Abstract
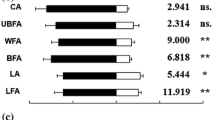
The parasitoids Trichogramma chilonis (Hymenoptera: Trichogrammatidae) and Cotesia plutellae (Hymenoptera: Braconidae), and the predator Chrysoperla carnea (Neuroptera: Chrysopidae), are potential biological control agents for the diamondback moth, Plutella xylostella (Lepidoptera: Yponomeutidae). We present studies on the interactions between these bioagents and various host-associated volatiles using a Y olfactometer. T. chilonis was attracted to a synthetic pheromone blend (Z11–16:Ald, Z11–16:Ac, and Z11–16:OH in a 1:1:0.01 ratio), to Z11–16:Ac alone, and to a 1:1 blend of Z11–16:Ac and Z11–16:Ald. C. plutellae responded to the blend and to Z11–16:Ac and Z11–16:Ald. Male and female C. carnea responded to the blend and to a 1:1 blend of the major components of the pheromone, although no response was elicited by single compounds. Among the four host larval frass volatiles tested (dipropyl disulfide, dimethyl disulfide, allyl isothiocyanate, and dimethyl trisulfide), only allyl isothiocyanate elicited significant responses in the parasitoids and predator, but C. plutellae and both sexes of C. carnea did respond to all four volatiles. Among the green leaf volatiles of cabbage (Brassica oleracea subsp. capitata), only Z3–6:Ac elicited significant responses from T. chilonis, C. plutellae, and C. carnea, but C. plutellae also responded to E2–6:Ald and Z3–6:OH. When these volatiles were blended with the pheromone, the responses were similar to those elicited by the pheromone alone, except for C. carnea males, which had an increased response. The effect of temperature on the response of the biological agents to a mixture of the pheromone blend and Z3–6:Ac was also studied. T. chilonis was attracted at temperatures of 25–35°C, while C. plutellae and C. carnea responded optimally at 30–35°C and 20–25°C, respectively. These results indicate that the sex pheromone and larval frass volatiles from the diamondback moth, as well as volatile compounds from cabbage, may be used by these natural enemies to locate their diamondback moth host.
Similar content being viewed by others
REFERENCES
Auger, J., Lecompte, C., Paris, J., and Thibout, E. 1989. Identification of leek-moth and diamondback moth frass volatiles that stimulate parasitoid, Diadromous pulchellus. J. Chem. Ecol. 15:1391–1398.
Bogahawatte, C. N. L. and Van Emden, H. F. 1996. The influence of the host plant of diamond-back moth (Plutella xylostella) on the plant preferences of its parasitoid Cotesia plutellae in Sri Lanka. Physiol. Entomol. 21:93–96.
Boo, K. S. and Yang, J. P. 2000. Kairomones used by Trichogramma chilonis to find Helicoverpa assulta eggs. J. Chem. Ecol. 26:359–375.
Chow, Y. S., Tin, Y. M., and Hsu, C. T. 1977. Sex pheromone of the diamondback moth (Lepidoptera: Plutellidae). Bull. Inst. Zool. Acad. Sin. 16:99–105.
Colazza, S., Rosi, M. C., and Clemente, A. 1997. Response of egg parasitoid Telenomus busseolae to sex pheromone of Sesamia nonagrioides. J. Chem. Ecol. 23:2437–2453.
Corbet, S. A. 1985. Insect chemosensory responses: A chemical legacy hypothesis. Ecol. Entomol. 10:143–153.
Fan, S. H. and Ho, K. K. 1971. A preliminary study on the life history, rearing method of Apanteles plutellae Kurdj and the effect of different insecticides to it. Plant Prot. Bull. Taiwan 13:151–161.
Flint, M. H., Salter, S. S., and Walters, S. 1979. Caryophyllene: An attractant for the green lacewing. Environ. Entomol. 8:1123–1125.
Guerin, P. M., Stadler, E., Gough, A. J. E., Staddon, B.W., and Games, D. E. 1983. Identification of host plant attractants for the carrot fly, Psila rosae. J. Chem. Ecol. 9:843–861.
Katsoyannos, B. I. and Guerin, P. M.1984. A potent attractant for the black fig fly, Silba adipata. Entomol. Exp. Appl. 35:71–74.
Koshihara, T. and Yamada, H. 1980. Attractant activity of the female sex pheromone of diamondback moth, Plutella xylostella (L.), and analogue. Jpn. J. Appl. Entomol. Zool. 24:6–12.
Koshihara, T., Yamada, H., Tamaki, Y., and Ando, T. 1978. Field attractiveness of the synthetic sex pheromone of the diamondback moth, Plutella xylostella (L.). Appl. Entomol. Zool. 13:138–141.
Lewis, W. J., Nordlund, D. A., Gueldner, R. C., Teal, P. E. A., and Tumlinson, J. H. 1982. Kairomones and their use for management of entomophagous insects: XIII. Kairomonal activity for Trichogramma spp. of abdominal tips, excretion, and a synthetic sex pheromone blend of Heliothis zea (Boddie) moths. J. Chem. Ecol. 8:1323–1331.
Lewis, W. J. and Martin, W. R., Jr. 1990. Semiochemicals for use with parasitoids: Status and future. J. Chem. Ecol. 16:3067–3089.
Liu, S. S., Wang, X. G., Guo, S. J., He, J. H., and Shi, Z.H. 2000. Seasonal abundance of the parasitoid complex associated with the diamondback moth, Plutella xylostella (Lepidoptera: Plutellidae) in Hangzhou, China. Bull. Entomol. Res. 90:221–231.
Loke, W. H. and Ashley, T. R. 1984. Potential uses of kairomones for behavioral manipulation of Cotesia marginiventris (Cresson). J. Chem. Ecol. 10:1377–1384.
Miura, K. and Kobayashi, M. 1998. Effects of host-egg on the parasitism by Trichogramma chilonis Ishii (Hymenoptera: Trichogrammatidae), an egg parasitoid of the diamondback moth. Appl. Entomol. Zool. 33:219–222.
New, T. R. R. 1988. Neuroptera, pp. 249–258, in A. K. Minks and P. Harrewijn (eds.). Aphids: Their Biology, Natural Enemies and Control, Vol. 2B. Elsevier, Amsterdam, The Netherlands.
Noldus, L. P. J. J. 1988. Response of the egg parasitoid Trichogramma pretiosum to the sex pheromone of its host Heliothis zea. Entomol. Exp. Appl. 48:293–300.
Noldus, L. P. J. J. and Van Lenteren, J. C. 1985. Kairomones for the egg parasite Trichogramma evanescensWestwood. I. Effect of volatile substances released by two of its hosts, Pieris brassicae L. and Mamestra brassicae L. J. Chem. Ecol. 11:781–791.
Nordlund, D. A., Lewis, W. J., and Gueldner, R.C. 1983. Kairomones and their use for management of entomophagous insects. XIV. Response of Telenomous remus to abdominal tips of Spodoptera frugiperda as well as to (Z)-9-tetradecen-1-ol acetate and (Z)-9-dodecen-1-ol acetate. J. Chem. Ecol. 9:695–701.
Nordlund, D. A., Chalfant, R. B., and Lewis, W. J. 1985. Response of Trichogramma pretiosum females to volatile synomones from tomato plants. J. Entomol. Sci. 20:372–376.
Potting, R. P. J., Poppy, G. M., and Schuler, T. H. 1999. The role of volatiles from cruciferous plants and pre-flight experience in the foraging behaviour of the specialist parasitoid Cotesia plutellae. Entomol. Exp. Appl. 93:87–95.
Reddy, G. V. P. and Guerrero, A. 2000a. Pheromone-based integrated pest management to control the diamondback moth, Plutella xylostella in cabbage fields. Pest Manage. Sci. 56:882–888.
Reddy, G. V. P. and Guerrero, A. 2000b. Behavioral responses of the diamondback moth to green leaf volatiles of Brassica oleracea subsp. capitata. J. Agric. Food Chem. 48:6025–6029.
Romeis, J., Shanower, T. G., and Zebitz, C. P. W. 1997. Volatile plant infochemicals mediate plant preference of Trichogramma chilonis. J. Chem. Ecol. 23:2455–2465.
Tabone, E., Pintureau, B., Pizzol, J., Michel, F., and Barnay, O. 1999. Ability of 17 strains of Trichogramma to control Plutella xylostella L. (Lep.: Yponomeutidae) in the laboratory. Ann. Soc. Entomol. Fr. 35(suppl.):427–433.
Talekar, N. S. and Shelton, A. M. 1993. Biology, ecology, and management of the diamondback moth. Annu. Rev. Entomol. 38:275–301.
Tamaki, Y., Kawasaki, K., Yamada, H., Koshihara, T., Osaki, N., Ando, T., Yashida, S., and Kakinohama, H. 1977. Z-11-Hexadecenal and Z-11-hexadecenyl acetate: Sex pheromone components of the diamondback moth (Lepidoptera: Plutellidae). Appl. Entomol. Zool. 12:208–210.
Verkerk, R. H. J. and Wright, D. J. 1996. Multitrophic interactions and management of the diamondback moth: A review. Bull. Entomol. Res. 86:205–216.
Vet, L. E. M. and Dicke, M. 1992. Ecology of infochemicals use by natural enemies in tritrophic context. Annu. Rev. Entomol. 37:141–172.
Vinson, S. B. 1985. The behavior of parasitoids, pp. 417–469, in G. A. Kerkut and L. I. Gilbert (eds.). Comprehensive Insect Physiology, Biochemistry and Pharmacology, Vol.9. Pergamon Press, Elmsford, New York.
Visser, J. H., Van Straten, S., and Maarse, H. 1979. Isolation and identification of volatiles in the foliage of potato, Solanum tubersoum, a host plant of the Colorado potato beetle, Leptinotarsa decemlineata. J. Chem. Ecol. 5:13–25.
Zhang, Q.-H., Schlyter, F., and Anderson, P. 1999. Green leaf volatiles interrupt pheromone response of spruce bark beetle, Ips typographus. J. Chem. Ecol. 25:2847–2861.
Zhu, J., CossÉ, A. A., Obrycki, J. J., Boo, K. S., and Baker, T. C. 1999. Olfactory reactions of the twelve-spotted lady beetle, Coleomegilla maculata and the green lacewing, Chrysoperla carnea to semiochemicals released from their prey and host plant: Electroantennogram and behavioral responses. J. Chem. Ecol. 25:1163–1177.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reddy, G.V.P., Holopainen, J.K. & Guerrero, A. Olfactory Responses of Plutella xylostella Natural Enemies to Host Pheromone, Larval Frass, and Green Leaf Cabbage Volatiles. J Chem Ecol 28, 131–143 (2002). https://doi.org/10.1023/A:1013519003944
Issue Date:
DOI: https://doi.org/10.1023/A:1013519003944