Abstract
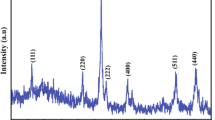
Magnetic Fe3O4 nanoparticles with size below 10 nm have been prepared by the aqueous phase coprecipitation method. The Fe3O4 nanoparticles show typical superparamagnetism. Comparison is made between the dispersed sample and the powder sample, and the results are discussed.
Similar content being viewed by others
REFERENCES
B. H. Sohn and R. E. Cohen, Chem. Mater. 9,2649 (1997).
B. Z. Tang, Y. Geng, et al. Chem. Mater. 11,1581–1589 (1997).
G. Beaucage (ed.), Material Research Society, Warrendale, PA, 1998.
P. Jena (Ed.), Nova Science, New York, 1996.
R. F. Ziolo, U. S. Patent 4,474, 866, 1984.
R. G. Audram and A. P. Huguenard, U. S. Patent 4,302, 523, 1981.
N. M. Pope, Y.-A. Chang, and A. K. J. Sonith, Biomed. Mater. Res. 28,449 (1994).
D. K. Kim, Y. Zhang, W. Viot, K. V. Rao, and M. Muhammed. J. Magn. Magn. Mater. 225,30–36 (2001).
Y. Liu, C. B. Wang, and J. H. Wang, Functional Mater. 30 (1999).
B. D. Cullity, Elements of X-Ray Diffraction, Addison-Wesley, Reading, MA, 1978.
D. H. Han and J. P. Luo, J. Magn. Magn. Mater. 136,176 (1994).
R. Massart, IEEE Trans. Magn. 17,1247 (1981).
R. Kaiser and G. Miskolczy, J. Appl. Phys. 41,1064 (1970).
B. D. Cullity, Introduction to Magnetic Materials, Addison-Wesley, Reading, MA, 1974.
G. A. J. Allan, Phys. Res. B 1,352 (1970).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, Z.L., Liu, Y.J., Yao, K.L. et al. Synthesis and Magnetic Properties of Fe3O4 Nanoparticles. Journal of Materials Synthesis and Processing 10, 83–87 (2002). https://doi.org/10.1023/A:1021231527095
Issue Date:
DOI: https://doi.org/10.1023/A:1021231527095