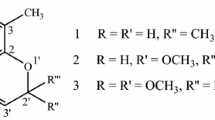Abstract
Four phenolic amides, dihydro-N-caffeoyltyramine (1), trans-N-feruloyloctopamine (2), trans-N-caffeoyltyramine (3), and cis-N-caffeoyltyramine (4), were isolated from an ethyl acetate extract of the root bark of Lycium chinense Miller. All had an anti-fungal effect; compounds 1-3 were potent at 5–10 μg ml−1 and were without hemolytic activity against human erythrocyte cells. Compound 4 was active at 40 μg ml−1. All four compounds impeded the dimorphic transition of pathogen, Candida albicans.
Similar content being viewed by others
References
Blondle SE, Houghten RA (1992) Design of model amphipathic peptides having potent antimicrobial activities. Biochemistry 31: 12688–12694.
Chen C-Y, Chang F-R, Yen H-F, Wu Y-C (1998) Amides from stems of Annona cherimola. Phytochemistry 45: 1443–1447.
Funayama S, Yoshida K, Konno H, Hikkino H (1980) Structure of Kukoamine A, a hypotensive principle of Lycium chinenseroot bark. Tetrahedron Lett. 21: 1355–1356.
Funayama S, Zhang G.-R, Nozoe S (1995) Kukoamine B, a spermine alkaloid from Lycium chinense. Phytochemistry 38: 1529–1531.
Han S-H, Lee H-H, Lee I-S, Moon Y-H, Woo E-R (2002) A new phenolic amide from Lycium chinenseMiller. Arch. Pharm. Res. 25: 433–437.
Kim SY, Choi Y-H, Huh H, Kim J, Kim YC, Lee HS (1997) New antihepatotoxic cerebroside from Lycium chinenseFruits. J. Nat. Prod. 60: 274–276.
Lajide L, Escoubas P, Mizutani J (1995) Termite antifeedant activity in Xylopia aethiopica. Phytochemistry 40: 1105–1112.
Lehrer R, Lichtenstein AK, Ganz T (1993) Defensins: antimicrobial and cytotoxic peptides of mammalian cells. Annu. Rev. Immunol. 11: 105–128.
Mclain N, Ascanio R, Baker C, Strohaver RA, Dolan JW (2000) Undecylenic acid inhibits morphogenesis of Candida albicans. Antimicrob. Agents Chemother. 44: 2873–2875.
Morota T, Sasaki H, Chin M, Sato T, Katayama N, Fukuyama K, Mitsuhashi H (1987) Studies on the crude drug containing the angiotensin I converting enzyme inhibitors(I) on the active principles of Lycium chinenseMiller. Shoyakugaku Zasshi. 41: 169–173.
Mühlenbeck U, Kortenbusch A, Barz W (1996) Formation of hydroxycinnamoyl-amides and a-hydroxyacetovanillone in cell cultures of Solanum khasianum. Phytochemistry 42: 1573–1579.
Negrel J, Pollet B, Lapierre C (1996) Ether-linked ferulic acid amides in natural and wound periderms of potato tuber. Phytochemistry 43: 1195–1199.
Sakakibara I, Katsuhara T, Ikeya Y, Hayashi K, Mitsuhashi H (1991) Cannabisin A, an arylnaphthalene lignanamide from fruits of Cannabis sativa. Phytochemistry 30: 3013–3016.
Sannai A, Fujimori T, Kato K (1982) Isolation of (-)-1,2-dehydro-a-cyperone and solavetivone from Lycium chinense. Phytochemistry 21: 2986–2987.
Santos LP, Boaventura MA, Oliveira AB, Cassady JM (1996) Grossamide and N-trans-caffeoyltyramine from Annona crassifloraseeds. Plant. Med. 62: 76. 1130
Terauchi M, Kanamori H, Nobuso M, Yahara S, Nohara T (1997) Detection and determination of antioxidative components in Lycium chinense. Nat. Med. 51: 387–391.
Terauchi M, Kanamori H, Nobuso M, Yahara S, Yamasaki K (1998) New acyclic diterpene glycoside, Lyciumoside IV-IX from Lycium chinenseMill. Nat. Med. 52: 167–171.
Wu Y-C, Chang G-Y, Ko F-N, Teng C-M (1995) Bioactive constituents from the stems of Annona montana. Plant. Med. 61: 146–149.
Yahara S, Shigeyama C, Ura T, Wakamatsu K, Yasuhara T, Nohara T (1993) Cyclic peptides, acyclic diterpene glycoside and other compounds from Lycium chinenseMill. Chem. Pharm. Bull. 41: 703–709.
Yoshihara T, Takamatsu S, Sakamura S (1978) Three new phenolic amides from the roots of eggplant (Solanum melongenaL.). Agric. Biol. Chem. 42: 623–627.
Yoshihara T, Yamaguchi K, Takamatsu S, Sakamura S (1981) A new lignan amide, grossamide from bell pepper (Capsicum annumvar. grossum). Agric. Biol. Chem. 45: 2593–2598.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lee, D.G., Park, Y., Kim, MR. et al. Anti-fungal effects of phenolic amides isolated from the root bark of Lycium chinense . Biotechnology Letters 26, 1125–1130 (2004). https://doi.org/10.1023/B:BILE.0000035483.85790.f7
Issue Date:
DOI: https://doi.org/10.1023/B:BILE.0000035483.85790.f7