Abstract
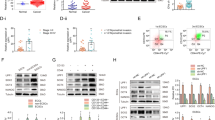
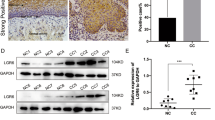
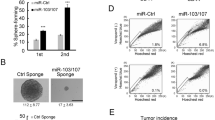
Let-7 miRNAs are involved in carcinogenesis and tumor progression through their roles in maintaining differentiation and normal development. However, there is little research focusing on the effects of let-7 on Wnt-activated self-renewal of breast cancer stem cells. By analyzing the expression levels of let-7 family members in clinical tissues, we found that higher expression levels of let-7b and let-7c were correlated with better clinical prognosis of patients with estrogen receptor (ER)α-positive breast tumor. Further, we found that only let-7c was inversely correlated with ERα expression, and there is corelationship between let-7c and Wnt signaling in clinical tissues. Aldehyde dehydrogenase (ALDH)1 sorting and mammosphere formation assays showed that let-7c inhibited the self-renewal of stem cells in ERα-positive breast cancer. Let-7c decreased ERα expression through directly binding to the 3’UTR (untranslated region), and let-7c inhibited the estrogen-induced activation of Wnt signaling. Depletion of ERα abolished let-7c functions in stem cell signatures, which further confirmed that let-7c inhibited estrogen-induced Wnt activity through decreasing ERα expression. Taken together, our findings identified a biochemical and functional link between let-7c with ERα/Wnt signaling in breast cancer stem cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Croker AK, Allan AL . Inhibition of aldehyde dehydrogenase (ALDH) activity reduces chemotherapy and radiation resistance of stem-like ALDHhiCD44(+) human breast cancer cells. Breast Cancer Res Treat 2012; 133: 75–87.
Sun X, Qin S, Fan C, Xu C, Du N, Ren H . Let-7: a regulator of the ERalpha signaling pathway in human breast tumors and breast cancer stem cells. Oncol Rep 2013; 29: 2079–2087.
Miki T, Yasuda SY, Kahn M . Wnt/beta-catenin signaling in embryonic stem cell self-renewal and somatic cell reprogramming. Stem Cell Rev 2011; 7: 836–846.
Ono M, Yin P, Navarro A, Moravek MB, Coon JS, Druschitz SA et al. Paracrine activation of WNT/β-catenin pathway in uterine leiomyoma stem cells promotes tumor growth. Proc Natl Acad Sci USA 2013; 110: 17053–17058.
Wray J, Hartmann C . WNTing embryonic stem cells. Trends Cell Biol 2012; 22: 159–168.
Anastas JN, Moon RT . WNT signalling pathways as therapeutic targets in cancer. Nat Rev Cancer 2013; 13: 11–26.
Hu X, Guo J, Zheng L, Li C, Zheng TM, Tanyi JL et al. The heterochronic microRNA let-7 inhibits cell motility by regulating the genes in the actin cytoskeleton pathway in breast cancer. Mol Cancer Res 2013; 11: 240–250.
Sun X, Fan C, Du N, Ren H . Possible carcinogenesis of tumor suppressor let-7. Med Hypotheses 2013; 81: 410–413.
Sun X, Tang S-C, Xu C, Wang C, Qin S, Du N et al. DICER1 regulated let-7 expression levels in p53-induced cancer repression requires cyclin D1. J Cell Mol Med 2015; 19: 1357–1365.
Sun X, Jiao X, Pestell TG, Fan C, Qin S, Mirabelli E et al. MicroRNAs and cancer stem cells: the sword and the shield. Oncogene 2014; 33: 4967–4977.
Sun X, Jiang S, Liu J, Wang H, Zhang Y, Tang SC et al. MiR-208a stimulates the cocktail of SOX2 and beta-catenin to inhibit the let-7 induction of self-renewal repression of breast cancer stem cells and formed miR208a/let-7 feedback loop via LIN28 and DICER1. Oncotarget 2015; 6: 32944–32954.
Trang P, Wiggins JF, Daige CL, Cho C, Omotola M, Brown D et al. Systemic delivery of tumor suppressor microRNA mimics using a neutral lipid emulsion inhibits lung tumors in mice. Mol Ther 2011; 19: 1116–1122.
Cai WY, Wei TZ, Luo QC, Wu QW, Liu QF, Yang M et al. The Wnt-beta-catenin pathway represses let-7 microRNA expression through transactivation of Lin28 to augment breast cancer stem cell expansion. J Cell Sci 2013; 126 (Pt 13): 2877–2889.
Malanchi I, Santamaria-Martinez A, Susanto E, Peng H, Lehr HA, Delaloye JF et al. Interactions between cancer stem cells and their niche govern metastatic colonization. Nature 2012; 481: 85–89.
Liu M, Casimiro MC, Wang C, Shirley LA, Jiao X, Katiyar S et al. p21CIP1 attenuates Ras- and c-Myc-dependent breast tumor epithelial mesenchymal transition and cancer stem cell-like gene expression in vivo. Proc Natl Acad Sci USA 2009; 106: 19035–19039.
Wu K, Li Z, Cai S, Tian L, Chen K, Wang J et al. EYA1 phosphatase function is essential to drive breast cancer cell proliferation through cyclin D1. Cancer Res 2013; 73: 4488–4499.
Casimiro MC, Wang C, Li Z, DiSante G, Willmart NE, Addya S et al. Cyclin D1 determines estrogen signaling in the mammary gland in vivo. Mol Endocrinol 2013; 27: 1415–1428.
Wang T, Liu Z, Guo S, Wu L, Li M, Yang J et al. The tumor suppressive role of CAMK2N1 in castration-resistant prostate cancer. Oncotarget 2014; 5: 3611–3621.
Yu Z, Wang L, Wang C, Ju X, Wang M, Chen K et al. Cyclin D1 induction of Dicer governs microRNA processing and expression in breast cancer. Nat Commun 2013; 4: 2812.
Liu H, Liang Y, Li Y, Wang J, Wu H, Wang Y et al. Gene silencing of BAG-1 modulates apoptotic genes and sensitizes lung cancer cell lines to cisplatin-induced apoptosis. Cancer Biol Ther 2010; 9: 832–840.
He J, Wang M, Jiang Y, Chen Q, Xu S, Xu Q et al. Chronic arsenic exposure and angiogenesis in human bronchial epithelial cells via the ROS/miR-199a-5p/HIF-1alpha/COX-2 pathway. Environ Health Perspect 2014; 122: 255–261.
Wang T, Guo S, Liu Z, Wu L, Li M, Yang J et al. CAMK2N1 inhibits prostate cancer progression through androgen receptor-dependent signaling. Oncotarget 2014; 5: 10293–10306.
Chen K, Wu K, Gormley M, Ertel A, Wang J, Zhang W et al. Acetylation of the cell-fate factor Dachshund determines p53 binding and signaling modules in breast cancer. Oncotarget 2013; 4: 923–935.
Zhao Y, Deng C, Wang J, Xiao J, Gatalica Z, Recker RR et al. Let-7 family miRNAs regulate estrogen receptor alpha signaling in estrogen receptor positive breast cancer. Breast Cancer Res Treat 2011; 127: 69–80.
Zhou J, Wang C, Wang Z, Dampier W, Wu K, Casimiro MC et al. Attenuation of Forkhead signaling by the retinal determination factor DACH1. Proc Natl Acad Sci USA 2010; 107: 6864–6869.
Taurin S, Sandbo N, Yau DM, Sethakorn N, Dulin NO . Phosphorylation of beta-catenin by PKA promotes ATP-induced proliferation of vascular smooth muscle cells. Am J Physiol Cell Physiol 2008; 294: C1169–C1174.
Sun Y, Wang Y, Fan C, Gao P, Wang X, Wei G et al. Estrogen promotes stemness and invasiveness of ER-positive breast cancer cells through Gli1 activation. Mol Cancer 2014; 13: 137.
Ginestier C, Hur MH, Charafe-Jauffret E, Monville F, Dutcher J, Brown M et al. ALDH1 is a marker of normal and malignant human mammary stem cells and a predictor of poor clinical outcome. Cell Stem Cell 2007; 1: 555–567.
Liu SY, Zheng PS . High aldehyde dehydrogenase activity identifies cancer stem cells in human cervical cancer. Oncotarget 2013; 4: 2462–2475.
Cicalese A, Bonizzi G, Pasi CE, Faretta M, Ronzoni S, Giulini B et al. The tumor suppressor p53 regulates polarity of self-renewing divisions in mammary stem cells. Cell 2009; 138: 1083–1095.
Wu K, Jiao X, Li Z, Katiyar S, Casimiro MC, Yang W et al. Cell fate determination factor Dachshund reprograms breast cancer stem cell function. J Biol Chem 2011; 286: 2132–2142.
MacDonald BT, Tamai K, He X . Wnt/β-catenin signaling: components, mechanisms, and diseases. Dev Cell 2009; 17: 9–26.
Amado NG, Predes D, Moreno MM, Carvalho IO, Mendes FA, Abreu JG . Flavonoids and Wnt/β-catenin signaling: potential role in colorectal cancer therapies. Int J Mol Sci 2014; 15: 12094–12106.
Merlo DF, Ceppi M, Filiberti R, Bocchini V, Znaor A, Gamulin M et al. Breast cancer incidence trends in European women aged 20–39 years at diagnosis. Breast Cancer Res Treat 2012; 134: 363–370.
Ferlay J, Shin H-R, Bray F, Forman D, Mathers C, Parkin DM . Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 2010; 127: 2893–2917.
Hermann PC, Bhaskar S, Cioffi M, Heeschen C . Cancer stem cells in solid tumors. Semin Cancer Biol 2010; 20: 77–84.
Velasco-Velázquez MA, Homsi N, De La Fuente M, Pestell RG . Breast cancer stem cells. Int J Biochem Cell Biol 2012; 44: 573–577.
Liu C, Tang DG . MicroRNA regulation of cancer stem cells. Cancer Res 2011; 71: 5950–5954.
Parashurama N, Lobo NA, Ito K, Mosley AR, Habte FG, Zabala M et al. Remodeling of endogenous mammary epithelium by breast cancer stem cells. Stem Cells 2012; 30: 2114–2127.
Jiao X, Katiyar S, Willmarth NE, Liu M, Ma X, Flomenberg N et al. c-Jun induces mammary epithelial cellular invasion and breast cancer stem cell expansion. J Biol Chem 2010; 285: 8218–8226.
Song SuJ, Poliseno L, Song Min S, Ala U, Webster K, Ng C et al. MicroRNA-antagonism regulates breast cancer stemness and metastasis via TET-family-dependent chromatin remodeling. Cell 2013; 154: 311–324.
Sun X, Fan C, Hu LJ, Du N, Xu CW, Ren H . Role of let-7 in maintaining characteristics of breast cancer stem cells. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi 2012; 28: 789–792.
Liu S, Cong Y, Wang D, Sun Y, Deng L, Liu Y et al. Breast cancer stem cells transition between epithelial and mesenchymal states reflective of their normal counterparts. Stem Cell Rep 2014; 2: 78–91.
Kwon M . Epithelial-to-mesenchymal transition and cancer stem cells: emerging targets for novel cancer therapy. Cancer Gene Ther 2014; 21: 179–180.
Gan XQ, Wang JY, Xi Y, Wu ZL, Li YP, Li L . Nuclear Dvl, c-Jun, beta-catenin, and TCF form a complex leading to stabilization of beta-catenin-TCF interaction. J Cell Biol 2008; 180: 1087–1100.
Taurin S, Sandbo N, Yau DM, Sethakorn N, Dulin NO . Phosphorylation of β-catenin by PKA promotes ATP-induced proliferation of vascular smooth muscle cells. Am J Physiol Cell Physiol 2008; 294: C1169–C1174.
Acknowledgements
The authors acknowledge assistants in the Key Laboratory (Ministry of Education) of Environment and Genes Related to Disease, and the Center for Translational Medicine of The First Affiliated Hospital of Xi’an Jiaotong University, for their technical assistance. This experiment was mainly supported by National Natural Science Foundation of China, grant no. 81272418 (HR). This work was also supported in part by National Science Foundation for Young Scientists of China, grant no. 81502227 (SHX) and 81402506 (SDQ). All co-authors implicated in this research approved this article to be published.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Cancer Gene Therapy website
Rights and permissions
About this article
Cite this article
Sun, X., Xu, C., Tang, SC. et al. Let-7c blocks estrogen-activated Wnt signaling in induction of self-renewal of breast cancer stem cells. Cancer Gene Ther 23, 83–89 (2016). https://doi.org/10.1038/cgt.2016.3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2016.3
This article is cited by
-
M6A associated TSUC7 inhibition contributed to Erlotinib resistance in lung adenocarcinoma through a notch signaling activation dependent way
Journal of Experimental & Clinical Cancer Research (2021)
-
The immunotherapy candidate TNFSF4 may help the induction of a promising immunological response in breast carcinomas
Scientific Reports (2021)
-
Mir-let-7a/g Enhances Uterine Receptivity via Suppressing Wnt/β-Catenin Under the Modulation of Ovarian Hormones
Reproductive Sciences (2020)
-
Prognostic value of Lin28A and Lin28B in various human malignancies: a systematic review and meta-analysis
Cancer Cell International (2019)
-
CCAT1 stimulation of the symmetric division of NSCLC stem cells through activation of the Wnt signalling cascade
Gene Therapy (2018)