Abstract
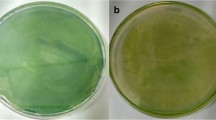
We investigated the allelopathic activity of two submersed macrophytes with different growth forms and nutrient uptake modes, Ceratophyllum demersum and Najas marina ssp. intermedia. A bioassay-directed method development revealed optimal extraction solvents for allelochemicals from both macrophytes. For Najas, 50% methanol and for Ceratophyllum 50% acetone yielded the strongest inhibition in the agar-diffusion assay with various filamentous or chroococcal cyanobacteria as target species. Further fractionation by liquid–liquid extraction (LLE) and solid phase extraction (SPE) procedures showed that both aquatic plants appear to have more than one active fraction, one being hydrophilic and one moderately lipophilic. The water-soluble allelochemicals may inhibit phytoplankton whereas the lipophilic allelochemicals may act through direct cell–cell contact, e.g., against epiphytes. Both macrophytes also exuded allelopathically active compounds into the surrounding medium as shown by SPE of their incubation water.
Similar content being viewed by others

References
Agami, M., S. Beer & Y. Waisel, 1980. Growth and photosynthesis of Najas marina L. as affected by light intensity. Aquat. Bot. 9: 285–289.
Agami, M., S. Beer & Y. Waisel, 1984. Seasonal variations in the growth capacity of Najas marina L. as a function of various water depth at the Yarkon Springs, Israel. Aquat. Bot. 19: 45–51.
Agami, M. & Y. Waisel, 1985. Inter-relationship between Najas marina L. and three other species of aquatic macrophytes. Hydrobiologia 126: 169–173.
Carignan, R. & J. Kalff, 1980. Phosphorus sources for aquatic weeds: water or sediments? Science 207: 987–989.
Denny, P., 1987. Mineral cycling by wetland plants – a review. Arch. Hydrobiol. Beih. Ergebn. Limnol. 27: 1–25.
Elakovich, S. D., 1989. Allelopathic aquatic plants for aquatic weed management. Biologia Plantarum 31: 479–486.
Elakovich, S. D. & J. W. Wooten, 1989. Allelopathic potential of 16 aquatic and wetland plants. J. Aquat. Plant Manag. 27: 78–84.
Fitzgerald, G. P., 1969. Some factors in the competition or antagonism among bacteria, algae and aquatic weeds. J. Phycol. 5: 351–359.
Flores, E. & C. P. Wolk, 1986. Production, by filamentous, nitrogen-fixing cyanobacteria, of a bacteriocin and of other antibiotics that kill related strains. Arch. Microbiol. 145: 215–219.
Gopal, B. & U. Goel, 1993. Competition and allelopathy in aquatic plant communities. Bot. Rev. 59: 155–210.
Gross, E. M., 1999. Allelopathy in benthic and littoral areas: Case studies on allelochemicals from benthic cyanobacteria and submersed macrophytes. In Inderjit, K. M., M. Dakshini & C. L. Foy (eds), Principles and Practices in Plant Ecology: Allelochemical Interactions. CRC Press, LLC, Boca Raton: 179–199.
Gross, E. M., H. Meyer & G. Schilling, 1996. Release and ecological impact of algicidal hydrolyzable polyphenols in Myriophyllum spicatum. Phytochemistry 41: 133–138.
Gross, E. M., C. P. Wolk & F. Jüttner, 1991. Fischerellin, a new allelochemical from the freshwater cyanobacterium Fischerella muscicola. J. Phycol. 27: 686–692.
Gross, E. M., C. Feldbaum & A. Graf, 2003. Epiphyte biomass and elemental composition on submersed macrophytes in shallow eutrophic lakes. Hydrobiologia 506–509: 559–565.
Herz, G., 2001. Aktionsprogramm zur Sanierung oberschwäbischer Weiher und Seen. Report. Tettnang/Tübingen, Germany.
Hutchinson, G. E., 1975. A Treatise on Limnology. Vol III. Limnological Botany. John Wiley & Sons, New York.
Jasser, I., 1994. Influence of Ceratophyllum demersum on phytoplankton community in experimental conditions. Verh. int. Ver. theor. angew. Limnol. 25: 2291–2295.
Jasser, I., 1995. The influence of macrophytes on a phytoplankton community in experimental conditions. Hydrobiologia 306: 21–32.
Jüttner, F., J. Leonhardt & S. Möhren, 1983. Environmental factors affecting the formation of mesityloxide, dimethylallylic alcohol and other volatile compounds excreted by Anabaena cylindrica. J. Gen. Microbiol. 129: 407–412.
Kogan, S. I. & G. A. Chinnova, 1972. Relations between Ceratophyllum demersum (L.) and some blue-green algae. Hydrobiol. J. 8: 14–19 (21–27).
Körner, S. & A. Nicklisch, 2002. Allelopathic growth inhibition of selected phytoplankton species by submerged macrophytes. J. Phycol. 38: 862–871.
Madsen, T. V. & K. Sand-Jensen, 1994. The interactive effects of light and inorganic carbon on aquatic plant growth. Plant Cell Environ. 17: 955–962.
Mjelde, M. & B. A. Faafeng, 1997. Ceratophyllum demersum hampers phytoplankton development in some small Norwegian lakes over a wide range of phosphorus concentrations and geographical latitude. Freshwat. Biol. 37: 355–365.
Nakai, S., Y. Inoue, M. Hosomi & A. Murakami, 1999. Growth inhibition of blue-green algae by allelopathic effects of macrophytes. Wat. Sci. Technol. 39: 47–53.
Sand-Jensen, K., 1990. Epiphyte shading – its role in resulting depth distribution of submerged aquatic macrophytes. Folia Geobotanica & Phytotaxonomica 25: 315–320.
Scheffer, M., S. H. Hosper, M. L. Meijer, B. Moss & E. Jeppesen, 1993. Alternative equilibria in shallow lakes. Trends Ecol. Evol. 8: 275–279.
Spencer, W. E. & R. G. Wetzel, 1993. Acclimation of photosynthesis and dark respiration of a submersed angiosperm beneath ice in a temperate lake. Plant Physiol. 101: 985–991.
Wium-Andersen, S., U. Anthoni & G. Houen, 1983. Elemental sulphur, a possible allelopathic compound from Ceratophyllum demersum. Phytochemistry 22: 2613.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gross, E.M., Erhard, D. & Iványi, E. Allelopathic activity of Ceratophyllum demersum L. and Najas marina ssp. intermedia (Wolfgang) Casper. Hydrobiologia 506, 583–589 (2003). https://doi.org/10.1023/B:HYDR.0000008539.32622.91
Issue Date:
DOI: https://doi.org/10.1023/B:HYDR.0000008539.32622.91