Abstract
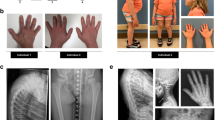
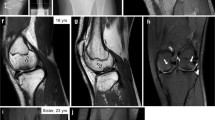
Members of the CCN (for CTGF, cyr61/cef10, nov) gene family encode cysteine-rich secreted proteins with roles in cell growth and differentiation1. Cell-specific and tissue-specific differences in the expression and function of different CCN family members suggest they have non-redundant roles. Using a positional-candidate approach, we found that mutations in the CCN family member WISP3 are associated with the autosomal recessive skeletal disorder progressive pseudorheumatoid dysplasia (PPD; MIM 208230). PPD is an autosomal recessive disorder that may be initially misdiagnosed as juvenile rheumatoid arthritis2,3,4,5. Its population incidence has been estimated at 1 per million in the United Kingdom4, but it is likely to be higher in the Middle East and Gulf States6. Affected individuals are asymptomatic in early childhood2,3. Signs and symptoms of disease typically develop between three and eight years of age. Clinically and radiographically, patients experience continued cartilage loss and destructive bone changes as they age2,3,4,5,6,7, in several instances necessitating joint replacement surgery by the third decade of life. Extraskeletal manifestations have not been reported in PPD. Cartilage appears to be the primary affected tissue, and in one patient, a biopsy of the iliac crest revealed abnormal nests of chondrocytes and loss of normal cell columnar organization in growth zones5. We have identified nine different WISP3 mutations in unrelated, affected individuals, indicating that the gene is essential for normal post-natal skeletal growth and cartilage homeostasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bork, P. The modular architecture of a new family of growth regulators related to connective tissue growth factor. FEBS Lett. 327, 125 –130 (1993).
El-Shanti, H.E., Omari, H.Z. & Qubain, H.I. Progressive pseudorheumatoid dysplasia: report of a family and review. J. Med. Genet. 34, 559–563 (1997).
Rezai-Delui, H., Mamoori, G., Sadri-Mahvelatie, E. & Noori, N.M. Progressive pseudorheumatoid chondrodysplasia: a report of nine cases in three families. Skeletal Radiol. 23, 411– 419 (1994).
Wynne-Davies, R., Hall, C. & Ansell, B.M. Spondylo-epiphysial dysplasia tarda with progressive arthropathy. A "new" disorder of autosomal recessive inheritance. J. Bone Joint Surg. Br. 64, 442–445 ( 1982).
Spranger, J., Albert, C., Schilling, F., Bartsocas, C. & Stöss, H. Progressive pseudorheumatoid arthritis of childhood (PPAC). A hereditary disorder simulating rheumatoid arthritis. Eur. J. Pediatr. 140, 34– 40 (1983).
Teebi, A.S. & Al-Awadi, S.A. Spondyloepiphyseal dysplasia tarda with progressive arthropathy: a rare disorder frequently diagnosed among Arabs. J. Med. Genet. 23, 189– 191 (1986).
Kozlowski, K., Kennedy, J. & Lewis, I.C. Radiographic features of progressive pseudorheumatoid arthritis. Australas. Radiol. 30, 244– 250 (1986).
El-Shanti, H. et al. Assignment of gene responsible for progressive pseudorheumatoid dysplasia to chromosome 6 and examination of COL10A1 as candidate gene. Eur. J. Hum. Genet. 6, 251–256 (1998).
Fischer, J. et al. Genetic linkage of progressive pseudorheumatoid dysplasia to a 3-cM interval of chromosome 6q22. Hum. Genet. 103, 60–64 (1998).
Pennica, D. et al. WISP genes are members of the connective tissue growth factor family that are up-regulated in Wnt-1-transformed cells and aberrantly expressed in human colon tumors. Proc. Natl Acad. Sci. USA 95 , 14717–14722 (1998).
O'Brien, T.P., Yang, G.P., Sanders, L. & Lau, L.F. Expression of cyr61, a growth factor-inducible immediate-early gene. Mol. Cell. Biol. 10, 3569–3577 ( 1990).
Ryseck, R.-P., Macdonald-Bravo, H., Mattei, M.-G. & Bravo, R. Structure, mapping, and expression of fisp-12, a growth factor-inducible gene encoding a secreted cysteine-rich protein. Cell Growth Differ. 2, 225–233 ( 1991).
Bradham, D.M., Igarashi A., Potter, R.L. & Grotendorst, G.R. Connective tissue growth factor: a cysteine-rich mitogen secreted by human vasular endothelial cells is related to the SRC-induced immediate early gene product CEF-10. J. Cell Biol. 114, 1285– 1294 (1991).
Joliot, V. et al. Proviral rearrangements and overexpression of a new cellular gene (nov) in myeloblastosis-associated virus type 1-induced nephroblastomas. Mol. Cell. Biol. 12, 10– 21 (1992).
Zhang, R. et al. Identification of rCop-1, a new member of the CCN protein family, as a negative regulator for cell transformation. Mol. Cell. Biol. 18, 6131–6141 ( 1998).
Hashimoto, Y. et al. Expression of the Elm1 gene, a novel gene of the CCN (connective tissue growth factor, Cyr61/Cef10, and neuroblastoma overexpressed gene) family, suppresses in vivo tumor growth and metastasis of K-1735 murine melanoma cells. J. Exp. Med. 187, 289– 296 (1998).
Yang, G.P. & Lau, L.F. Cyr61, product of a growth factor-inducible immediate early gene, is associated with the extracellular matrix and the cell surface. Cell Growth Differ. 2, 351 –357 (1991).
O'Brien, T.P. & Lau, L.F. Expression of the growth factor-inducible immediate early gene cyr61 correlates with chondrogenesis during mouse embryonic development. Cell Growth Differ. 3, 645–654 (1992).
Kireeva, M.L., Mo, F., Yang, G.P. & Lau, L.F. Cyr61, a product of a growth factor-inducible immediate-early gene, promotes cell proliferation, migration, and adhesion. Mol. Cell. Biol. 16, 1326–1334 (1996).
Martinerie, C., Chevalier, G., Rauscher, F.J. III & Perbal, B. Regulation of nov by WT1: a potential role for nov in nephrogenesis. Oncogene 12, 1479– 1492 (1996).
Wong, M., Kireeva, M.L., Kolesnikova, T.V. & Lau, L.F. Cyr61, a product of a growth factor-inducible immediate-early gene, regulates chondogenesis in mouse limb bud mesenchymal cells. Dev. Biol. 192, 492–508 (1997).
Grotendorst, G.R. Connective tissue growth factor: a mediator of TGF-β action on fibroblasts. Cytokine Growth Factor Rev. 8, 171– 179 (1997).
Kothapalli, D., Frazier, K.S., Welply, A., Segarini, P.R. & Grotendorst, G.R. Transforming growth factor B induces anchorage-independent growth of NRK fibroblasts via a connective tissue growth factor-dependent signaling pathway. Cell Growth Differ. 8, 61–68 (1997 ).
Schütze, N. et al. The human analog of murine cystein rich protein 16 ( sic) is a 1∝,25-dihydroxyvitamin D3 responsive immediate early gene in human fetal osteoblasts: regulation by cytokines, growth factors, and serum. Endocrinology 139, 1761– 1770 (1998).
Erlebacher, A., Filvaroff, E.H., Gitelman, S.E. & Derynck, R. Toward a molecular understanding of skeletal development. Cell 80, 371–378 ( 1995).
Brigstock, D.R. et al. Purification and characterization of novel heparin-binding growth factors in uterine secretory fluids. J. Biol. Chem. 272, 20275–20282 (1997).
Dale, T.C. Signal transduction by the Wnt family of ligands. Biochem. J. 329, 209–223 (1998).
Parr, B.A., Avery, E.J., Cygan, J.A. & McMahon, A.P. The classical mouse mutant postaxial hemimelia results from a mutation in the Wnt 7a gene. Dev. Biol. 202, 228–234 (1998).
Yoo, J.U. et al. The chondrogenic potential of human bone-marrow-derived mesenchymal progenitor cells. J. Bone Joint Surg. Am. 80, 1745–1757 (1998).
Legius, E., Mulier, M., Van Damme, B. & Fryns, J.P. Progressive pseudorheumatoid arthritis of childhood (PPAC) and normal adult height. Clin. Genet. 44, 152– 155 (1993).
Altschul, S.F., Gish, W., Miller, W., Myers, E.W. & Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 ( 1990).
Vikkula, M. et al. Autosomal dominant and recessive osteochondrodysplasias associated with the COL11A2 locus. Cell 80, 431– 437 (1995).
Carrel, L. et al. X inactiviation analysis and DNA methylation studies of the ubiquitin activating enzyme E1 and PCTAIRE-1 genes in human and mouse. Hum. Mol. Genet. 5, 391–401 (1996).
Acknowledgements
We thank the patients and their families for participating in this study, and J. Marcelino, Y. Gong, S. Gregory, P. Modaff, T. Haqqi, B. Johnstone, F.M. Pope and A. Richards for sharing clinical and scientific expertise. This work was supported by NIH grant AR43827 and by a biomedical sciences grant from the March of Dimes Birth Defects Foundation (M.L.W.), grant 45-401.95 from the Swiss National Foundation (A.S.-F.) and a concerted-action grant from the University of Antwerp (W.V.H.).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Rights and permissions
About this article
Cite this article
Hurvitz, J., Suwairi, W., Van Hul, W. et al. Mutations in the CCN gene family member WISP3 cause progressive pseudorheumatoid dysplasia. Nat Genet 23, 94–98 (1999). https://doi.org/10.1038/12699
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/12699
This article is cited by
-
Mesenchymal stromal cells from a progressive pseudorheumatoid dysplasia patient show altered osteogenic differentiation
European Journal of Medical Research (2022)
-
Inflammatory conditions of the pediatric hand and non-inflammatory mimics
Pediatric Radiology (2022)
-
The matricellular protein CCN6 differentially regulates mitochondrial metabolism in normal epithelium and in metaplastic breast carcinomas
Journal of Cell Communication and Signaling (2022)
-
Cellular communication network factor 3 in cartilage development and maintenance
Journal of Cell Communication and Signaling (2021)
-
Skeletal phenotype/genotype in progressive pseudorheumatoid chondrodysplasia
Clinical Rheumatology (2020)