related to the death domain. Journal of Biological Chemistry1995;270: 7795–7798.
Chinnaiyan AM, O'Rourke K, Tewari M, Dixit VM. FADD, a novel death domain-containing protein, interacts with the death domain of Fas and initiates apoptosis. Cell1995;81: 505–512.
Muzio M, Stockwell BR, Stennicke HR, Salvesen GS, Dixit VM. An induced proximity model for caspase-8 activation. Journal of Biological Chemistry1998;273:2926–2930.
Martin DA, Siegel RM, Zheng L, Lenardo MJ. Membrane oligomerization and cleavage activates the caspase-8 (FLICE/MACH1) death signal. Journal of Biological Chemistry1998;273:4345–4349.
Kischkel FC, Hellbardt S, Behrmann I, et alCytotoxicitydependent APO-1 (Fas/CD95)-Associated proteins form a death-inducing signaling complex (DISC) with the receptor. EMBO Journal1995;14:5579–5588.
Medema JP, Scaffidi C, Kischkel FC, et alFLICE is activated by association with the CD95 death-inducing signaling complex (DISC). EMBO Journal1997;16:2794–2804.
Newton K, Harris AW, Bath ML, Smith KGC, Strasser A. A dominant interfering mutant of FADD/Mort1 enhances deletion of autoreactive thymocytes and inhibits proliferation of mature T lymphocytes. EMBO Journal1998;17: 706–718.
Yeh WC, Pompa JL, McCurrach ME. FADD: Essential for embryo development and signaling from some, but not all, inducers of apoptosis. Science1998;279:1954–1958.
Zhang J, Cado D, Chen A, Kabra NH, Winoto A. Fasmediated apoptosis and activation-induced T-cell proliferation are defective in mice lacking FADD/Mort1. Nature1998; 392:296–300.
Smith KGC, Strasser A, Vaux DL. CrmA expression in T lymphocytes of transgenic mice inhibits CD95 (Fas/APO-1)-transduced apoptosis, but does not cause lymphadenopathy or autoimmune disease. EMBO Journal1996;15:5167–5176.
Varfolomeev EE, Schuchmann M, Luria M. Targeted disruption of the mouse Caspase 8 gene ablates cell death induction by the TNF receptors, Fas/Apo1, and DR3 and is lethal prenatally. 1998;9:267–276.
Hsu H, Shu H-B, Pan M-G, Goeddel DV. TRADD-TRAF2 and TRADD-FADD interactions define two distinct TNF Receptor 1 signal transduction pathways. Cell1996;84: 299–308.
Bodmer JL, Holler N, Reynard S, et alTRAIL receptor-2signals apoptosis through FADD and caspase-8. Nature Cell Biology2000;2:241–243.
Kischkel FC, Lawrence DA, Chuntharapai A, Schow P, Kim KJ, Ashkenazi A. Apo2L/TRAIL-dependent recruitment of endogenous FADD and caspase-8 to death receptors 4 and 5. Immunity2000;12:611–620.
Sprick MR, Weigand MA, Rieser E, et alFADD/MORT1 and caspase-8 are recruited to TRAIL receptors 1 and 2 and are essential for apoptosis mediated by TRAIL receptor 2. Immunity2000;12:599–609.
Vanhaesebroeck B, Reed JC, de Valck D, et alEffect of bcl-2 proto-oncogene expression on cellular sensitivity to tumor necrosis factor-mediated cytotoxicity. Oncogene1993;8: 1075–1081.
Strasser A, Harris AW, Huang DCS, Krammer PH, Cory S. Bcl-2 and Fas/APO-1 regulate distinct pathways to lymphocyte apoptosis. EMBO Journal1995;14:6136–6147.
Traver D, Akashi K, Weissman IL, Lagasse E. Mice defective in two apoptosis pathways in the myeloid lineage develop acute myeloblastic leukemia. Immunity1998;9:47–57.
Newton K, Strasser A. Ionizing radiation and chemotherapeutic drugs induce apoptosis in lymphocytes in the absence of fas or FADD/MORT1 signaling: Implications for cancer therapy. Journal of Experimental Medicine2000;191:195–200.
Villunger A, O'Reilly LA, Holler N, Adams JM, Strasser A. Fas ligand, Bcl-2, G-CSF and p38 MAPK-Regulators of distinct cell death and survival pathways in granulocytes. Journal of Experimental Medicine, 2000;192:647–657.
Adams JM, Cory S. The Bcl-2 protein family: Arbiters of cell survival. Science1998;281:1322–1326.
Vaux DL, Cory S, Adams JM. Bcl-2 gene promotes haemopoietic cell survival and cooperates with c-myc to immortalize pre-B cells. Nature1988;335:440–442.
Oltvai ZN, Korsmeyer SJ. Checkpoints of dueling dimers foil death wishes. Cell1994;79:189–192.
Cecconi F, Alvarez-Bolado G, Meyer BI, Roth KA, Gruss P. Apaf-1 (CED-4 homologue) regulates programmed cell death in mammalian development. Cell1998;94:727–737.
Yoshida H, Kong Y-Y, Yoshida R, et alApaf1 is required for mitochondrial pathways of apoptosis and brain development. Cell1998;94:739–750.
Sentman CL, Shutter JR, Hockenbery D, Kanagawa O, Korsmeyer SJ. bcl-2 inhibits multiple forms of apoptosis but not negative selection in thymocytes. Cell1991;67: 879–888.
Strasser A, Harris AW, Cory S. Bcl-2 transgene inhibits T cell death and perturbs thymic self-censorship. Cell1991;67:889–899.
Lagasse E, Weissman IL. Enforced expression of Bcl-2 in monocytes rescues macrophages and partially reverses osteopetrosis in op/op mice. Cell1997;89:1021–1031.
Akashi K, Kondo M, von Freeden-Jeffry U, Murray R, Weissman IL. Bcl-2 rescues T lymphopoiesis in interleukin-7 receptor-deficient mice. Cell1997;89:1033–1041.
Maraskovsky E, O'Reilly LA, Teepe M, Corcoran LM, Peschon JJ, Strasser A. Bcl-2 can rescue T lymphocyte development in interleukin-7 receptor-deficient mice but not in mutant mice. Cell1997;89:1011–1019.
Ogilvy S, Metcalf D, Print CG, Bath ML, Harris AW, Adams JM. Constitutive bcl-2 expression throughout the hematopoietic compartment affects multiple lineages and enhances progenitor cell survival. Proceedings of the National Academy of Sciences of the United States of America1999;96: 14943–14948.
O'Connor L, Strasser A, O'Reilly LA, et al. Bim: A novel member of the Bcl-2 family that promotes apoptosis. EMBO Journal1998;17:384–395.
Bouillet P, Metcalf D, Huang DCS, et alProapoptotic Bcl-2 relative Bim required for certain apoptotic responses, leukocyte homeostasis, and to preclude autoimmunity. Science1999; 286:1735–1738.
Green DR, Reed JC. Mitochondria and apoptosis. Science1998;281:1309–1311.
Gross A, McDonnell JM, Korsmeyer SJ. BCL-2 family members and the mitochondria in apoptosis. Genes and Development1999;13:1899–1911.
Zou H, Henzel WJ, Liu X, Lutschg A, Wang X. Apaf-1, a human protein homologous to C. elegans CED-4, participates in cytochrome c-dependent activation of Caspase-3. Cell1997; 90:405–413.
Li P, Nijhawan D, Budihardjo I, et alCytochrome c and dATP-dependent formation of Apaf-1/Caspase-9 complex initiates an apoptotic protease cascade. Cell1997;91:479–489.
Antonsson B, et alInhibition of Bax channel-forming activity by Bcl-2. Science1997;277:370–372.
Yang J, Liu XS, Bhalla K, et alPrevention of apoptosis by Bcl-2-Release of cytochrome c from mitochondria blocked. Science1997;275:1129–1132.
Kluck RM, Bossy-Wetzel E, Green DR, Newmeyer DD. The release of cytochrome c from mitochondria-A primary site for Bcl-2 regulation of apoptosis. Science1997;275: 1132–1136.
Shimizu S, Narita M, Tsujimoto Y. Bcl-2 family proteins regulate the release of apoptogenic cytochrome c by the mitochondrial channel VDAC. Nature1999;399:483–487.
Chinnaiyan AM, O'Rourke K, Lane BR, Dixit VM. Interaction of CED-4 with CED-3 and CED-9: A molecular framework for cell death. Science1997;275:1122–1126.
Wu DY, Wallen HD, Nuñez G. Interaction and regulation of subcellular localization of CED-4 by CED-9. Science1997; 275:1126–1129.
Spector MS, Desnoyers S, Hoeppner DJ, Hengartner MO. Interaction between the C. elegans cell-death regulators CED-9 and CED-4. Nature1997;385:653–656.
Imaizumi K, Tsuda M, Imai Y, Wanaka A, Takagi T, Tohyama M. Molecular cloning of a novel polypeptide, DP5, induced during programmed neuronal death. Journal of Biological Chemistry1997;272: 18842–18848.
Conradt B, Horvitz HR. The C. elegans protein EGL-1 is required for programmed cell death and interacts with the Bcl-2-like protein CED-9. Cell1998;93:519–529.
Zha J, Harada H, Yang E, Jockel J, Korsmeyer SJ. Serine phosphorylation of death agonist BAD in response to survival factor results in binding to 14–3–3 not BCL-XL. Cell1996; 87:619–628.
Gross A, Jockel J, Wei MC, Korsmeyer SJ. Enforced dimerization of BAX results in its translocation, mitochondrial dysfunction and apoptosis.EMBOJournal1998;17:3878–3885.
Puthalakath H, Huang DCS, O'Reilly LA, King SM, Strasser A. The pro-apoptotic activity of the Bcl-2 family member Bim is regulated by interaction with the dynein motor complex. Molecular Cell1999;3:287–296.
Davidson FF, Steller H. Blocking apoptosis prevents blindness in Drosophila retinal degeneration mutants. Nature1998;391: 587–591.
Sugimoto A, Friesen PD, Rothman JH. Baculovirus p35 prevents developmentally programmed cell death and rescues a ced-9 mutant in the nematode Caenorhabditis elegans. EMBO Journal1994;13:2023–2028.
Hisahara S, Araki T, Sugiyama F. Targeted expression of baculovirus p35 caspase inhibitor in oligodendrocytes protects mice against autoimmune-mediated demyelination. EMBO Journal2000;19:341–348.
Hengartner MO. Death cycle and Swiss army knives. Nature1998;391:441–442.
Fulda S, Scaffidi C, Pietsch T, Krammer PH, Peter ME, Debatin K-M. Activation of the CD95 (APO-1/Fas) pathway in drug-and °-irradiation-induced apoptosis of brain tumor cells. Cell Death and Differentiation1998;5:884–893.
Sheard MA, Vojtesek B, Janakova L, Kovarik J, Zaloudik J. Up-regulation of Fas (CD95) in human p53(wild-type) cancer cells treated with ionizing radiation. International Journal of Cancer1997;73:757–762.
Fulda S, Sieverts H, Friesen C, Herr I, Debatin K-M. The CD95 (APO-1/Fas) system mediates drug-induced apoptosis in neuroblastoma cells. Cancer Research1997;57:3823–3829.
Friesen C, Herr I, Krammer PH, Debatin K-M. Involvement of the CD95 (APO-1/Fas) receptor/ligand system in druginduced apoptosis in leukemia cells. Nature Medicine1996;2: 574–577.
Kasibhatla S, Brunner T, Genestier L, Echeverri F, Mahboubi A, Green DR. DNA damaging agents induce expression of Fas ligand and subsequent apoptosis in T lymphocytes via the activation of NF-B and AP-1. Molecular Cell1998;1: 543–551.
Srivastava RK, Sasaki CY, Hardwick JM, Longo DL. Bcl-2-mediated drug resistance: Inhibition of apoptosis by blocking nuclear factor of activated T lymphocytes (NFAT)-induced fas ligand transcription. Journal of Experimental Medicine1999; 190:253–266.
Micheau O, Solary E, Hammann A, Dimanche-Boitrel M-T. Fas ligand-independent, FADD-mediated activation of the Fas death pathway by anticancer drugs. Journal of Biological Chemistry1999;274:7987–7992.
Posovszky C, Friesen C, Herr I, Debatin K-M. Chemotherapeutic drugs sensitize pre-B ALL cells for CD95-and cytotoxic T-lymphocyte-mediated apoptosis. Leukemia1999;13: 400–409.
M¨ uller M, Wilder, S., Bannasch, D. p53 activates the CD95 (APO-1/Fas) gene in response to DNA damage by anticancer drugs. Journal of Experimental Medicine1998;188:2033–2045.
Reinke V, Lozano G. The p53 targets mdm2 and Fas are not required as mediators of apoptosis in vivo. Oncogene1997;15: 1527–1534.
Wu GS, et al. KILLER/DR5 is aDNAdamage-inducible p53-regulated death receptor gene. Nature Genetics1997;17: 141–143.
Herr I, Wilhelm D, B¨ohler T, Angel P, Debatin K-M. Activation of CD95 (APO-1/Fas) signaling by ceramide mediates cancer therapy-induced apoptosis. EMBO Journal1997;16: 6200–6208.
Kolbus A, Herr I, Schreiber M, Debatin KM, Wagner EF, Angel P. c-jun-dependent CD95-L expression is a ratelimiting step in the induction of apoptosis by alkylating agents. Molecular and Cellular Biology2000;20:575–582.
Gamen S, Anel A, Lasierra P, et alDoxorubicin-induced apoptosis in human T-cell leukemia is mediated by caspase-3 activation in a Fas-independent way. FEBS Letters1997;417: 360–364.
McGahon AJ, Costa Pereira AP, Daly L, Cotter TG. Chemotherapeutic drug-induced apoptosis in human leukaemic cells is independent of the Fas (APO-1/CD95) receptor/ligand system. British Journal of Haematology1998;101:539–547.
Ruiz-Ruiz MC, Lopez-Rivas A. p53-mediated up-regulation of CD95 is not involved in genotoxic drug-induced apoptosis of human breast tumor cells. Cell Death and Differentiation1999;6:271–280.
Fiedler P, Schaetzlein CE, Eibel H. Constitutive expression of Fas ligand in thyrocytes. Science1998;279: http://www.sciencemag.org/content/vol279/issue5359/.
Stokes TA, Rymaszewski M, Arscott PL, et al. Constitutive expression of Fas ligand in thyrocytes. Science1998;279: http://www.sciencemag.org/content/vol279/issue5359/.
Smith D, Sieg S, Kaplan D. Technical note: Aberrant detection of cell surface Fas ligand with anti-peptide antibodies. Journal of Immunology1998;160:4159–4160.
Baker Jr JR, Bretz JD. Specificity questions concerning the clone 33 anti-fas ligand antibody. Cell Death and Differentiation2000;7:8–9.
Herr I, Posovsky C, B¨ohler T, Debatin K-M. mAb 33 from Transduction Laboratories specifically binds to human CD95-L. Cell Death and Differentiation2000;7:129–130.
Fiedler P, Eibel H. Antibody mAb33 from Transduction Laboratories detects human CD95L in ELISA but not in immunoblots. Cell Death and Differentiation2000;7:126–128.
Villunger A, Egle A, Kos M, et alDrug-induced apoptosis is associated with enhanced Fas (Apo-1/CD95) ligand expression but occurs independently of Fas (Apo-1/CD95) signaling in human T-acute lymphatic leukemia cells. Cancer Research1997;57:3331–3334.
Eischen CM, Kottke TJ, Martins LM, et alComparison of apoptosis in wild-type and Fas-resistant cells-Chemotherapy-induced apoptosis is not dependent on Fas/Fas ligand interactions. Blood1997;90:935–943.
Landowski TH, Shain KH, Oshiro MM, Buyuksal I, Painter JS, Dalton WS. Myeloma cells selected for resistance to CD95-mediated apoptosis are not cross-resistant to cytotoxic drugs: Evidence for independent mechanisms of caspase activation. Blood1999;94:265–274.
Rehemtulla A, Hamilton CA, Chinnaiyan AM, Dixit VM. Ultraviolet radiation-induced apoptosis is mediated by activation of CD-95 (Fas/APO-1). Journal of Biological Chemistry1997;272: 25783–25786.
Thome M, et alViral FLICE-inhibitory proteins (FLIPs) prevent apoptosis induced by death receptors. Nature1997;386: 517–521.
Irmler M, et alInhibition of death receptor signals by cellular FLIP. Nature1997;388:190–194.
Enari M, Hug H, Nagata S. Involvement of an ICE-like protease in Fas-mediated apoptosis. Nature1995;375:78–81.
Kataoka T, Schroter M, Hahne M, et alFLIP prevents apoptosis induced by death receptors but not by perforin/granzyme B, chemotherapeutic drugs, and gamma irradiation. Journal of Immunology1998;161:3936–3942.
Villunger A, Huang DCS, Holler N, Tschopp J, Strasser A. Fas ligand-induced c-Jun kinase activation in lymphoid cells requires extensive receptor aggregation but is independent of DAXX, and Fas-mediated cell death does not involve DAXX, RIP or RAIDD. Journal of Immunology, 2000;165: 1337–1343.
Datta R, Kojima H, Banach D, et alActivation of a CrmAinsensitive, p35-sensitive pathway in ionizing radiationinduced apoptosis. Journal of Biological Chemistry1997;272: 1965–1969.
Strasser A, Harris AW, Bath ML, Cory S. Novel primitive lymphoid tumours induced in transgenic mice by cooperation between timyc and bcl-2. Nature1990;348:331–333.
Soengas MS, Alarc´on RM, Yoshida H, et alApaf-1 and caspase-9 in p53-dependent apoptosis and tumor inhibition. Science1999;284:156–159.
Watanabe-Fukunaga R, Brannan CI, Copeland NG, Jenkins NA, Nagata S. Lymphoproliferation disorder in mice explained by defects in Fas antigen that mediates apoptosis. Nature1992;356:314–317.
Fuchs EJ, McKenna KA, Bedi A. p53-dependent DNA damage-induced apoptosis requires Fas/APO-1-independent activation of CPP32. Cancer Research1997;57:2550–2554.
O'Connor L, Strasser A. Fas, p53, and apoptosis. Science1999; 284: 1430.
O'Connor L, Harris AW, Strasser A. CD95 (Fas/APO-1) and p53 signal apoptosis independently in diverse cell types. Cancer Research2000;60:1217–1220.
Takahashi T, Tanaka M, Brannan CI, et alGeneralized lymphoproliferative disease in mice, caused by a point mutation in the Fas ligand. Cell1994;76:969–976.
Newton K, Harris AW, Strasser A. FADD/MORT1 regulates the pre-TCR checkpoint and can function as a tumour suppressor. EMBO Journal2000;19:931–941.
Strasser A, Whittingham S, Vaux DL, et alEnforced TIBCL2 expression in B-lymphoid cells prolongs antibody responses and elicits autoimmune disease. Proceedings zof the National Academy of Sciences of the USA1991;88:8661–8665.
Domen J, Gandy KL, Weissman IL. Systemic overexpression of BCL-2 in the hematopoietic system protects transgenic mice from the consequences of lethal irradiation. Blood1998; 91:2272–2282.
Grillot DAM, Merino R, Nuñez G. Bcl-xL displays restricted distribution during T cell development and inhibits multiple forms of apoptosis but not clonal deletion in transgenic mice. Journal of Experimental Medicine1995;182:1973–1983.
Miyashita T, Reed JC. bcl-2 gene transfer increases relative resistance of S49.1 and WEHI17.2 lymphoid cells to cell death and DNA fragmentation induced by glucocorticoids and multiple chemotherapeutic drugs. Cancer Research1992; 52:5407–5411.
Gibson L,et al. bcl-w, a novel member of the bcl-2 family, promotes cell survival. Oncogene1996;13:665–675.
Huang DCS, Cory S, Strasser A. Bcl-2, Bcl-XL and adenovirus protein E1B19kD are functionally equivalent in their ability to inhibit cell death. Oncogene1997;14:405–414.
Veis DJ, Sorenson CM, Shutter JR, Korsmeyer SJ. Bcl-2-deficient mice demonstrate fulminant lymphoid apoptosis, polycystic kidneys, and hypopigmented hair. Cell1993;75: 229–240.
Ma A, Pena JC, Chang B, et alBclx regulates the survival of double-positive thymocytes. Proceedings of the National Academy of Sciences of the United States of America1995;92: 4763–4767.
Nakayama K-i, et alDisappearance of the lymphoid system in Bcl-2 homozygous mutant chimeric mice. Science1993;261: 1584–1588.
Pritchard DM, Print C, O'Reilly L, Adams JM, Potten CS, Hickman JA. Bcl-w is an important determinant of damageinduced apoptosis in epithelia of small and large intestine. Oncogene2000;19:3955–3959.
Wang Y, Szekely L, Okan I, Klein G, Wiman KG. Wildtype p53-triggered apoptosis is inhibited by bcl-2 in a vmyc-induced T-cell lymphoma line. Oncogene1993;8: 3427–3431.
Chiou S-K, Rao L, White E. Bcl-2 blocks p53-dependent apoptosis. Molecular, Cellular, Biology1994;14:2556–2563.
D'Sa-Eipper C, Subramanian T, Chinnadurai G. ibfl-1, a bcl-2 homologue, suppresses p53-induced apoptosis and exhibits potent cooperative transforming activity. Cancer Research1996;56:3879–3882.
Miyake H, Hanada N, Nakamura H. Overexpression of Bcl-2 in bladder cancer cells inhibits apoptosis induced by cisplatin and adenoviral-mediated p53 gene transfer. Oncogene1998;16: 933–943.
Memon SA, Moreno MB, Petrak D, Zacharchuk CM. Bcl-2 blocks glucocorticoid-but not Fas-or activation-induced apoptosis in a T cell hybridoma. Journal of Immunology1995; 115:4644–4652.
Chiu VK, Walsh CM, Liu CC, Reed JC, Clark WR. Bcl-2 blocks degranulation but not fas-based cell-mediated cytotoxicity. Journal of Immunology1995;154:2023–2032.
Reap EA, Felix NJ, Wolthusen PA, et al bcl-2 transgenic Lpr mice show profound enhancement of lymphadenopathy. Journal of Immunology1995;155:5455–5462.
Tamura A, Katsumata M, Greene MI, Yui K. Inhibition of apoptosis and augmentation of lymphoproliferation in bcl-2 transgenic Fas/Fas ligand-defective mice. Cellular Immunol1996;168:220–228.
Hakem R, et alDifferential requirement for caspase 9 in apoptotic pathways in vivo. Cell1998;94:339–352.
Kuida K, Haydar TF, Kuan C-Y, et alReduced apoptosis and cytochrome c-mediated caspase activation in mice lacking caspase 9. Cell1998;94:325–337.
Miyashita T, Reed JC. Tumor suppressor p53 is a direct transcriptional activator of the human bax gene. Cell1995;80: 293–299.
Kitada S, Krajewski S, Miyashita T, Krajewska M, Reed JC. radiation induces upregulation of Bax protein and apoptosis in radiosensitive cells in vivo Oncogene1996;12:187–192.
Schmidt T, Korner K, Karsunky H, Korsmeyer S, Muller R, Moroy T. The activity of the murine bax promoter is regulated by Sp1/3 and E-box binding proteins but not by p53. Cell Death and Differentiation1999;6:873–882.
Knudson CM, Tung KSK, Tourtellotte WG, Brown GAJ, Korsmeyer SJ. Bax-deficient mice with lymphoid hyperplasia and male germ cell death. Science1995;270:96–99.
Brady HJM, Salomns GS, Bobeldijk RC, Berns AJM. T cells from bax® transgenic mice show accelerated apoptosis in response to stimuli but do not show restored DNA damageinduced cell death in the absence of p53. EMBO Journal1996; 15:1221–1230.
Chong MJ, Murray MR, Gosink EC, et alAtm and bax cooperate in ionizing radiation-induced apoptosis in the central nervous system. Proceedings of the National Academy of Sciences of the United States of America2000;97:889–894.
Oda E, Ohki R, Murasawa H, et alNoxa, a BH3-only member of the bcl-2 family and candidate mediator of p53-induced apoptosis. Science2000;288:1053–1058.
Lowenthal JW, Harris AW. Activation of mouse lymphocytes inhibits induction of rapid cell death by X-irradiation. J Immunol1985;135:1119–1125.
Boise LH, Gonzalez-Garcia M, Postema CE, et al. bcl-x, a bcl-2-related gene that functions as a dominant regulator of apoptotic cell death. Cell1993;74:597–608.
Grumont RJ, Rourke IJ, O'Reilly LA, et alB lymphocytes differentially use the Rel and nuclear factor ·B1 (NF-B1) transcription factors to regulate cell cycle progression and apoptosis in quiescent and mitogen-activated cells. Journal of Experimental Medicine1998;187:663–674.
Grumont RJ, Rourke IJ, Gerondakis S. Rel-dependent induction of TIA1 transcription is required to protect B cells from antigen receptor ligation-induced apoptosis. Genes and Development1999;13:400–411.
O'Reilly LA, Cullen L, Visvader J, et al. The pro-apoptotic BH3-only protein Bim is expressed in hemopoietic, epithelial, neuronal and germ cells. American Journal of Pathology2000; 157:449–461.
Xu Y. ATM in lymphoid development and tumorigenesis. Advances in Immunology1999;72:179–189.
Barlow C, Hirotsune S, Paylor R. Atm-deficient mice: A paradigm of ataxia telangiectasia. Cell1996;86:159–171.
Elson A, Wang Y, Daugherty CJ, et alPleiotropic defects in ataxia-telangiectasia protein-deficient mice. Proceedings of the National Academy of Sciences of the United States of America1996; 93: 13084–13089.
Liao M-J, Van Dyke T. Critical role for Atm in suppressing TIV(D)J recombination-driven thymic lymphoma. Genes and Development1999;13:1246–1250.
Jhappan C, Morse HC, Fleischmann RD, Gottesman MM, Merlino G. DNA-PKcs: A T-cell tumour suppressor encoded at the mouse scid locus. Nature Genetics1997;17:483–486.
Li GC, Ouyang H, Li X, iet al. Ku70: A candidate tumor suppressor gene for murine T cell lymphoma. Molecular Cell1998;2:1–8.
Difilippantonio MJ, Zhu J, Chen HT, et alDNA repair protein Ku80 suppresses chromosomal aberrations and malignant transformation. Nature2000;404:510–514.
Kinzler KW, Vogelstein B. Lessons from hereditary colorectal cancer. Cell1996;87:159–170.
de Wind N, Dekker M, Berns A, Radman M, te Riele H. Inactivation of the mouse Msh2 gene results in mismatch repair deficiency, methylation tolerance, hyperrecombination, and predisposition to cancer. Cell1995;82:321–330.
Reitmair AH, iet al. MSH2 deficient mice are viable and susceptible to lymphoid tumours. Nature Genetics1995;11: 64–70.
Baker SM, et alMale mice defective in the DNA mismatch repair gene TIPMS2 exhibit abnormal chromosome synapsis in meiosis. Cell1995;82:309–319.
deWind N, et al. HNPCC-like cancer predisposition in mice through simultaneous loss of Msh3 and Msh6 mismatchrepair protein functions. Nature Genetics1999;23:359–362.
Bell DW, Varley TM, Szydlo TE. Heterozygous germ line hCHK2 mutations in Li-Fraumeni syndrome. Science1999; 286:2528–2531.
Hollstein M, Sidransky D, Vogelstein B, Harris CC. p53 mutations in human cancers. Science1991;253:49–53.
Levine AJ, Momand J, Finlay CA. The p53 tumour suppressor gene. Nature1991;351:453–456.
Malkin D, Li FP, Strong LC. Germ line p53 mutations in a familial syndrome of breast cancer, sarcomas, and other neoplasms. Science1990;250:1233–1238.
Jacks T, Remington L, Williams BO, et alTumor spectrum analysis in p53-mutant mice. Curr Biol1994;4:1–7.
Nacht M, Jacks T. V(D)J recombination is not required for the development of lymphoma in p53-deficient mice. Cell Growth and Differentiation1998;9:131–138.
McDonnell TJ, Korsmeyer SJ. Progression from lymphoid hyperplasia to high-grade malignant lymphoma in mice transgenic for the t(14;18). Nature1991;349:254–256.
Strasser A, Harris AW, Cory S. E-tibcl-2 transgene facilitates spontaneous transformation of early pre-B and immunoglobulin-secreting cells but not T cells. Oncogene1993;8:1–9.
Linette GP, Hess JL, Sentman CL, Korsmeyer SJ. Peripheral T-cell lymphoma in lckpr-bcl-2 transgenic mice. Blood1995; 86:1255–1260.
Hermeking H, Eick D. Mediation of c-Myc-induced apoptosis by p53. Science1994;265:2091–2043.
Nacht M, Strasser A, Chan YR, et alMutations in the p53 and SCID genes cooperate in tumorigenesis. Genes and Development1996;10:2055–2066.
Guidos CJ, Williams CJ, Grandal I, Knowles G, Huang MTF, Danska JS. V(D)J recombination activates a p53-dependent DNA damage checkpoint in scid lymphocyte precursors. Genes and Development1996;10:2038–2054.
Westphal CH, Schmaltz C, Rowan S, Elson A, Fisher DE, Leder P. Genetic interactions between atm and ip53 influence cellular proliferation and irradiation-induced cell cycle checkpoints. Cancer Research1997;57:1664–1667.
Gao Y, et alA critical role for DNA end-joining proteins in both lymphogenesis and neurogenesis. Cell1998;95: 891–902.
Frank KM, Sharpless NE, Gao Y. DNA Ligase IV defi-ciency in mice leads to defective neurogenesis and embryonic lethality via the p53 pathway. Molecular Cell2000;5: 993–1002.
GaoY, et alInterplay of p53 and DNA-repair protein XRCC4 in tumorigenesis, genomic stability and development. Nature2000;404:897–900.
Lowe SW, Ruley HE, Jacks T, Housman DE. p53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell1993;74:957–967.
Villunger A, Strasser A. Does 'death receptor'-signalling play a role in tumourigenesis and cancer therapy? Oncology Research1998;10:541–550.
Huang DGS, Strasser A. BMS-only proteins-essential initiators of apoptotic cell death. Cell 2000;103:839–842.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Coultas, L., Strasser, A. The molecular control of DNA damage-induced cell death. Apoptosis 5, 491–507 (2000). https://doi.org/10.1023/A:1009617727938
Issue Date:
DOI: https://doi.org/10.1023/A:1009617727938