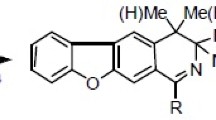
Abstract
A mixture of isomeric phenyloxazines (2, 3) in a 5:4 ratio was synthesized via a [4 + 2] addition reaction of a heterocyclic diene-precursor prepared from bromoacetophenone oxime and the methyl ester of chrysenequinonecarboxylic acid (1). The structures of the synthesized compounds were confirmed using spectral methods.
Similar content being viewed by others
REFERENCES
G. A. Tolstikov, E. E. Shul'ts, T. Sh. Mukhametyanova, I. P. Baikova, and L. V. Spirikhin, Zh. Org. Khim., 29, No. 4, 698 (1993).
G. A. Tolstikov, E. E. Shul'ts, T. Sh. Mukhametzyanova, V. S. Sultanova, and L. V. Spirikhin, Zh. Org. Khim., 28, No. 6, 1310 (1992).
R. C. Cookson, E. Grundwell, R. R. Hill, and J. Hudec, J. Chem. Soc., 9, 3062 (1964).
V. Nechepurenko, E. E. Shul'ts, and G. A. Tolstikov, International Conference on Natural Products and Physiologically Active Substances (ICNPAC-98), Book of Abstracts (1998), 122.
E. V. Tret'yakova, Candidate Dissertation in Chemical Sciences, Ufa (2003).
A. Tahdi, S. L. Titouani, and M. Soufiaoui, Tetrahedron, 54, 65 (1998).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tret'yakova, E.V., Flekhter, O.B., Galin, F.Z. et al. Synthesis of Phenyl-2,3-oxazine Derivatives of Chrysenequinonecarboxylic Acid. Chemistry of Natural Compounds 40, 391–393 (2004). https://doi.org/10.1023/B:CONC.0000048255.87504.d0
Issue Date:
DOI: https://doi.org/10.1023/B:CONC.0000048255.87504.d0