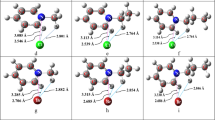
Abstract
We investigated the electronic structure and chemical bonding of the B3 −, Al3 −, and Ga3 − anions, and the gas phase NaB3, NaAl3, and NaGa3 molecules. We found that the ground state of the neutral gas phase salts contains an equilateral triangular anion interacting with a Na+ cation. The B3 −, Al3 −, and Ga3 − anions possess two delocalized π electrons and are found to be aromatic. The triangular anions have been shown to be related to recently synthesized organometallic compound containing an aromatic -Ga3 2− unit, but they are differ from them by four valence electrons. The reason for earlier appearance of the π-orbital in the B3 −, Al3 −, and Ga3 − anions is discussed.
Similar content being viewed by others
REFERENCES
van Zandwijk, G.; Janssen, R. A. J.; Buck, H. M., J. Amer. Chem. Soc. 1990, 112, 4155.
Schleyer, P. v. R.; Jiao, H.; van E. Hommes, N. J. R.; Malkin, V. G.; Malkina, O. L., J. Amer. Chem. Soc. 1997, 119, 12669.
Minkin, V. I.; Glukhovtsev, M. N.; Simkin, B. Y., Aromaticity and Antiaromaticity; Wiley: New York, 1994.
Li, X. W.; Pennington, W. T.; Robinson, G. H., J. Amer. Chem. Soc. 1995, 117, 7578.
Xie, Y.; Schreiner, P. R.; Schaefer, H. F. III; Li, X. W.; Robinson, G. H., J. Amer. Chem. Soc. 1996, 118, 10635.
Robinson, G. H., Account. Chem. Res. 1999, 32, 773.
Li, X.; Kuznetsov, A. E.; Zhang, H. F.; Boldyrev, A. I.; Wang, L. S., Science 2001, 291, 859.
Li, X.; Zhang, H. F.; Wang, L. S.; Kuznetsov, A. E.; Cannon, N. A.; Boldyrev, A. I., Angew. Chem. Intern. Ed. 2001, 40, 1867.
Boldyrev, A. I.; Simons, J.; Li, X.; Chen, W.; Wang, L. S., J. Chem. Phys. 1999, 110, 8980.
Kuznetsov, A. E.; Boldyrev, A. I.; Li, X.; Wang, L. S., J. Amer. Chem. Soc., in press.
Twamley, B.; Power, P. P., Angew. Chem. Intern. Ed. 39, 3500 (2000).
McLean, A. D.; Chandler, G. S., J. Chem. Phys. 1980, 72, 5639.
Clark, T.; Chandrasekhar, J.; Spitznagel, G. W.; Schleyer, P. v. R., J. Comput. Chem. 1983, 4, 294.
Frisch, M. J.; Pople, J. A.; Binkley, J. S., J. Chem. Phys. 1984, 80, 3265.
Parr, R. G.; Yang, W., Density-Functional Theory of Atoms and Molecules; Oxford Univ. Press: Oxford, 1989.
Becke, A. D., J. Chem. Phys. 1993, 98, 5648.
Perdew, J. P.; Chevary, J. A.; Vosko, S. H.; Jackson, K. A.; Pederson, M. R.; Singh, D. J.; Fiolhais, C., Phys. Rev. B 1992, 46, 6671.
Martin, J. M. L.; Francois, J. P.; Gijbels, R., J. Chem. Phys. 1989, 90, 6469.
Baeck, K. K.; Bartlett, R. J., J. Chem. Phys. 1998, 109, 1334.
Meier, U.; Peyerimhoff, S. D.; Grein, F., Z. Phys. D 1990, 17, 209.
Cizek, J., Adv. Chem. Phys. 1969, 14, 35.
Purvis, G. D., III; Bartlett, R. J., J. Chem. Phys. 1982, 76, 1910.
Scuseria, G. E.; Janssen, C. L.; Schaefer, H. F., III, J. Chem. Phys. 1988, 89, 7282.
Cederbaum, L. S., J. Phys. 1975, B8, 290.
Niessen, W. von; Shirmer, J.; Cederbaum, L. S., Comput. Phys. Rep. 1984, 1, 57.
Zakrzewski, V. G.; Ortiz, J. V., Intern. J. Quant. Chem. Quant. Chem. Symp. 1994, 28, 23.
Zakrzewski, V. G.; Ortiz, J. V., Intern. J. Quant. Chem. 1995, 53, 583.
For recent review see: Ortiz, J. V.; Zakrzewski, V. G.; Dolgunitcheva, O. in Conceptual Trends in Quantum Chemistry, Kryachko, E. S., ed.; Kluver: Dordrecht, 1997; Vol. 3; p. 463.
Frisch, M. J.; Trucks, G. M.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Zakrzewski, V. G.; Montgomery, J. A., Jr.; Stratmann, R. E.; Burant, J. C.; Dapprich, S.; Millam, J. M.; Daniels, A. D.; Kudin, K. N.; Strain, M. C.; Farkas, O.; Tomasi, J.; Barone, V.; Cossi, M.; Cammi, R.; Mennucci, B.; Pomelli, C.; Adamo, C.; Clifford, S.; Ocheterski, J.; Petersson, G. A.; Ayala, P. Y.; Cui, Q.; Morokuma, K.; Malick, D. K.; Rabuck, A. D.; Raghavachari, K.; Foresman, J. B.; Cioslowski, J.; Ortiz, J. V.; Baboul, A. G.; Stefanov, B. B.; Liu, G.; Liashenko, A.; Piskorz, P.; Komaromi, I.; Gomperts, R.; Martin, R. L.; Fox, D. J.; Keith, T.; Al-Laham, M. A.; Peng, C. Y.; Nanayakkara, A.; Gonzales, C.; Challacombe, M.; Gill, P. M. W.; Johnson, B. G.; Chen, W.; Wong, M. W.; Andres, J. L.; Head-Gordon, M.; Replogle E. S.; Pople, J. A., GAUSSIAN 98 (Revision A.7); Gaussian, Inc., Pittsburgh, PA, 1998.
Schaftenaar, G. MOLDEN3.4, CAOS/CAMM Center, The Netherlands (1998).
Li, X.; Wu, H.; Wang, X. B.; Wang, L. S., Phys. Rev. Lett. 1998, 81, 1909.
Wu, H.; Li, X.; Wang, X. B.; Ding, C.-F. Wang, L. S., J. Chem. Phys. 1998, 109, 449.
Villalta, P. W.; Leopold, D. G., unpublished data quoted in Ref. [19].
Cha, C.-Y.; Gantefor, G.; Eberhardt, W., J. Chem. Phys. 1994, 100, 995.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kuznetsov, A.E., Boldyrev, A.I. Theoretical Evidence of Aromaticity in X3 − (X = B, Al, Ga) Species. Structural Chemistry 13, 141–148 (2002). https://doi.org/10.1023/A:1015704515336
Issue Date:
DOI: https://doi.org/10.1023/A:1015704515336