Abstract
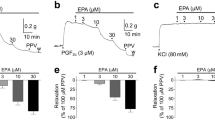
STIMULATION of the endothelial lining of arteries with acetyl-choline results in the release of a diffusible substance that relaxes and hyperpolarizes the underlying smooth muscle1–9. Nitric oxide (NO) has been a candidate for this substance, termed endothelium-derived relaxing factor10,11. But there are several observations that argue against the involvement of NO in acetylcholine-induced hyperpolarization. First, exogenous NO has no effect on the membrane potential of canine mesenteric arteries7. Second, although haemoglobin (believed to bind and inactivate NO (refs 11–15)) and methylene blue (which prevents the stimulation of guanylate cyclase11–16) inhibit relaxation7,12–14, neither has an effect on hyperpolarization5,7,8. Finally, nitroprusside, thought to generate NO in vascular smooth muscle14,16, relaxes rat aorta without increasing rubidium efflux17. Nevertheless, nitrovasodilators, nitroprusside and nitroglycerine cause hyperpolarization in some arteries18–20. NO might therefore be responsible for at least part of the hyperpolarization induced by acetylcholine. We now report that hyperpolarization and relaxation evoked by acetylcholine are reduced by NG-monomethyl-L-arginine, an inhibitor of NO biosynthesis from L-arginine21–25. Thus NO derived from the endothelium can cause hyperpolarization of vascular smooth muscle, which might also contribute to relaxation by closing voltage-dependent calcium channels. Our findings raise the possibility that hyperpolarization might be a component of NO signal transduction in neurons or inflammatory cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Furchgott, R. F. & Zawadzki, J. V. Nature 288, 373–376 (1980).
Kuriyama, H. & Suzuki, H. Br. J. Pharmac. 64, 493–501 (1978).
Bolton, T. B., Lang, R. J. & Takewaki, T. J. Physiol., Lond. 351, 549–572 (1984).
Komori, K. & Suzuki, H. Br. J. Pharmac. 92, 657–664 (1987).
Chen, G., Suzuki, H. & Weston, A. H. Br. J. Pharmac. 95, 1165–1174 (1988).
Feletou, M. & Vanhoutte, P. M. Br. J. Pharmac. 93, 515–524 (1988).
Komori, K., Lorenz, R. R. & Vanhoutte, P. M. Am. J. Physiol. 255, H207–212 (1988).
Chen, G. & Suzuki, H. J. Physiol., Lond. 410, 91–106 (1989).
Parkington, H. C. et al. Proc. Aust. Physiol. Pharmac. Soc. 20, 17P (1989).
Palmer, R. M. J., Ferrige, A. G. & Moncada, S. Nature 327, 524–526 (1987).
Ignarro, L. J., Buga, G. M., Wood, K. S., Byrns, R. E. & Chaudhuri, G. Proc. natn. Acad. Sci. U.S.A. 84, 9265–9269 (1987).
Dusting, G. J., Read, M. A. & Stewart, A. G. Clin. exp. Pharmac. Physiol. 15, 83–92 (1988).
Martin, W., Villani, G. M., Jothianandan, D. & Furchgott, R. F. J. Pharmac. exp. Ther. 232, 708–716 (1985).
Ignarro, L. J. & Kadowitz, P. J. A. Rev. Pharmac. Tox. 25, 171–191 (1985).
Feelisch, M. & Noack, E. A. Eur. J. Pharmac. 139, 19–30 (1987).
Waldman, S. A. & Murad, F. Pharmac. Rev. 39, 163–196 (1987).
Taylor, S. G., Southerton, J. S., Weston, A. H. & Baker, J. R. J. Br. J. Pharmac. 94, 853–863 (1988).
Itoh, Y., Suzuki, H. & Kuriyama, H. J. Pharmac. exp. Ther. 207, 1022–1031 (1978).
Cheung, D. W. & Mackay, M. J. Br. J. Pharmac. 86, 117–124 (1985).
Itoh, Y., Kitamura, K. & Kuriyama, H. Br. J. Pharmac. 70, 197–204 (1980).
Palmer, R. M. J., Ashton, D. S. & Moncada, S. Nature 333, 664–666 (1988).
Palmer, R. M. J., Rees, D. D., Ashton, D. S. & Moncada, S. Biochem. biophys. Res. Commun. 153, 1251–1256 (1988).
Sakuma, I., Stuehr, D. J., Gross, S. S., Nathan, C. & Levi, R. Proc. natn. Acad. Sci. U.S.A. 85, 8664–8667 (1988).
Palmer, R. M. J. & Moncada, S. Biochem. biophys. Res. Commun. 158, 345–352 (1989).
Rees, D. D., Palmer, R. M., Hodson, H. F. & Moncada, S. Br. J. Pharmac. 96, 418–424 (1989).
Itoh, Y., Kitamura, H. & Kuriyama, H. J. Physiol., Lond. 309, 171–183 (1980).
Beny, J. L. & Burnet, P. C. Blood Vessels 25, 308–311 (1988).
Huang, A. H., Busse, R. & Bassenge, E. Naunyn-Schmeideberg, Archs Pharmac. 338, 438–442 (1988).
Mulvany, M. J. & Halpern, W. Circulation Res. 41, 19–26 (1977).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tare, M., Parkington, H., Coleman, H. et al. Hyperpolarization and relaxation of arterial smooth muscle caused by nitric oxide derived from the endothelium. Nature 346, 69–71 (1990). https://doi.org/10.1038/346069a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/346069a0
This article is cited by
-
Multidrug-resistant protein inhibitor and phosphodiesterase inhibitor potentiate the vasodilator effect induced by photobiomodulation in isolated aortic rings
Lasers in Medical Science (2022)
-
Functional and molecular characterization of endothelium-dependent and endothelium-independent relaxant pathways in uterine artery of non-pregnant buffaloes
Naunyn-Schmiedeberg's Archives of Pharmacology (2020)
-
Molecular control of nitric oxide synthesis through eNOS and caveolin-1 interaction regulates osteogenic differentiation of adipose-derived stem cells by modulation of Wnt/β-catenin signaling
Stem Cell Research & Therapy (2016)
-
Intracellular Acidification Alters Myogenic Responsiveness and Vasomotion of Mouse Middle Cerebral Arteries
Journal of Cerebral Blood Flow & Metabolism (2014)
-
Sodium nitroprusside activates potassium channels in the vena cava in normotensive but not in hypertensive rats
Hypertension Research (2013)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.