Abstract
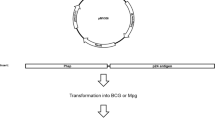
SEVERAL viral and bacterial live recombinant vaccine vehicles are being developed to produce a new generation of vaccines against a broad spectrum of infectious diseases1. The human tuberculosis vaccine Mycobacterium bovis bacillus Calmette–Guerin (BCG)2 has features that make it a particularly attractive live recombinant vaccine vehicle. BCG and other mycobacteria are highly effective adjuvants, and the immune response to mycobacteria has been studied extensively. With nearly two billion immunizations, BCG has a long record of safe use in man3,4. It is one of the few vaccines that can be given at birth, it engenders long-lived immune responses with only a single dose, and there is a worldwide distribution network with experience in BCG vaccination. Recently developed molecular genetic tools and methods for mycobacteria have provided the means to introduce foreign genes into BCG5–8. Here we report that a variety of human immunodeficiency virus type 1 polypeptides can be expressed in BCG recombinants under the control of the mycobacterial hspTO promoter and that the foreign polypeptides produced in BCG can induce antibody and T-cell responses. These results demonstrate that BCG can be used as a live recombinant vaccine vehicle to induce immune responses to pathogen proteins produced by the bacillus.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bloom, B. R. Nature 342, 115–120 (1989).
Calmette, A., Guerin, C. & Weill-Halle, B. Bull. Acad. natn. Med (Paris) 91, 787–796 (1924).
Luelmo, F. Am. Rev. resp. Dis. 125, 70–72 (1982).
Lotte, A. et al. Adv. Tuberc. Res. 21, 107–193 (1984).
Jacobs, W. R. Jr, Truckman, R. & Bloom, B. R. Nature 327, 532–535 (1987).
Snapper, S. B. et al. Proc natn Acad. Sci. U.S.A. 85, 6987–6991 (1988).
Husson, R. N., James, B. E. & Young, R. A. J. Bact. 172, 519–524 (1990).
Martin, C. et al. Nature 345, 739–743 (1990).
Neidhardt, F. C. & VanBogelen, R. A. In Escherichia coli and Salmonella typhimurium Cellular and Molecular Biology (eds Neidhardt, F. C., Ingraham, J. L., Low, K. B., Magasanik, B. & Schaechter, M.) 1654 (American Society for Microbiology, Washington, DC, 1987).
Buchmeier, N. A. & Heffron, F. Science 248, 730–732 (1990).
Young, R. A. A. Rev. Immun 8, 401–420 (1990).
Lamb, J. R. & Young, D. B. Molec biol. Med 7, 311–321 (1990).
Kaufmann, S. H. E. Immun. Today 11, 129–136 (1990).
Mosmann, T. R. & Coffmann, R. L. Adv. Immun. 46, 111–147 (1989).
Swain, S. L., McKenzie, D. T., Weinberg, A. D. & Hancock, W. J. Immun. 141, 3445–3455 (1988).
Mosmann, T. J. Immunol. Meth. 65, 55–63 (1983).
Nagler-Anderson, C., Verret, C. R., Firmenich, A. A., Berne, M. & Eisen, H. N. J. Immun. 141, 3299–3305 (1988).
Walker, B. D. in Techniques in HIV Research (eds Aldovini, A. & Walker, B. D.) 201–210 (Stockton, New York, 1990).
Pierce, E., James, S. L., Heiny, S., Lanar, D. E. & Sher, A. Proc. natn. Acad. Sci. U.S.A. 85, 5678–5682 (1988).
Desrosiers, R. C. et al. Proc. natn. Acad. Sci. U.S.A. 86, 6353–6357 (1989).
Murphey-Corb, M. et al. Science 246, 1293–1297 (1989).
Bloom, B. R. J. Immun 137, 1–10 (1986).
Wyler, D. J., Beller, D. I. & Sypek, J. P. J. Immun. 138, 1246–1249 (1987).
Ratner, L. et al. Nature 313, 277–284 (1985).
Fisher, A. G., Collati, E., Ratner, L., Gallo, R. C. & Wong-Staal, F. Nature 316, 262–265 (1985).
Maniatis, T., Fritsch, E. F. & Sambrook, J. Molecular Cloning: A Laboratory Manual. (Cold Spring Harbor Laboratory, Cold Spring Harbor, New York, 1982).
Laemli, U. K. Nature 227, 680–685 (1970).
Aldovini, A. & Young, R. A. J. Virol. 64, 1920–1926 (1990).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Aldovini, A., Young, R. Humoral and cell-mediated immune responses to live recombinant BCG–HIV vaccines. Nature 351, 479–482 (1991). https://doi.org/10.1038/351479a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/351479a0
This article is cited by
-
Vaccine strategies for the Mtb/HIV copandemic
npj Vaccines (2020)
-
Engineering new mycobacterial vaccine design for HIV–TB pediatric vaccine vectored by lysine auxotroph of BCG
Molecular Therapy - Methods & Clinical Development (2014)
-
Prime-boost vaccination with plasmid DNA and a chimeric adenovirus type 5 vector with type 35 fiber induces protective immunity against HIV
Gene Therapy (2005)
-
Annulling a dangerous liaison: vaccination strategies against AIDS and tuberculosis
Nature Medicine (2005)
-
NALT- versus PEYER'S-patch-mediated mucosal immunity
Nature Reviews Immunology (2004)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.