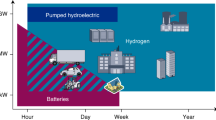
Abstract
Mobility — the transport of people and goods — is a socioeconomic reality that will surely increase in the coming years. It should be safe, economic and reasonably clean. Little energy needs to be expended to overcome potential energy changes, but a great deal is lost through friction (for cars about 10 kWh per 100 km) and low-efficiency energy conversion. Vehicles can be run either by connecting them to a continuous supply of energy or by storing energy on board. Hydrogen would be ideal as a synthetic fuel because it is lightweight, highly abundant and its oxidation product (water) is environmentally benign, but storage remains a problem. Here we present recent developments in the search for innovative materials with high hydrogen-storage capacity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Winter, C. J. & Nitsch, J. Hydrogen as an Energy Carrier: Technologies, Systems, Economy (Springer, 1988).
Bain, A. & Van Vorst, W. D. Int. J. Hydrogen Energy 24, 399–403 (1999).
Shell Hydrogen, Hydro-Québec (HQ) & Gesellschaft für Elektrometallurgie (GfE). Hydrogen storage joint venture to be established. 〈http://www.shell.com〉 Press release (12-07-2001).
Nellis, W. J., Louis, A. A. & Ashcroft, N. W. Metallization of fluid hydrogen. Phil. Trans. R. Soc. Lond. A 356, 119–135 (1998).
Orimo, S.-I. et al. Hydrogen in the mechanically prepared nanostructured graphite. Appl. Phys. Lett. 75, 3093 (1999).
Orimo, S., Matsushima, T., Fujii, H., Fukunaga, T. & Majer, G. Defective carbon for hydrogen storage—thermal desorption property of the mechanically prepared nanostructured graphite. J. Appl. Phys. (in the press).
Stan, G. & Cole, M. W. Hydrogen adsorption in nanotubes. J. Low Temp. Phys. 110, 539–544 (1998).
Hirscher, M. (ed.) Hydrogen storage in nanoscale carbon and metals. Appl. Phys. A (special issue) 72, 2 (2001).
Sholl, C. A. & Gray, E. MacA. (eds) Proc. Int. Symp. Metal Hydrogen Systems—Fundamentals and Applications, Noosa, Australia, 1–6 October 2000. J. Alloys Compounds (in the press).
Chambers, A., Park, C., Baker, R. T. K. & Rodriguez, N. M. Hydrogen storage in graphite nanofibers. Phys. Chem. B 102, 4253–4256 (1998).
Dillon, A. C. et al. Storage of hydrogen in single-walled carbon nanotubes. Nature 386, 377–379 (1997).
Dillon, A. C. et al. Carbon nanotube materials for hydrogen storage. Proc. 2000 DOE/NREL Hydrogen program review, 8–10 May 2000.
Hirscher, M. et al. Hydrogen storage in sonicated carbon materials. Appl. Phys. A 72, 129–132 (2001).
Züttel A. et al. Hydrogen sorption by carbon nanotubes and other carbon nanostructures. J. Alloys Compounds (in the press).
Chen, P., Wu, X., Lin, J. & Tan, K. L. High H2 uptake by alkali-doped carbon nanotubes under ambient pressure and moderate temperatures. Science 285, 91–93 (1999).
Hirscher, M. et al. Hydrogen storage in carbon nanostructures. J. Alloys Compounds (in the press).
Nijkamp, M. G., Raaymakers, J. E. M. J., Van Dillen, A. J. & De Jong, K. P. Hydrogen storage using physisorption—materials demands. Appl. Phys. A 72, 619–623 (2001).
Züttel, A. et al. Hydrogen storage in carbon nanostructures. Int. J.Hydrogen Energy (in the press).
Enoki, T., Shindo, K. & Sakamoto, N. Electronic properties of alkali-metal-hydrogen-graphite intercalation compounds. Z. Phys. Chem. 181, 75–82 (1993).
Schlapbach, L. (ed.) Hydrogen in Intermetallic Compounds I. Electronic, Thermodynamic, and Crystallographic Properties, Preparation (Topics in Applied Physics Vol. 63) (Springer, 1988).
Schlapbach, L. (ed.) Hydrogen in Intermetallic Compounds II. Surface and Dynamic Properties, Applications (Topics in Applied Physics Vol. 67) (Springer, 1992).
Sandrock, G. & Thomas, G. The IEA/DOC/SNL on-line hydride databases. Appl. Phys. A 72, 153–155 (2001).
Sakai, T., Natsuoka, M. & Iwakura, C. Rare earth intermetallics for metal–hydrogen batteries. Handb. Phys. Chem. Rare Earths 21, 135–180 (1995).
Latroche, M., Percheron-Guegan, A. & Chabre, Y. Influence of cobalt content in MmNi(4.3–x)Mn0.3Al0.4Cox alloy (x = 0.36 and 0.69) on its electrochemical behaviour studied by in situ neutron diffraction. J. Alloys Compounds 295, 637–642 (1999).
Schlapbach, L., Felix Meli, F., Züttel, A., Westbrook, J. H. & Fleischer, R. L. (eds) in Intermetallic Compounds: Principles and Practice Vol. 2, Ch. 22 (Wiley, 1994).
Zaluska, A., Zaluski, L. & Stroem-Olsen, J. O. Structure, catalysis and atomic reactions on the nano-scale: a systematic approach to metal hydrides for hydrogen storage. Appl. Phys. A 72, 157 (2001).
Yvon, K. Complex transition metal hydrides. Chimia 52, 613–619 (1998).
Liu, F. J. & Suda, S. A method for improving the long-term storability of hydriding alloys by air water exposure. J. Alloys Compounds 231, 742–750 (1995).
Akiba, E. & Iba, H. Hydrogen absorption by Laves phase related BCC solid solution. Intermetallics 6, 461–470 (1998).
Kuriiwa, T. et al. New V-based alloys with high protium absorption and desorption capacity. J. Alloys Compounds 295, 433–436 (1999).
Tsukahara, M. et al. Hydrogen storage and electrode properties of V-based solid solution type alloys prepared by a thermic process. J. Electrochem. Soc. 147, 2941–2944 (2000).
Inoue, H. et al. Effect of ball-milling with Ni and Raney Ni on surface structural characteristics of TiV2.1Ni0.3 alloy. J. Alloys Compounds 325, 299–303 (2001).
Bogdanovic, B. & Schwickardi, M. Ti-doped alkali metal aluminium hydrides as potential novel reversible hydrogen storage materials. J. Alloys Compounds 253, 1–9 (1997).
Acknowledgements
We thank the Swiss Federal Office of Energy (BFE), in contract with IEA, the Swiss Federal Office of Education and Science (BBW), and the University of Fribourg and EMPA for support of our hydrogen-storage research projects.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schlapbach, L., Züttel, A. Hydrogen-storage materials for mobile applications. Nature 414, 353–358 (2001). https://doi.org/10.1038/35104634
Issue Date:
DOI: https://doi.org/10.1038/35104634
This article is cited by
-
Microstructure and hydrogen storage properties of the Mg2−xYxNi0.9Co0.1 (x = 0, 0.2, 0.3, and 0.4) alloys
Scientific Reports (2024)
-
Reversible and high-density energy storage with polymers populated with bistable redox sites
Polymer Journal (2024)
-
Connectionist technique estimates of hydrogen storage capacity on metal hydrides using hybrid GAPSO-LSSVM approach
Scientific Reports (2024)
-
Optimizing FeCoNiCrTi high-entropy alloy with hydrogen pumping effect to boost de/hydrogenation performance of magnesium hydride
Rare Metals (2024)
-
Enhanced hydrogen storage performance of g-C3N4/CoMn2O4 nanocomposites
Journal of Materials Science: Materials in Electronics (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.