Abstract
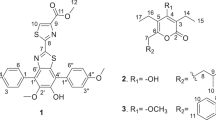
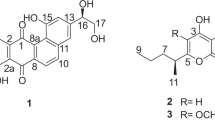
Three new naphtho-γ-pyrones, 5-hydroxy-6,8-dimethoxy-2-hydroxymethyl-4H-naphtho[2,3-b]pyran-4-one (1, nigerasperone A), 3,3′-dihydro-2,2′,5,5′-tetrahydroxy-8,8′,10,10′- tetramethoxy-2,2′-dimethyl-(6′,9-bi-4H-naphtho[1,2-b]pyran)-4,4′-dione (2, nigerasperone B), and 3′-hydro-2′,5,5′,8-tetrahydroxy-6,6′,8′-trimethoxy-2,2′-dimethyl-(7,10′-bi-4H-naphtho[2,3-b]pyran)-4,4′-dione (3, nigerasperone C), together with nine related known compounds were characterized from Aspergillus niger EN-13, an endophytic fungus isolated from the marine brown alga Colpomenia sinuosa. Their structures were elucidated by detailed analysis of spectroscopic data and by comparison with literature reports. In the cytotoxic assay, these compounds did not show remarkable inhibitory effects against A549 and SMMC-7721 tumor cell lines. However, 3 and several known compounds showed weak antifungal activity against Candida albicans and moderate activity on DPPH scavenging.
Similar content being viewed by others
Article PDF
References
Varoglu M, Corbett TH, Valeriote FA, Crews P . Asperazine, a selective cytotoxic alkaloid from a sponge-derived culture of Aspergillus niger. J Org Chem 62: 7078–7079 ( 1997)
Hiort J, Maksimenka K, Reichert M, Perovic-Ottstadt S, Lin WH, Wray V, Steube K, Schaumann K, Weber H, Proksch P, Ebel R, Muller WEG, Bringmann G . New natural products from the sponge-derived fungus Aspergillus niger. J Nat Prod 67: 1532–1543 ( 2004)
Bugni TS, Abbanat D, Bernan VS, Maiese WM, Greenstein M, Wagoner RMV, Ireland CM . Yanuthones: novel metabolites from a marine isolate of Aspergillus niger. J Org Chem 65: 7195–7200 ( 2000)
Ui H, Shiomi K, Yamaguchi Y, Masuma R, Nagamitsu T, Takano D, Sunazuka T, Namikoshi M, Ōmura S . Nafuredin, a novel inhibitor of NADH-fumarate reductase, produced by Aspergillus niger FT-0554. J Antibiot 54: 234–238 ( 2001)
Priestap HA . New naphthopyrones from Aspergillus fonsecaeus. Tetrahedron 40: 3617–3624 ( 1984)
Ehrlich KC, Delucca II AJ, Ciegler A . Naphtho-γ-pyrone production by Aspergillus niger isolated from stored cottonseed. Appl Environ Microbiol 48: 1–4 ( 1984)
Akiyama K, Teraguchi Y, Hamasaki Y, Mori M, Tatsumi K, Ohnishi K, Hayashi H . New dimeric naphthopyrones from Aspergillus niger. J Nat Prod 66: 136–139 ( 2003)
Skehan P, Storeng R, Scudiero D, Monks A, McMahon J, Vistica D, Warren JT, Bokesch H, Kenney S, Boyd MR . New colorimetric cytotoxicity assay for anticancer-drug screening. J Natl Cancer Inst 82: 1107–1112 ( 1990)
Al-Burtamani SKS, Fatope MO, Marwah RG, Onifade AK, Al-Saidi SH . Chemical composition, antibacterial and antifungal activities of the essential oil of Haplophyllum tuberculatum from Oman. J Ethnopharmacol 96: 107–112 ( 2005)
Duan XJ, Zhang WW, Li XM, Wang BG . Evaluation of antioxidant property of extract and fractions obtained from a red alga, Polysiphonia urceolata. Food Chem 95: 37–43 ( 2006)
Priestap HA . 13C NMR spectroscopy of naphtho-γ-pyrones. Magn Reson Chem 24: 875–878 ( 1986)
Galmarini OL, Stodola FH . Fonsecin, a pigment from an Aspergillus fonsecaeus Mutant. J Org Chem 30: 112–115 ( 1965)
Tanaka H, Wang PL, Yamada O, Tamura T . Yellow pigments of Aspergillus niger and A. awamori. Part I. Isolation of aurasperone A and related pigments. Agric Biol Chem 30: 107–113 ( 1966)
Galmarini OL, Mastronardi IO, Priestap HA . Two novel metabolites of Aspergillus fonsecaeus. Experientia 30: 586 ( 1974)
Ghosal S, Biswas K, Chakrabarti DK . Toxic naphtho-γ-pyrones from Aspergillus niger. J Agric Food Chem 27: 1347–1351 ( 1979)
Bouras N, Mathieu F, Coppel Y, Lebrihi A . Aurasperone F-a new member of the naphtho-gamma-pyrone class isolated from a cultured microfungus, Aspergillus niger C-433. Nat Prod Res 19: 653–659 ( 2005)
Ernst-Russell MA, Chai CLL, Wardlaw JH, Elix JA . Euplectin and coneuplectin, new naphthopyrones from the lichen Flavoparmelia euplecta. J Nat Prod 63: 129–131 ( 2000)
Coelho RG, Vilegas W, Devienne KF, Raddi MSG . A new cytotoxic naphthopyrone dimer from Paepalanthus bromelioides. Fitoterapia 71: 497–500 ( 2000)
Barbosa FG, Oliveira MCF, Braz-Filho R, Silveira ER . Anthraquinones and naphthopyrones from Senna rugosa. Biochem Syst Ecol 32: 363–365 ( 2004)
Song YC, Li H, Ye YH, Shan CY, Yang YM, Tan RX . Endophytic naphthopyrone metabolites are co-inhibitors of xanthine oxidase, SW1116 cell and some microbial growths. FEMS Microbiol Lett 241: 67–72 ( 2004)
Sakurai M, Kohno J, Yamamoto K, Okuda T, Nishio M, Kawano K, Ohnuki T . TMC-256A1 and C1, new inhibitors of IL-4 signal transduction produced by Aspergillus niger var. niger TC 1629. J Antibiot 55: 685–692 ( 2002)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, Y., Li, XM. & Wang, BG. Nigerasperones A~C, New Monomeric and Dimeric Naphtho-γ-pyrones from a Marine Alga-derived Endophytic Fungus Aspergillus niger EN-13. J Antibiot 60, 204–210 (2007). https://doi.org/10.1038/ja.2007.24
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ja.2007.24
Keywords
This article is cited by
-
Antibacterial Activity and AbFtsZ Binding Properties of Fungal Metabolites Isolated from Mexican Mangroves
Revista Brasileira de Farmacognosia (2024)
-
Impact of Fungal Extracts on the Physical and Antioxidant Properties of Bioactive Films Based on Enzymatically Hydrolyzed Yeast Cell Wall
Journal of Polymers and the Environment (2021)
-
Antimicrobial compounds from marine fungi
Phytochemistry Reviews (2021)
-
Asperitaconic acids A–C, antibacterial itaconic acid derivatives produced by a marine-derived fungus of the genus Aspergillus
The Journal of Antibiotics (2018)
-
Safety of the fungal workhorses of industrial biotechnology: update on the mycotoxin and secondary metabolite potential of Aspergillus niger, Aspergillus oryzae, and Trichoderma reesei
Applied Microbiology and Biotechnology (2018)