Abstract
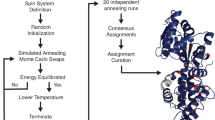
We present a computer algorithm for the automated assignment of polypeptide backbone and13Cβ resonances of a protein of known primary sequence. Input to the algorithm consistsof cross peaks from several 3D NMR experiments: HNCA, HN(CA)CO, HN(CA)HA,HNCACB, COCAH, HCA(CO)N, HNCO, HN(CO)CA, HN(COCA)HA, and CBCA(CO)NH.Data from these experiments performed on glutamine-binding protein are analyzed statisticallyusing Bayes' theorem to yield objective probability scoring functions for matching chemicalshifts. Such scoring is used in the first stage of the algorithm to combine cross peaks fromthe first five experiments to form intraresidue segments of chemical shifts{Ni,HiN,Ciα,Ciβ,C′i}, while the latter five are combined into interresiduesegments {Ciα,Ciβ,C′i,Ni+1,HNi+1}. Given a tentative assignment of segments,the second stage of the procedure calculates probability scores based on the likelihood ofmatching the chemical shifts of each segment with (i) overlapping segments; and (ii) chemicalshift distributions of the underlying amino acid type (and secondary structure, if known). Thisjoint probability is maximized by rearranging segments using a simulated annealing program,optimized for efficiency. The automated assignment program was tested using CBCANH andCBCA(CO)NH cross peaks of the two previously assigned proteins, calmodulin and CheA.The agreement between the results of our method and the published assignments wasexcellent. Our algorithm was also applied to the observed cross peaks of glutamine-bindingprotein of Escherichia coli, yielding an assignment in excellent agreement with that obtainedby time-consuming, manual methods. The chemical shift assignment procedure described hereshould be most useful for NMR studies of large proteins, which are now feasible with the useof pulsed-field gradients and random partial deuteration of samples.
Similar content being viewed by others
References
Bax, A., Ikura, M., Kay, L.E., Torchia, D.A. and Tschudin, T. (1990) J. Magn. Reson., 86, 304–318.
Bax, A. and Grzesiek, S. (1993) Acc. Chem. Res., 26, 131–138.
Cieslar, C., Clore, G.M. and Gronenborn, A.M. (1988) J. Magn. Reson., 80, 119–127.
Clore, G.M. and Gronenborn, A.M. (1991) Science, 252, 1390–1399.
Clubb, R.T., Thanabal, V. and Wagner, G. (1992a) J. Biomol. NMR, 2, 203–210.
Clubb, R.T., Thanabal, V. and Wagner, G. (1992b) J. Magn. Reson., 97, 213–217.
Clubb, R.T. and Wagner, G. (1992) J. Biomol. NMR, 2, 389–394.
Dijkstra, K., Kroon, G.J.A., Van Nuland, N.A.J. and Scheek, R.M. (1994) J. Magn. Reson., A107, 102–105.
Eads, C.D. and Kuntz, I.D. (1989) J. Magn. Reson., 82, 467–482.
Eccles, C., Güntert, P., Billeter, M. and Wüthrich, K. (1991) J. Biomol. NMR, 97, 111–130.
Friedrichs, M.S., Mueller, L. and Wittekind, M. (1994) J. Biomol. NMR, 4, 703–726.
Grzesiek, S. and Bax, A. (1992a) J. Magn. Reson., 96, 432–440.
Grzesiek, S. and Bax, A. (1992b) J. Am. Chem. Soc., 114, 6291–6293.
Grzesiek, S. and Bax, A. (1992c) J. Magn. Reson., 99, 201–207.
Grzesiek, S. and Bax, A. (1993) J. Biomol. NMR, 3, 185–204.
Hing, A.W., Tjandra, N., Cottam, P.F., Schaeffer, J. and Ho, C. (1994) Biochemistry, 33, 8651–8661.
Hsiao, C.-D. (1993) Ph.D. Thesis, University of Pittsburgh, Pittsburgh, PA, U.S.A.
Ikura, M., Kay, L.E. and Bax, A. (1990) Biochemistry, 29, 4659–4667.
Kirkpatrick, S., Gelatt Jr., C.D. and Vecchi, M.P. (1983) Science, 220, 671–680.
Kleywegt, G.J., Boelens, R., Cox, M., Llinás, M. and Kaptein, R. (1991) J. Biomol. NMR, 1, 23–47.
Leopold, M.F., Urbauer, J.L. and Wand, A.J. (1994) Mol. Biotechnol., 2, 61–93.
McEvoy, M.M., Zhou, H., Roth, A.F., Lowry, D.F., Morrison, T.B., Kay, L.E. and Dahlquist, F.W. (1995) Biochemistry, 34, 13871–13880.
McEvoy, M.M., Muhandiram, D.R., Kay, L.E. and Dahlquist, F.W. (1996) Biochemistry, 35, 5633–5640.
Meadows, R.P., Olejniczak, E.J. and Fesik, S.W. (1994) J. Biomol. NMR, 4, 79–96.
Metropolis, N., Rosenbluth, A.W., Rosenbluth, M.N., Teller, A.H. and Teller, E. (1953) J. Chem. Phys., 21, 1087–1092.
Morelle, M., Brutscher, B., Simorre, J.-P. and Marion, D. (1995) J. Biomol. NMR, 5, 154–160.
Olejniczak, E.T., Xu, R.X., Petros, A.M. and Fesik, S.W. (1992) J. Magn. Reson., 100, 444–450.
Olson, J.B. and Markley, J.L. (1994) J. Biomol. NMR, 4, 385–410.
Powers, R., Gronenborn, A.M., Clore, G.M. and Bax, A. (1991) J. Magn. Reson., 94, 209–213.
Press, S.J. (1989) Bayesian Statistics: Principles, Models, and Applications, Wiley, New York, NY, U.S.A.
Press, W.H., Teukolsky, S.A., Vetterling, W.T. and Flannery, B.P. (1992) Numerical Recipes in Fortran, 2nd ed., Cambridge University Press, Cambridge, U.K., pp. 436–438.
Seavey, B.R., Farr, E.A., Westler, W.M. and Markley, J.L. (1991) J. Biomol. NMR, 1, 217–236.
Shen, Q., Simplaceanu, V., Cottam, P.F. and Ho, C. (1989a) J. Mol. Biol., 210, 849–857.
Shen, Q., Simplaceanu, V., Cottam, P.F., Wu, J.-L., Hong, J.-S. and Ho, C. (1989b) J. Mol. Biol., 210, 859–867.
Spera, S. and Bax, A. (1991) J. Am. Chem. Soc., 113, 5490–5492.
Tjandra, N., Simplaceanu, V., Cottam, P.F. and Ho, C. (1992) J. Biomol. NMR, 2, 149–160.
Tjandra, N. (1993) Ph.D. Thesis, Carnegie Mellon University, Pittsburgh, PA, U.S.A.
Tjandra, N., Kuboniwa, H., Ren, H. and Bax, A. (1995) Eur. J. Biochem., 230, 1014–1024.
Van de Ven, F.J.M. (1990) J. Magn. Reson., 86, 633–644.
Weber, P.L., Malikayil, J.A. and Mueller, L. (1988) J. Magn. Reson., 82, 419–426.
Wishart, D.S., Sykes, B.D. and Richards, F.M. (1991) J. Mol. Biol., 222, 311–333.
Wishart, D.S. and Sykes, B.D. (1994) Methods Enzymol., 23 9, 363–392.
Wishart, D.S., Bigham, C.G., Yao, J., Abildgaard, F., Dyson, H.J., Oldfield, E., Markley, J.L. and Sykes, B.D. (1995) J. Biomol. NMR, 6, 135–140.
Wittekind, M. and Mueller, L. (1993) J. Magn. Reson., B101, 201–205.
Xu, J., Straus, S.K., Sanctuary, B.C. and Trimble, L. (1994) J. Magn. Reson., B103, 53–58.
Yu, J., Tjandra, N., Simplaceanu, V., Cottam, P.F., Lukin, J.A. and Ho, C. (1995) Biophys. J., 68, 420.
Yu, J., Simplaceanu, V., Tjandra, N.L., Cottam, P.F., Lukin, J.A. and Ho, C. (1997) J. Biomol. NMR, 9, 167–180.
Zimmerman, D.E. and Montelione, G.T. (1995) Curr. Opin. Struct. Biol., 5, 664–673.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lukin, J.A., Gove, A.P., Talukdar, S.N. et al. Automated probabilistic method for assigning backbone resonances of (13C,15N)-labeled proteins. J Biomol NMR 9, 151–166 (1997). https://doi.org/10.1023/A:1018602220061
Issue Date:
DOI: https://doi.org/10.1023/A:1018602220061