Abstract
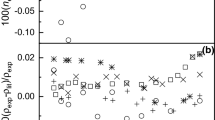
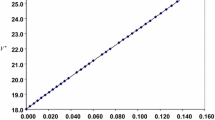
The densities, refractive indexes, and sound velocities for mixtures of ethanol + methanol + dibutyl ether at 25°C and atmospheric pressure, were determined and used to calculate molar volumes, molar refractions, and isentropic compressibilities. The excess molar volumes and the deviations of molar refractions and isentropic compressibilities from mole fraction and volume fraction averages, respectively, of these properties of the pure components were satisfactorily correlated with the composition data by means of the Redlich–Kister polynomial.
Similar content being viewed by others
REFERENCES
I. L. Teng and W. E. Acree Jr., Phys. Chem. Liq. 24, 249 (1992).
J. Singh, I. M. Joshi, and S. C. Sharma, J. Chem. Thermodyn. 25, 1049 (1993).
A. Serna, L. García de la Fuente, J. A. González, J. C. Cobos, and C. Casanova, Fluid Phase Equilib. 110, 361 (1995).
E. Jiménez, C. Franjo, L. Segade, J. L. Legido, and M. I. Paz Andrade, Fluid Phase Equilib. 133, 173 (1997).
J. A. Riddick, W. B. Bunger, and T. Sakano, Organic Solvents, 4th. edn. (Wiley, New York, 1986).
T. M. Aminabhavi, M. Y. Aralaguppi, Sh. B. Harogoppad, and R. H. Balundgi, J. Chem. Eng. Data 38, 31 (1993).
A. Arce, J. Mendoza, J. Martínez-Ageitos, and A. Soto, J. Chem. Eng. Data 41, 724 (1996).
A. Arce, E. Rodil, and A. Soto, J. Chem. Eng. Data 42, 721 (1997).
O. J. Redlich and A. T. Kister, Ind. Eng. Chem. 40, 345 (1948).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Arce, A., Rodil, E. & Soto, A. Molar Volume, Molar Refraction, and Isentropic Compressibility Changes of Mixing at 25°C for the System Ethanol + Methanol + Dibutyl Ether. Journal of Solution Chemistry 27, 911–923 (1998). https://doi.org/10.1023/A:1022611228690
Issue Date:
DOI: https://doi.org/10.1023/A:1022611228690