Abstract
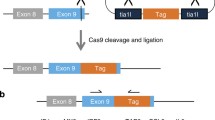
The gene trap approach is based on the integration of a gene trap vector into the genome. This can be done either by electroporation of a plasmid construct or by infection with a viral vector. Commonly used viral gene trap vectors have been shown to select for integrations near the 5′ end of genes. To date, no plasmid vector with a similar tendency has been reported. In this paper we describe a new plasmid vector, pKC199βgeo. This vector contained a short splice acceptor fragment from the Hoxc9 gene, a full length lacZ gene, including an ATG, and a reduced activity, mutant neomycin phosphotransferase gene as a selectable marker. This vector enriched the population of trapped genes in our gene trap screen for insertion events in the 5′ end of genes. In the two cases examined the β-galactosidase activity pattern accurately reflected the endogenous promotor activity.
Similar content being viewed by others
References
Adams ME, Kerlavage AR, Fleischmann RD, Fuldner RA, Bult CJ, Lee NH, et al. (1995) Initial assessment of human gene diversity and expression patterns based upon 83 million nucleotides of cDNA sequence. Nature 377, 3–174.
Bauer H, Sonnleitner U, Lametschwandtner A, Steiner M, Adam H and Bauer HC (1995) Ontogenic expression of the erythroid-type glucose transporter (Glut 1) in the telencephalon of the mouse: correlation to the tightening of the blood-brain barrier. Develop Brain Res 86, 317–325.
Chirgwin JM, Przybyla AE, MacDonald RJ and Rutter WJ (1979) Isolation of biologically active ribonucleic acid from sources enriched in ribonucleases. Biochemistry 18, 5294–5299.
Chowdhury K, Bonaldo P, Torres M, Stoykova A and Gruss P (1997) Evidence for the stochastic integration of gene trap vectors into the mouse germline. Nucleic Acids Res 25, 1531–1536.
Coccia EM, Cicala C, Charlesworth A, Ciccarelli C, Rossi GB, Philipson L, et al. (1992) Regulation and expression of a growth arrest-specific gene (gas5) during growth, differentiation, and development. Mol Cell Biol 12(8), 3514–3521.
Devaskar S, Zahm DS, Holtzclaw L, Chundu K and Wadzinski BE (1991) Developmental regulation of the distribution of rat brain insulin-insensitive (Glut 1) glucose transporter. Endocrinology 129, 1530–1540.
Erselius JR, Goulding MD and Gruss P (1990) Structure and expression pattern of the murine Hox-3.2 gene. Development 110, 629–642.
Friedrich G and Soriano P (1991) Promoter traps in embryonic stem cells: a genetic screen to identify and mutate developmental genes in mice. Genes Dev 5, 1513–1523.
Frohman MA, Dush MK and Martin GR (1988) Rapid production of full-length cDNAs from rare transcripts: Amplification using a single gene-specific oligonucleotide primer. Proc Natl Acad Sci USA 85, 8998–9002.
Hawkins JD (1988) A survey on intron and exon lengths. Nucleic Acids Res 16, 9893–9908.
Hogan BLM, Constantini F and Lacy E (1986) Manipulating the Mouse Embryo: A Laboratory Manual. Cold Spring Harbor Laboratory, New York.
Krainer AR and Maniatis T (1988) RNA splicing. In: Hames BD and Glover DM (eds) Transcription and Splicing. Oxford, IRL Press.
Langley KE, Villarejo MR, Fowler AV, Zamenhof P and Zabin I (1975) Molecular basis of β-Galactosidase alpha complementation. Proc Natl Acad Sci USA 72, 1254–1257.
Nagy A and Rossant J (1993) Production of completely ES cell-derived fetuses. In: Joyner JL (ed.) Gene Targeting. A Practical Approach. (pp. 147–178) Oxford, IRL Press.
Reed BC, Shade D, Alperovich F and Vang M (1990) 3T3-L1 adipocyte glucose transporter (HepG2 class): sequence and regulation of protein and mRNA expression by insulin, differentiation, and glucose starvation. Arch Biochem Biophys 279, 261–274.
Sambrook J, Fritsch EF and Maniatis T (1989) Molecular Cloning. A Laboratory Manual. Cold Spring Harbor Laboratory, Cold Spring Harbor.
Sanger F, Nicklen S and Coulsen AR (1977) DNA sequencing with chain-termination inhibitors. Proc Natl Acad Sci USA 74, 5463–5467.
Skarnes WC, Moss JE, Hurtley SM and Beddington RS (1995) Capturing genes encoding membrane and secreted proteins important for development. Proc Natl Acad Sci USA 92, 6592–6596.
Soininen R, Schoor M, Henseling U, Tepe C, Kisters-Woike B, Rossant J and Gossler A (1992) The mouse Enhancer trap locus 1 (Etl-1): a novel mammalian gene related to Drosophila and yeast transcriptional regulator genes. Mech Dev 39, 111–123.
Surh LC, Beaudet AL, O'Brien WE (1991) Molecular characterization of the murine argininosuccinate synthetase locus. Gene 99, 181–189.
Takeuchi T, Yamazyki Y, Katoh-Fukui Y, Tsuchiya R, Kondo S, Motoyama J, et al. (1995) Gene trap capture of a novel mouse gene, jumoniji, required for neural tube formation. Genes Develop 9, 1211–1222.
Smith DE And Gridley T (1992) Differential screening of a PCR generated mouse embryo cDNA library: glucose transporters are differentially expressed in the early postimplantation mouse embryo. Development 116, 555–561.
Torres M, Stoykova A, Huber O, Chowdhury K, Bonaldo P, Mansouri A, et al. (1997) An alpha-E-catenin gene trap mutation defines its function in preimplantation development. Proc Natl Acad Sci USA 94, 901–906.
Voss AK, Thomas T and Gruss P (1997) Germ line chimeras from female ES cells. Exp Cell Res 230, 45–49.
Voss AK, Thomas T and Gruss P (1998) Efficiency assessment of the gene trap approach. Develop Dyn 284, 171–180.
Yenofsky RL, Fine M and Pellow JW (1991) A mutant neomycin phosphotransferase II gene reduces the resistance of transformants to antibiotic selection pressure. Proc Natl Acad Sci USA 87, 3435–3439.
Zambrowicz BP, Friedrich GA, Buxton EC, Lilleberg SL, Person C and Sands AT (1998) Disruption and sequence identification of 2,000 genes in mouse embryonic stem cells. Nature 392, 608–611.
Zipser D (1963) A study of the urea-produced subunits of β-galactosidase. J Mol Biol 7, 113–121.